Do Clinical Symptoms and Signs Predict Reduced Renal Function Among Hospitalized Adults?
- *Corresponding Author:
- Dr. Sunil Kumar
Department of Medicine, Jawahar Lal Nehru Medical College, DMIMS, Sawangi, Wardha, Maharashtra, India.
E-mail: sunilkumarmed@gmail.com
Abstract
Background: Reduced renal function manifests as reduced glomerular filtration rate (GFR), which is estimated using the serum creatinine levels. This condition is frequently encountered among hospitalized adults. Renal dysfunction remains clinically asymptomatic, until late in the course of disease, and its symptoms and screening strategies are poorly defined. Aim: We conducted this study to understand if the presence of renal dysfunction related clinical symptom and signs (either alone or in combination) can predict reduced GFR. Further, we aimed to determine if the combination of symptoms and signs are useful for prediction of different levels of reduced GFR. Subjects and Methods: We performed a cross‑sectional clinical prediction study and included all consecutive patients admitted to the medical wards of the hospital. We used a renal dysfunction related clinical predictors as index tests and low estimated GFR ([eGFR] < 60 ml/min/1.73 m2) as a reference standard. We identified symptoms with a high likelihood ratio (LR) for prediction of low eGFR and constructed different risk score models. We plotted receiver operating curves for each score and used area under the curve (AUC) for comparison. The score with the highest AUC was considered as most discriminant. All statistical analysis was performed using the statistical software STATA (version 11.0, lake drive, Texas, USA). Results: A total of 341 patients participated in the study. None of the predictor variables had statistically significant LRs for eGFR less than 60 ml/min or eGFR less than 30 ml/min. Positive LRs were significant for prediction of eGFR < 15 ml/min for the presence of hypertension, vomiting pruritis, peripheral edema, hyperpigmentation, peripheral neuropathy and severe anemia. The best predictive model for eGFR less than 15 ml/min/1.73 m2, included Age > 45 years, the presence of hypertension, vomiting, peripheral edema, hyperpigmentation, and severe anemia and had AUC of 0.82. Conclusion: Clinical symptoms and signs are poorly predictive of reduced renal function, except for very low eGFR of less than 15 ml/min/1.73 m2.
Keywords
Clinical predictor, Glomerular filtration rate, Renal dysfunction
Introduction
Reduced renal function is commonly encountered as acute, chronic or acute-on-chronic subtypes, manifesting as reduced glomerular filtration rate (GFR). Progressive renal dysfunction results in declining GFR. While a number of clinical symptoms are associated with renal dysfunction, these are largely known to be non-specific, and many of these manifest with advanced dysfunction. If GFR continues to remain low for at least 3 months, individuals are classified to have chronic kidney disease (CKD), which is a global public health problem. It is estimated that approximately 100,000 new cases of end stage renal disease (ESRD) develop annually in India.[1]
Although, it is important to screen for reduced renal function and then to follow-up individuals with reduced GFR to establish chronicity, screening strategies are poorly defined. Current screening strategies are either disease based (example screening individuals with hypertension or diabetes mellitus), or opportunistic (example to systematically screen individuals seeking health-care for any cause). Symptom based screening is more ill-defined and is usually contingent upon thresholds individual physicians have for CKD related symptoms.
Clinical symptom and sign based screening is challenging as renal function is preserved until late in the course of the disease.[2] Symptoms usually associated with long standing renal dysfunction (such as anemia, fatigue, edema, polydipsia, nocturia, anorexia, nausea, vomiting, bone pain, and pruritus) are either nonspecific or are usually seen only during ESRD. We do-not know, which symptoms alone or in combination can best predict presence of renal dysfunction. The research question for the current study is “among adults seeking in-hospital care, does the presence of certain renal dysfunction related symptoms or simple measurements (or combinations thereof) as compared to the absence of such symptoms help accurately predict reduced GFR.”
Subjects and Methods
We performed a cross-sectional study in a teaching hospital in central India. All consecutive adults, admitted to medical wards of the hospital over a 2-month period (July-August 2011) were included in the study. Patients primarily admitted for intensive care, dialysis services, those with previously known CKD kidney disease and those who were in delirium or were comatose were excluded. These exclusions were carried out hence as to minimize the bias in symptom ascertainment either on the part of the interviewer or the respondent. A written informed consent was sought from all eligible participants. The study design was approved by the institutional review board for research in human subjects.
All eligible and consenting participants were administered a structured pre-tested questionnaire translated in local language on the day of their admission by a study investigator (VJ). This questionnaire included the following 18 symptoms and signs:
• Do you currently smoke or consume tobacco products (smoking)?
• Have you consumed alcohol more than once in the past 12 months (alcohol)?
• Were you ever told by a doctor that you have high blood pressure (hypertension)?
• Were you ever told by a doctor that you have diabetes (diabetes)?
• Did you have vomiting, on most days in past 3 months (vomiting)?
• Do you feel pain over any of your limbs, in past 3 months (bone pain)?
• Do you have pain/discomfort over your calves (muscle cramp)?
• Do you feel tired most of the times in past 3 months (fatigue)?
• Do you feel that in past 3 months your appetite has reduced (anorexia)?
• Do you feel thirsty more often than people around you (polydipsia)?
• Do you need to get up two times or more from sleep to pass urine (nocturia)?
• Does your skin feel itchy (pruritus)?
In addition, following simple measurements/clinical signs were recorded
• Presence of pitting on the shin of the tibia after pressing for 30 s with thumb (pitting edema)
• Presence of puffiness of face as evidenced by loss of facial wrinkles (puffiness) ?
• Presence of a knuckle pigmentation (hyperpigmentation)
• Absence of bilateral ankle reflex (peripheral neuropathy)
• Presence of a rub on cardiac auscultation (pericardial rub)
• Presence of hemoglobin level of 7 g/dL or less on automated cell counter as recorded on the patient’s chart (severe anemia).
Response to all of the above items was recorded as either present or absent. The investigator who administered this questionnaire was blinded to serum chemistry or imaging. Another investigator (SK) retrieved electronic hospital records of all included patients and abstracted the on-admission serum creatinine values. GFR for all study participants was estimated using the Modified Diet in Renal Disease equation [3] as follows:
Estimated GFR (eGFR) (ml/min/1.73 m2) =186 × (PCr)−1.154 × (age)−0.203 (×0.742 for women)
Each of the 18 symptoms or signs was coded as a dichotomous variable. We used three meaningful cut-offs for eGFR (less than 15, Less than 30 and less than 60 ml/min/1.73 m2) as reference values for different levels of renal dysfunction. We constructed 2 × 2 tables for each symptom or sign as an index test and at different levels of renal dysfunction. We estimated positive and negative likelihood ratios (LR) and their confidence intervals for each symptom or sign. Since none of the symptoms or signs had a significant positive or negative predictive values at eGFR cut-off of less than 60 or less than 30, we selected those symptoms or signs, which had a significant LR (confidence interval of the LR did not include 1.0), and the point estimate of LR of greater than 2.0. Seven symptoms and signs met these criteria (vomiting, pruritus, hypertension, edema, peripheral neuropathy, hyperpigmentation and anemia). Since age is another important predictor of reduced GFR, in addition to these seven predictors, we used age more than 45 years as eighth variable to construct subsequent predictive models. We used different combinations of these eight variables, in a logistic regression model, with reduced eGFR of less than 15 ml/min/1.73 m2 and less than 30 ml/min/1.73 m2 as reference standards. For each model, we plotted receiver operating curves and used area under the curve (AUC) for comparison. The model with best AUC was considered as most discriminant. All statistical analysis was performed using the statistical software STATA (version 11.0, lake drive, Texas, USA).
Results
A total of 341 patients were included in the study (mean age 35.4 [12.8], 48% women). Of all participants 55.1% (188/341) had eGFR greater than 60 ml/min, and 44.9% (153/341) had renal dysfunction as evidenced by eGFR less than 60 ml/min. A total of 117 34.4% (117/153) individuals had eGFR between 30 ml/min and 60 ml/min, 5.8% (20/153) had eGFR between 15 ml/min and 30 ml/min and 4.7% (16/153) had eGFR of less than 15 ml/min. The distribution of various symptoms and signs is summarized in Table 1
| Clinical variable | All (n=341)(%) | eGFR>60 (n=188)(%) | eGFR 30-60 (n=117)(%) | eGFR15-30 (n=20)(%) | eGFR<15 (n=16)(%) |
|---|---|---|---|---|---|
| Smoking | 75(21.9) | 39(20.7) | 33(28.2) | 2 (10) | 1 (6.2) |
| Alcohol intake | 76(22.2) | 38(20.2) | 31(26.4) | 4 (20) | 3 (18.7) |
| Hypertension | 72(21.1) | 21(11.1) | 37(31.6) | 6 (20) | 8 (50) |
| Diabetes | 48(14.0) | 19(10.1) | 23(19.6) | 3 (15) | 3 (18.7) |
| Vomiting | 88(25.8) | 49(26.0) | 23(19.6) | 6 (30) | 10 (62.5) |
| Bone pain | 112(32.8) | 51(27.1) | 47(40.1) | 8 (40) | 6 (37.5) |
| Muscle cramp | 170(49.8) | 62(32.9) | 84(71.7) | 12(60) | 12 (75) |
| Fatigue | 177(51.9) | 77(40.9) | 78(66.6) | 11(55) | 11 (68.7) |
| Anorexia | 120(35.1) | 60(46.8) | 45(38.4) | 6 (30) | 9 (56.2) |
| Polydipsia | 98(28.7) | 36(19.1) | 46(39.3) | 10(50) | 6 (37.1) |
| Nocturia | 91(26.6) | 32(17.0) | 47(40.1) | 5 (25) | 7 (43.7) |
| Pruritus | 50(14.6) | 17 (9.0) | 25(21.3) | 2 (10) | 6 (37.5) |
| Pedal edema | 66(19.3) | 26(13.8) | 29(24.7) | 3 (15) | 8 (50) |
| Puffiness of face | 17(4.9) | 5(2.6) | 9(7.6) | 1(5) | 2 (12.5) |
| Hyperpigmentation | 23(6.7) | 8(4.2) | 9(7.6) | 1(5) | 5 (31.2) |
| Peripheral neuropathy | 29(8.5) | 8(4.2) | 13(11.1) | 4 (20) | 4 (25) |
| Pericardial rub | 7(2.0) | 2(1.0) | 5(4.2) | 0(0) | 0 (0) |
| Anemia (Hb<7 g/dL) | 153(44.8) | 114 (60.6) | 24(20.5) | 6 (30) | 9 (56.2) |
Hb: Hemoglobin, eGFR: Estimated glomerular filteration rate. eGFR is expressed as ml/min/1.73 m2 body surface area
Table 1: Distribution of clinical features by eGFR
We estimated LRs and their 95% confidence intervals for each of these clinical symptoms and signs, with different levels of reduced eGFR as the reference standard. The positive and negative LRs are summarized in Table 2. None of the predictor variables had statistically significant LRs for eGFR less than 60 ml/min or eGFR less than 30 ml/min. Positive LRs were significant for prediction of eGFR < 15 ml/min for presence of hypertension (2.54 [95% CI 1.48-4.34], vomiting [2.57 (95% CI 1.68-3.93]), pruritis (2.77 [95% CI 1.79-5.52]), peripheral edema (2.80 [95% CI 1.63-4.82]), hyperpigmentation (5.64 [95% CI 2.4-13.3], peripheral neuropathy (3.25 [95% CI 1.28-8.22]) and anemia (Hb < 7 g/dL) (4.64 [95% CI 1.79-12.0]).
| Variables | eGFR less than 60 versus eGFR>60 ml/min/1.73 m2 | eGFR less than 30 versus eGFR>0 ml/min/1.73 m2 | eGFR<15 versus eGFR>15 ml/min/1.73 m2 | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TP | FP | FN | TN | LR+ | LR– | TP | FP | FN | TN | LR+ | LR– | TP | FP | FN | TN | LR+ | LR– | ||
| Smoking | 40 | 35 | 113 | 153 | 1.4 (0.94-2.1) | 0.90 (0.80-1.02) | 7 | 68 | 29 | 237 | 0.87 (0.43-1.75) | 1.04 (0.87-1.23) | 1 | 74 | 15 | 15 | 0.27 (0.04-1.85) | 1.21 (1.06-1.4) | |
| Alcohol | 38 | 38 | 114 | 150 | 1.24 (0.83-1.84) | 0.94 (0.83-1.06) | 7 | 69 | 29 | 235 | 0.85 (0.42-1.72) | 1.04 (0.87-1.24) | 3 | 73 | 13 | 251 | 0.83 (0.29-2.35) | 1.05 (0.82-1.34) | |
| Hypertension | 43 | 29 | 110 | 159 | 1.82 (1.2-2.77) | 0.85 (0.75-0.95) | 14 | 58 | 22 | 247 | 2.05 (1.28-3.27) | 0.75 (0.57-0.98) | 8 | 64 | 8 | 261 | 2.54 (1.48-4.34) | 0.63 (0.38-1.02) | |
| Diabetes | 29 | 19 | 124 | 169 | 1.88 (1.1-3.21) | 0.90 (0.82-0.98) | 6 | 42 | 30 | 263 | 1.21 (0.55-2.65) | 0.96 (0.82-1.13) | 3 | 45 | 13 | 280 | 1.35 (0.47-3.89) | 0.94 (0.74-1.2) | |
| Vomiting | 47 | 42 | 106 | 146 | 1.38 (0.96-0.97) | 0.89 (0.78-1.02) | 16 | 73 | 20 | 232 | 1.86 (1.22-2.82) | 0.73 (0.54-0.98) | 10 | 79 | 6 | 246 | 2.57 (1.68-3.93) | 0.49 (0.26-0.93) 0.93 (0.63-1.37) | |
| Bone pain | 55 | 57 | 98 | 129 | 1.17 (0.86-1.59) | 0.92 (0.79-1.08) | 14 | 98 | 22 | 205 | 1.2 (0.77-1.87) | 0.90 (0.68-1.19) | 6 | 106 | 10 | 217 | 1.14 (0.59-2.19) | ||
| Muscle cramp | 96 | 74 | 57 | 113 | 1.59 (1.28-1.97) | 0.61 (0.48-0.78) | 24 | 146 | 12 | 158 | 1.39 (1.07-1.8) | 0.64 (0.39-1.03) | 12 | 158 | 4 | 166 | 1.54 (1.13-2.08) | 0.48 (0.20-1.15) | |
| Fatigue | 89 | 88 | 64 | 99 | 1.24 (1.01-1.51) | 0.79 (0.62-0.99) | 22 | 155 | 14 | 149 | 1.2 (0.90-1.59) | 0.79 (0.51-1.21) | 11 | 166 | 5 | 158 | 1.34 (0.94-1.9) | 0.64 (0.30-1.34) | |
| Anorexia | 60 | 60 | 93 | 128 | 1.23 (0.92-1.64) | 0.89 (0.76-1.05) | 15 | 105 | 21 | 200 | 1.21 (0.798-1.84) | 0.89 (0.66-1.19) | 9 | 111 | 7 | 214 | 1.65 (1.04-2.6) | 0.66 (0.37-1.16) | |
| Polydypsia | 56 | 42 | 97 | 146 | 1.64 (1.17-2.3) | 0.81 (0.70-0.94) | 16 | 82 | 20 | 223 | 1.65 (1.1-2.49) | 0.76 (0.56-1.03) | 6 | 92 | 10 | 233 | 1.32 (0.68-2.55) | 0.87 (0.59-1.28) | |
| Nocturia | 52 | 39 | 101 | 148 | 1.63 (1.14-2.33) | 0.83 (0.72-00.95) | 12 | 79 | 24 | 225 | 1.28 (0.77-2.11) | 0.90 (0.70-1.15) | 7 | 84 | 9 | 240 | 1.69 (0.94-3.03) | 0.75 (0.49-1.18) | |
| Pruritus | 27 | 23 | 126 | 165 | 1.44 (0.86-2.41) | 0.93 (0.85-1.03) | 8 | 42 | 28 | 263 | 1.61 (0.82-3.16) | 0.90 (0.75-1.08) | 6 | 44 | 10 | 281 | 2.77 (1.39-5.52) | 0.72 (0.49-1.06) | |
| Edema | 40 | 26 | 113 | 162 | 1.89 (1.21-2.95) | 0.85 (0.76-0.95) | 11 | 55 | 25 | 250 | 1.69 (0.98-2.93) | 0.84 (0.67-1.06) | 8 | 58 | 8 | 267 | 2.8 (1.63-4.82) | 0.60 (0.37-0.99) | |
| Puffiness | 10 | 7 | 143 | 181 | 1.76 (0.68-4.5) | 0.97 (0.92-1.02) | 3 | 14 | 33 | 291 | 1.82 (0.54-6.02) | 0.96 (0.86-1.06) | 2 | 15 | 14 | 310 | 2.71 (0.67-10.8) | 0.91 (0.76-1.11) | |
| Hyperpigmentation | 10 | 13 | 143 | 175 | 0.94 (0.42-2.1) | 1 (0.94-1.06) | 6 | 17 | 30 | 288 | 2.99 (1.26-7.1) | 0.88 (0.76-1.02) | 5 | 18 | 11 | 307 | 5.64 (2.4-13.3) | 0.72 (0.52-1.01) | |
| Peripheral neuropathy | 17 | 12 | 136 | 176 | 1.74 (0.85-3.53) | 0.94 (0.88-1.02) | 8 | 21 | 28 | 284 | 3.23 (1.54-6.75) | 0.83 (0.7-0.99) | 4 | 25 | 12 | 300 | 3.25 (1.28-8.22) | 0.81 (0.61-1.08) | |
| Pericardial rub | 5 | 2 | 148 | 186 | 3.07 (0.06-15.6) | 0.97 (0.94-1.07) | 0 | 7 | 36 | 294 | 0 | 1.02 (1.01-1.04) | 0 | 7 | 16 | 318 | 0 | 1.02 (1.01-1.04) | |
| Anemia (Hb<7g/dL) | 39 | 114 | 35 | 153 | 1.23 (0.95-1.0) | 0.82 (0.63-1.07) | 15 | 21 | 59 | 246 | 2.58 (1.4-4.71) | 0.86 (0.76-6.97) | 9 | 7 | 65 | 260 | 4.64 (1.79-12) | 0.90 (0.82-0.98) | |
Hb: Hemoglobin, eGFR: Estimated glomerular filtration rate, TP: True positive , FP: False positive, FN: False negative, TN: True negative, LR: Likelihood ratio
Table 2: Diagnostic accuracy of clinical symptoms and signs for detection of reduced eGFR
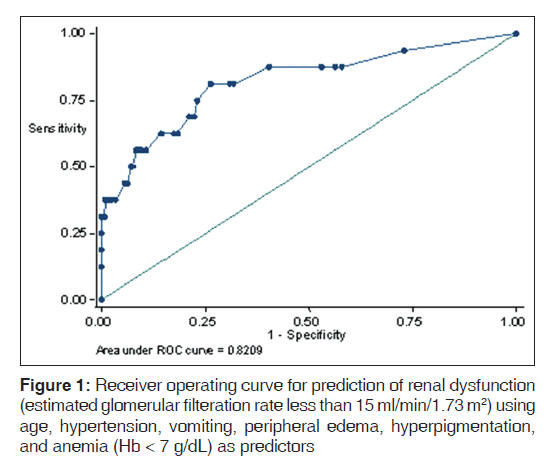
We used the above variables along with age, in predictive models, to identify the best combination of clinical symptoms and signs which may be used to screen for reduced renal function. Using logistic regression techniques, the best predictive model for eGFR less than 15 ml/min/1.73 m2, included Age > 45 years, presence of hypertension, vomiting, peripheral edema, hyperpigmentation, and anemia (Hb < 7 g/dL). The AUC for this model was 0.82 [Figure 1].
The same model was predictive of eGFR less than 30 ml/min/1.73 m2, with an AUC of 0.69. The different models and their AUCs are summarized in Table 3. Overall the predictability for eGFR less than 15 ml/min was better as compared to 30 ml/min.
| Model | Age morethan 45 | Hypertension | Anemia | Peripheralneuropathy | Vomiting | Hyperpigmentation | Pruritis | Edema | AUC |
|---|---|---|---|---|---|---|---|---|---|
| Models for prediction of eGFR less than 15 ml/min/1.73 m2 | |||||||||
| 1 | × | × | × | × | × | 0.7915 | |||
| 2 | × | × | × | × | × | 0.7934 | |||
| 3 | × | × | × | × | × | × | 0.8088 | ||
| 4 | × | × | × | × | × | × | 0.8209 | ||
| 5 | × | × | × | × | 0.7965 | ||||
| 6 | × | × | × | 0.7738 | |||||
| 7 | × | × | × | × | 0.7929 | ||||
| Models for prediction of eGFR less than 30 ml/min/1.73 m2 | |||||||||
| 1 | × | × | × | × | × | 0.6803 | |||
| 2 | × | × | × | × | × | 0.6634 | |||
| 3 | × | × | × | × | × | × | 0.7003 | ||
| 4 | × | × | × | × | × | × | 0.6909 | ||
| 5 | × | × | × | × | 0.6785 | ||||
| 6 | × | × | × | 0.6643 | |||||
| 7 | × | × | × | × | 0.6744 | ||||
The ‘×’ symbol indicates that the variable was used in the predictive model, veGFR: Estimated glomerular filtration rate, AUC: Area under the curve
Table 3: Different predictive models for detection of reduced renal function
Discussion
In the current study, we demonstrated that clinical symptoms and signs suggestive of reduced eGFR donot predict eGFR less than 60 ml/min; however predictability improves for very low eGFR (less than 15 ml/min/1.73 m2). Individuals above 45 years of age, who have hypertension, vomiting, peripheral edema, and hemoglobin less than 7 g/dL are likely to have eGFR less than 15 ml/min/1.73 m2.
Lack of predictability for eGFR less than 60 ml/min/1.73 m2 emphasizes that symptom based screening for renal dysfunction is a poor technique, and will have low sensitivity. Thus, opportunistic and disease based on screening remain important for screening for renal dysfunction. Serum creatinine measurement hence remains only screening tool, as detection of renal dysfunction before development of overt clinical symptoms and signs is the key to prevent further deterioration in the renal functions.
Given the huge burden of chronic diseases, such as diabetes mellitus and hypertension, and increasing burden of CKD, simpler tools to assess serum creatinine levels are required. As of now, there is no point of care test to estimate serum creatinine levels. However developments in the field of lateral flow technologies, microfluidics, and dry chemistry could make such a measurement possible. Examples of such a development in technology are availability of point of care natriuretic peptide and glycosylated hemoglobin levels. Similar test, if made available for serum creatinine levels, can supplement current proteinuria based screening for renal dysfunction.
Our study has several strengths. This is one of the first attempts to quantify various signs and symptoms for diagnosis of reduced renal function. While various risk scores have previously been reported in relation to CKD,[4-7] but these all predict future cardiovascular events in those with manifest CKD. In one study by Taal and Brenner risk scores have been emphasized as practical tools to help stratify patients at increased risk of CKD.[8] We evaluated different symptoms and signs in a blind and independent manner and hence as to reduce interviewer/ observer bias in their interpretation. We excluded patients at a high-risk for acute renal failure (such as critically ill), and those who were previously known to have CKD and were on dialysis therapy to reduce reporting bias in these symptoms.
However, our study also has some important limitations. First, we did not evaluate the type of renal dysfunction by either etiology, or chronicity. eGFR measurements exhibit a high degree of intra individual variability and ideally require second measurements to accurately represent kidney function.,[9,10] Second, study was among hospitalized adults and hence individuals with or without renal dysfunction would have a high prevalence of symptoms and signs. Each of the 18 clinical features, we evaluated is non-specific and have multiple possible etiologies. However, both these limitations mimic real clinical scenarios, where myarid presentations often masquerade renal dysfunction. Third, although the symptoms and signs were best predictive of eGFR less than 15 ml/min/1.73 m2, less than 5% patients belonged to this group. This is likely to result in imprecise estimates and wide confidence intervals. Fourth, we did not measure certain other parameters such as a socio-economic status, height, weight, or body mass index, which could have been predictive. Last, we estimated renal dysfunction based on MDRD formula, which has its own limitations that it is most accurate for eGFR less than 30 ml/min/1.73 m2.[11] The impact of calibration on the accuracy of the GFR estimating equation is now well-recognized. Errors in GFR estimates using uncaliberated serum creatinine concentration are larger at higher levels of GFR and although are smaller near 60 ml/min/1.73 m2, they can still cause substantial differences in estimated prevalence of CKD when applied to large populations. These errors were the lowest in individuals with a GFR less than 60 ml/min/1.73 m2. For example, a GFR estimates of 30 ml/min/1.73 m2 would have an expected range of 25-31 ml/min/1.73 m2 (−17- +3%), which is not likely to be clinically significant. However, an eGFR of 90 ml/min/1.73 m2 was associated with an error range of 62-100 ml/min/1.73 m2 (−31-11%), which is likely to be significant.
Conclusion
This study evaluates various symptom-sign-combinations, which may be helpful to screen for renal dysfunction in hospitalized adults. Clinical symptoms and signs are poorly predictive of reduced renal function, except for very low eGFR of less than 15 ml/min/1.73 m2. However, early diagnosis of renal dysfunction cannot reliably be made using the classical clinical features and hence invasive techniques remain the mainstay for screening.
Source of Support: Nil.
Conflict of Interest: None declared.
References
- Kher V. End-stage renal disease in developing countries. Kidney Int 2002;62:350-62.
- Coresh J, Astor BC, Greene T, Eknoyan G, Levey AS. Prevalence of chronic kidney disease and decreased kidney function in the adult US population: Third National Health and Nutrition Examination Survey. Am J Kidney Dis 2003;41:1-12.
- Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D. A more accurate method to estimate glomerular filtration rate from serum creatinine: A new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 1999;130:461-70.
- Le A, Wilson R, Douek K, Pulliam L, Tolzman D, Norman D, et al. Prospective risk stratification in renal transplant candidates for cardiac death. Am J Kidney Dis 1994;24:65-71.
- Lewis MS, Wilson RA, Walker KW, Wilson DJ, Norman DJ, Barry JM, et al. Validation of an algorithm for predicting cardiac events in renal transplant candidates. Am J Cardiol 2002;89:847-50.
- Foley RN, Parfrey PS, Hefferton D, Singh I, Simms A, Barrett BJ. Advance prediction of early death in patients starting maintenance dialysis. Am J Kidney Dis 1994;23:836-45.
- Armstrong KA, Rakhit DJ, Case C, Johnson DW, Isbel NM, Marwick TH. Derivation and validation of a disease-specific risk score for cardiac risk stratification in chronic kidney disease. Nephrol Dial Transplant 2005;20:2097-104.
- Taal MW, Brenner BM. Predicting initiation and progression of chronic kidney disease: Developing renal risk scores. Kidney Int 2006;70:1694-705.
- Rule AD, Larson TS, Bergstralh EJ, Slezak JM, Jacobsen SJ, Cosio FG. Using serum creatinine to estimate glomerular filtration rate: Accuracy in good health and in chronic kidney disease. Ann Intern Med 2004;141:929-37.
- Perrone RD, Madias NE, Levey AS. Serum creatinine as an index of renal function: New insights into old concepts. Clin Chem 1992;38:1933-53.
- Stevens LA, Stoycheff N. Standardization of serum creatinine and estimated GFR in the Kidney Early Evaluation Program (KEEP). Am J Kidney Dis 2008;51:S77-82.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.