An Assessment of Relevance of Sputum Sample Examination in Patients with Tuberculous Pleural Effusion
- *Corresponding Author:
- Vishnu Prasad Shenoy
Department of Microbiology, Kasturba Medical College, Manipal, Karnataka, India.
Tel: 9886616197
E-mail:vishnuprasad.shenoy@rediffmail.com
Citation: Shenoy VP, Patra S, Tellapragada C, Shenoy RP, Mukhopadhyay C. An Assessment of Relevance of Sputum Sample Examination in Patients with Tuberculous Pleural Effusion. Ann Med Health Sci Res. 2017; 7:65-68
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix,tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
Abstract
Background: Utility of chest-X ray findings as a reference diagnostic modality in cases of tubercular pleural effusion is well acknowledged. In advent of increasing reports, regarding the absence of visible lung lesions among patients suffering with tubercular pleural effusion, reliability on radiographic findings alone is questionable. In this context, need for a more reliable diagnostic tool in addressing this diagnostic challenge arises. Aim: The present study was undertaken to evaluate the relevance and utility of sputum specimen and adenosine deaminase (ADA) level examination in patients with tubercular pleural effusion, having no visible lesions on radiological examination. Materials and Methods: A total 81 cases of suspected tubercular pleural effusion were selected from the inpatient wards of a tertiary care hospital, over a period of eight months (May to December, 2013). Sputum and pleural fluid specimens from all these patients were collected for microbiological investigations such as smear microscopy using Ziehl-Neelsen and Auramine-O staining, mycobacterial culture using conventional method. ADA level was also tested from all pleural fluid specimens. Results: Of the 81 cases, culture positivity from sputum specimens was observed among 13 (16.1%), smear positivity from sputum specimens (3, 3.7%) and smear and culture positivity from pleural fluid specimens (1, 1.23%) cases. High levels of ADA, ranging from 115 IU/L to 203 IU/L were observed among (8, 22.2%) subjects. Conclusion: Our study findings suggest the reliability of smear examination and culture from sputum specimens over pleural fluids specimens and also high sensitivity of ADA as a biomarker in the diagnosis of tubercular pleural effusion.
Keywords
Tuberculous pleural effusion, Mycobacterium tuberculosis, Diagnosis, Adenosine deaminase (ADA)
Introduction
Tuberculosis (TB) presenting in its pulmonary and extra pulmonary forms is one among the major public health concerns in India. Tuberculous pleural effusion is the second common form of extra pulmonary TB after lymphadenopathy.[1,2] Pleural effusion is a consequence of various medical conditions and patients with pleural effusion presents with cough, chest pain and dyspnea that generally increases with daily activities.[3] While the diagnosis of tubercular pleural effusion primarily relies on chest x-ray findings (gold Standard method), adjunct of cytological, microbiological and biochemical testing of the pleural fluid specimens can help in enhancing the case detection. In spite of its invasive nature, pleural biopsy still remains the most effective diagnostic specimen for the tuberculous pleural effusion.[4] Diagnosis of pleural effusion with early TB pose a challenge due to the low culture positivity rate of pleural fluid specimens, leading to delays in diagnosis and treatment. Recently, there is an increase in the number of reports having cases of tubercular pleural effusion with no visible lung lesions. [5,6] In this context, need for a more reliable diagnostic modality for the early diagnosis of tubercular pleural effusion is evident. With this background, we intended to assess the relevance and utility of microbiological examination of sputum specimens among the patients suspected with tubercular pleural effusion with no radiological evidence.
Materials and Methods
Study details
A hospital based prospective study for duration of eight months (May-December) was undertaken. Study population comprised of subjects suspected with tubercular pleural effusion, having no visible lesions on radiological examination. The study protocol was approved by the Institutional ethical committee and an informed written consent was obtained from all the study participants.
Study specimens
Pleural fluid specimens from patients suspected with tubercular pleural effusion were collected during the study period. Two consecutive days sputum samples were also collected from all the study participants using standard specimen collection techniques. Specimens were collected prior to the administration of anti-tubercular therapy.
Microbiological examination of sputum and pleural fluid specimens
Direct microscopy of pleural fluid and sputum specimens was performed using Ziehl-Neelsen and Auramine O fluorescence staining techniques. Stained smears were graded according to RNTCP guidelines.[7] Decontamination and concentration of sputum specimens were done using modified Petroff’s method [8] and pleural fluid specimens were centrifuged and sediments were used for culture on solid media (Lowenstein-Jensen medium). Cultured LJ bottles were incubated at 37°C for upto 8 weeks.[8] Isolated organisms were subjected to Ziehl-Neelsen staining to confirm the isolation of acid fast bacilli. Identification and differentiation of all the isolates were done using rapid immunochromatographic identification test (SD BIOLINE TB Ag MPT64).[9] In addition to the microbiological investigations, levels of adenosine deaminase (ADA) were tested from the pleural fluid specimens using colorimetric method.
Results
During the study period, 81 suspected cases of tubercular pleural effusion with no visible parenchymal lesion on chest X-ray examination were included in the study. Majority (51.8%) of the study subjects were within the age of 30-60 years. Male to female ratio was 1.5:1. Radiological examination revealed: 57 (70.4%) patients with unilateral and 24 (29.6%) patients with bilateral pleural effusion. Table 1 describes all etiologies of pleural effusion in our study.
| Cause of pleural effusion (N = 81) | No. of cases n (%) | Median ADA values | |
| Tuberculosis | Culture confirmed TB | 13 (16.1) | 34 (27, 94) |
| Suspected TB | 10 (12.3) | 45 (25, 117) | |
| Causes other than TB
Other bacterial etiology Congestive heart failure Pulmonary thromboembolism Empyema Malignancy Cirrhosis Others |
13 (16.1) 12 (14.8) 10 (12.3) 9 (11.1) 7 (8.6) 5 (6.2) 2 (2.4) |
34 (27, 79) 35 (26, 62) 33 (27, 43) 23 (22, 33) 35 (25, 61) 34 (32.5, 79.5) 32 (30, 34) |
|
Table 1: Etiologic distribution of pleural effusion with mean ADA values
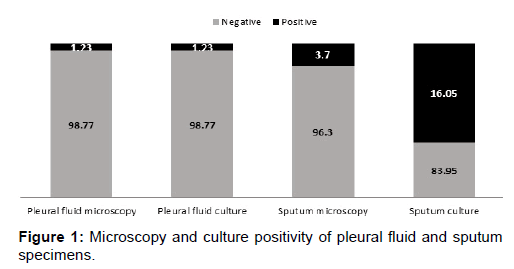
Overall the detection rate of AFB by smear microscopy from sputum specimens in the present study was 3.7% in comparison to the detection rate of 1.23% from pleural fluid. Among the sputum and pleural fluid specimens collected from the 81 subjects, presence of acid fast bacilli from both the specimens was observed in one (1.23%) patient. Smear examination of the sputum specimens could help in the detection of AFB in two more patients whose pleural fluid microscopy revealed no AFB. Culture positivity for M. tuberculosis from pleural fluid specimens was 1.23% (1/81), while the culture positivity rate from sputum specimens was 16.05% (13/81) [Figure 1]. All isolates were confirmed as the members of Mycobacterium tuberculosis complex using MPT 64 antigen detection.
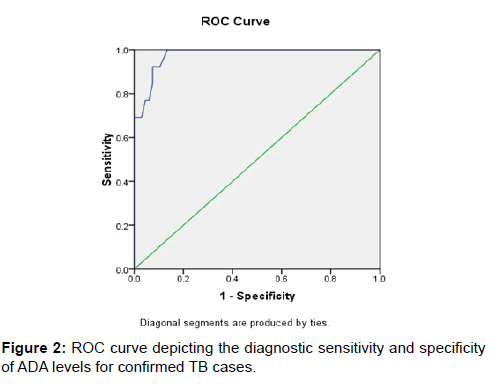
Further, high levels of Adenosine deaminase (ADA) from the plural fluid specimens, ranging between 115-203 IU/L was observed among 18 (22.2%) of the subjects and low levels (<50 IU/L) of ADA among 63 (77.8%) of the study subjects. Using ROC curve analysis, the sensitivity, specificity and area under curve for ADA levels in culture confirmed TB cases was 100%, 84% and 97% with the optimal cutoff limit of 52 IU/L [Figure 2].
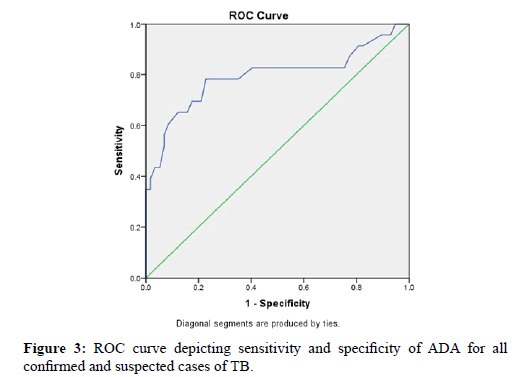
Using ROC curve analysis, the sensitivity, specificity and area under curve for ADA levels in both culture confirmed and clinically suspected cases of TB was 78.3%, 87.2% and 79.3% with the optimal cutoff limit of 44 IU/L [Figure 2].
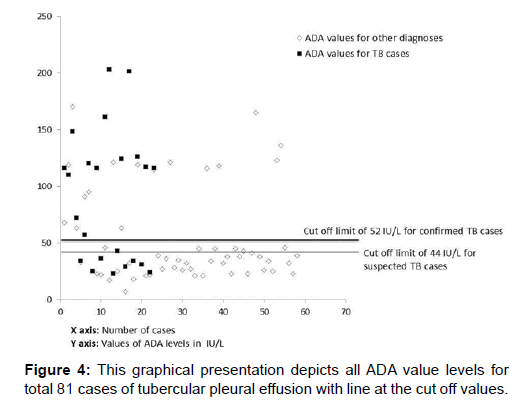
Figure 3 represents all ADA value levels for total 81 cases of tubercular pleural effusion, with a demarcation to visualise the false negative and false positive values [Figure 4].
Discussion
In the present study, we evaluated the utility of microbiological examination of sputum specimens in adjunct to the pleural fluid examination for diagnosis of tubercular pleural effusion. From this hospital based study, we observed that the majority (42/81, 51.8%) of suspected patients were within 30-60 years of age. Similar observation regarding the high-incidence of tubercular pleural effusion among adult population with in the age group of 30-59 years was previously reported.[10] However, a study from the United States of America reported the highest incidence among patients older than 65 years of age.[11] From all these study findings including the present study, it can be emphasized that tuberculosis forms an unavoidable differential diagnosis among older patients with pleural effusion.
Microbiological examination of pleural fluid specimens for tubercle bacilli demonstrated a low rate of detectability (1.2%) in the present study. Low detection rate (<30%) of tubercle bacilli either by culture or smear examination of the pleural fluid specimens was reported previously.[4,10] From a similar study which examined the presence of M. tuberculosis using smear microscopy of patients suspected with tubercular pleural effusion, detection rate of 0% from pleural fluids specimens was reported.[12] All these study findings underscore the low sensitivity of microbiological examination of pleural fluid specimens alone in the diagnosis of tubercular pleural effusion. Smear and culture positivity rates of 3.7% and 16.2% respectively for M. tuberculosis using sputum specimens as observed in our study can be matched with the 22% of culture positivity reported previously from sputum specimens.[4] Detection of Mycobacteria by microscopy and culture in pleural effusion patients was enhanced by examining pleural fluid as well as sputum specimen. Another study compared three detection methods and showed detection of Mycobacterium tuberculosis was enhanced by the Nested PCR assay in 70.6% of the samples compared to 22.4% by culture and 10.3% by AFB smear microscopy.[13]
With a diagnostic cut off of 36 IU/L, the sensitivity and specificity of ADA levels for the diagnosis of TB pleural effusion was reported as 100% and 77% respectively.[14] In our study, the sensitivity and specificity of ADA levels was 100% and 84% respectively analyzed through ROC Curve analysis, when the diagnostic cut off level was 52 IU/L. Among the 81 subjects recruited in the study, 18 (22%) of them had ADA levels suggestive of Tubercular pleural effusion. Among the 18 subjects with elevated ADA levels in our study, microbiological evidence in the form of M. tuberculosis smear or culture positivity was observed in 13 (72.2%) patients. On contrary, none of the patients with lower levels of ADA than the diagnostic cut off (52 IU/L) had either smear or culture positivity for tubercle bacilli. Our study findings emphasize the high sensitivity of ADA as a biomarker for the diagnosis of TB pleural effusion. However, we also foresee the probability of over diagnosis of TB pleural effusion when the diagnosis is solely based on elevated ADA levels due to its lower diagnostic specificity.
Diagnostic accuracies of sputum and pleural fluid microbiological examination for the diagnosis of tubercular pleural effusion could not be estimated. Lack of power in the sample size could be attributed for this limitation. Despite this limitation, our study findings could underscore the utility of microbiological examination of sputum specimens for the diagnosis of tubercular pleural effusion.
Acknowledgement
This study was supported by Manipal University, Manipal. We thank department of microbiology, Kasturba medical college for providing us all the facilities and resources.
References
- Light RW. Update on tuberculous pleural effusion. Respirology. 2010;15:451-8.
- Sharma SK, Mohan A. Extrapulmonary tuberculosis. Indian J Med Res. 2004;120:16-353.
- Yu H. Management of pleural effusion, empyema, and lung abscess. Semin Intervent Radiol. 2011;28:75-86.
- Mihmanli A, Ozseker F, Baran A, K���ker F, Atik S, Akkaya E. Evaluation of 105 cases with tuberculous pleurisy. TuberkToraks. 2004;52:137-44.
- Chaudhuri AD, Bhuniya S, Pandit S, Dey A, Mukherjee S, Bhanja P. Role of sputum examination for acid fast bacilli in tuberculous pleural effusion. Lung India. 2011;28:21-4.
- Ghosal AG, Ghosh S, Guhathakurta R, Sinha A, Banerjee AK, Chakrabarti A K. Sputum AFB positivity in tuberculous pleural effusion with no radiologically apparent parenchymal lung lesion. Ind J Tub. 1997;44:13-5.
- Manual for sputum smear fluorescens microscopy. Central TB Division. Directorate general of health services, ministry of health and family welfare. India.
- Sharma M, Misra RN, Gandham NR, Jadhav SV, Wilson V. Comparison of modified Petroff?s and N-acetyl-L-cysteine-sodium hydroxide method for sputum decontamination in tertiary care hospital in India. Medical Journal of Dr. D.Y.Patil University. 2012;5:97-100.
- Seiscento M, Conde MB, Dalcolmo MM. Tuberculous pleural effusions. J Bras Pneumol. 2006;4:174-81.
- Seiscento M, Vargas FS, Rujula MJ, Bombarda S, Uip DE, Galesi VM. Epidemiological aspects of pleural tuberculosis in the state of S�o Paulo, Brazil (1998-2005). J Bras Pneumol. 2009;35:548-54.
- Gopi A, Madhavan SM, Sharma SK, Sahn SA. Diagnosis and treatment of tuberculous pleural effusion in 2006. Chest. 2007;131:880-9.
- Kumar P, Sen MK, Chauhan DS, Katoch VM, Singh S, Prasad HK. Assessment of the N-PCR assay in diagnosis of pleural tuberculosis: Detection of M.tuberculosis in pleural fluid and sputum collected in tandem. PLoS One. 2010;5:e10220.
- Baumann MH, Nolan R, Petrini M, Lee YC, Light RW, Schneider E. Pleural tuberculosis in the United States: incidence and drug resistance. Chest. 2007;131:1125-32.
- Verma SK, Dubey AL, Singh PA, Tewerson SL, Sharma D. Adenosine deaminase (ADA) Level in tubercular pleural effusion. Lung India. 2008;25:109-10.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.