Effects of Coenzyme Q10 Supplementation on Anti-Müllerian Hormone Levels in Patients with Polycystic Ovarian Syndrome
2 Department of Anesthesiology and Critical Care, Arak University of Medical Sciences, Arak, Iran, Email: dralirezakamalianesthesiology@gmail.com
Citation: Shokrpour M, et al. Effects of Coenzyme Q10 Supplementation on Anti-Mullerian Hormone Levels in Patients with Polycystic Ovarian Syndrome. Ann Med Health Sci Res. 2019;9:440-442
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
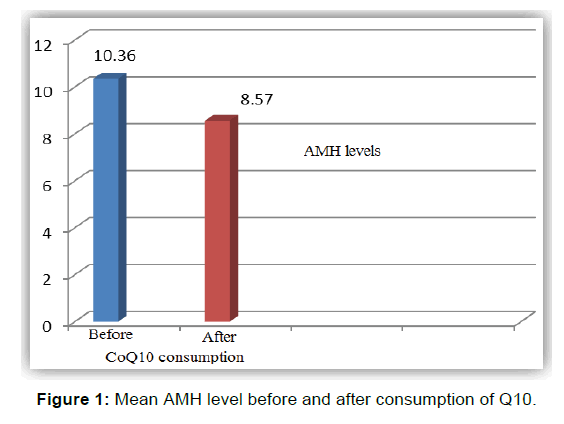
Background: Polycystic ovarian syndrome (PCOS) is a complicated endocrine disorder characterized by metabolic and reproductive disorders. This syndrome is the most common cause of infertility. The aim of this study was to investigate the effect of Coenzyme Q10 on anti-Müllerian hormone (AMH) level as an indicator of PCOS patients. Methods and Methods: We designed an interventional clinical trial study on 30 women with PCOS. The variables studied were age, marriage status, number of pregnancies and history of PCOS. All patients were evaluated for AMH by Electrochemiluminescence Immune assay. Patients with a serum AMH level of 5 and above were treated with CoQ- 10 pills 100 mg daily for 3 months. Results: The mean age of patients was 23.3 ± 4.7 and the duration of PCOS was 4.76 ± 0.76 years. 55% of patients were single and 78% of cases referred with Oligomenorrhea. The mean level of AMH in patients with PCOS showed that Q10 significantly decreased the AMH (2.2 ± 10.36 vs. 1.4 ± 8.57, p value = 0.02). Conclusion: The effect of Q10 intake as an inexpensive and effective therapy significantly reduced the level of AMH in patients with PCOS.
Keywords
Anti-Müllerian hormone; Coenzyme Q10; Polycystic ovary syndrome; Infertility
Introduction
Polycystic ovarian syndrome (PCOS) is a complex condition characterized by high levels of androgen, menstrual disorder, or small cysts in one or both ovaries. [1] This complication is a heterogeneous disorder that affects at least 7% of adult women. [2] According to the World Health Organization, nearly 5 million pregnant women in the United States are exposed to PCOS, with a detection and the management costs of over 4 billion $ a year to identify and manage. [3] In addition, researches have shown that PCOS increases the metabolic risk factor, type 2 diabetes and possibly cardiovascular disease. About a third of patients with PCOS have impaired glucose tolerance. Therefore, the central role of insulin resistance in the emergence of PCOS manifests itself as a primary objective for management of this disease. [4] Various studies have shown that resistance to insulin not only causes metabolic problems but also plays a role in the development of fertility disorders in PCOS women. Studies have shown that insulin has a profound effect on both the levels of ovarian and folic acid stroma. Insulin secretes androgens in the ovary, and the increase in androgens results in the analysis of growing follicles and interfere with the appearance of a dominant follicle. Increasing the number of follicles and increasing the androgens in PCOS patients leads to an increase in the production of anti-Müllerian hormone (AMH). [5,6]
AMH is produced from Granulosa cells of the antral ovarian follicles and its amount represents the size of growing follicles and ovarian reserve function. Previous studies have shown that the level of AMH in PCOS is increased by 2-3 times and is associated with various metabolic, endocrine and reproductive disorders (such as ovarian dysfunction, hyperandrogenism, and infertility). [7] Evidence suggests that elevated levels of AMH in women with PCOS are likely to play a role in the follicular infertility and decrease the sensitivity of growing follicles to follicular stimulatory hormone. Recently, the determination of serum AMH level as a valid indicator of ovarian function has been suggested for female infertility and PCOS diagnosis, and possibly the possibility of targeted treatment of infertility. [8-10]
Now-a-days in various studies the effect of non-pharmacological therapies through the regimen on reproductive performance and the reduction of metabolic heart risk factors has been shown. [11] Previous studies have shown that taking coenzyme Q1O (CoQ10) supplements with antioxidant effects greatly improves blood glucose control (glucose concentration, insulin concentration, insulin resistance and HbA1c), blood pressure, dyslipidemia (TG, chol), ovulation induction and preserving the ovarian reserve. [12,13] Animal studies have reported that CoQ10 increases the reproductive life of female rats up to 30%, and animals that produce more CoQ10 are more likely to ovulate and improve ovarian responses and hormonal changes in them. [14] Therefore, due to the high prevalence of PCOS and its complications, as well as the significant effect of CoQ10 on various disorders in these patients and the lack of adequate and human studies in this field, we have investigated the effect of Q10 on AMH levels in patients with PCOS.
Materials and Methods
Study selection
This study was an interventional randomized clinical trial in which the effect of coenzyme Q10 on PCOS was measured over a period of 3 months. Patients who participated in this study were selected from all women with PCOS who were referred to the Kowsar Medical Center (Arak University of Medical Sciences, Iran). All the patients have proved to have PCOS according to Rotterdam Criteria. There were 30 women eligible to participate in the study.
Inclusion criteria: Have at least 2 of 3 Rotterdam Criteria;
• Oligo-ovarian manifestations and ovulation failure
• Clinical or laboratory symptoms of hyperandrogenism and
• Polycystic ovary specimen in ultrasound as a criterion for PCOS)
Exclusion criteria: CoQ10 supplementation in the last 3 months, Sensitivity to Q10 and lack of consent to participate in the study.
After selecting patients, the variables including age, marriage status, history of pregnancies and the history of PCOS were extracted from their medical records. All patients were evaluated for AMH by Electrochemiluminescence Immune assay (ECLIA) using Cobas e411 instrument.
AMH levels based on current kits include natural ovarian reserve, AMH >1 and high reserve (often in PCOS patients) with an AMH> 3 and a normal lower limit of 0.10-9.0. Given the constant amount of AMH over the entire menstrual cycle, this test can be done at any time from the menstrual cycle.
All the patients with a serum AMH levels of 5 and above were treated with CoQ10 pills (made in Zist Takhmir Co. Iran) for 3 months at a daily rate of 100 mg (one tablet). At the end of the third month, the patients returned to the laboratory and their AMH levels were measured again.
Ethical considerationsThe aim of the study was described for all patients, and then they were asked to attend the study by filling out the Consent Form. Patients were also free to withdraw from the study if they had adverse effects due to drug use or if they did not wish to continue their collaboration.
Statistics analysis
Data were analyzed using SPSS software (version 20) and also using statistical tests such as t- test and ANOVA, and the level of significance was considered to be 0.05.
Results
In this study, 30 patients were selected among PCOS patients referred to Kawsar Medical Center (Arak, Iran). The mean age of patients was 23.3 ± 4.7 and the mean duration of PCOS was 4.8 years. The marriage status and the reason for referral were also studied in patients, which are listed in Table 1. 55% of patients were single and 45% of them were married. 78% of cases referred due to Oligomenorrhea. The effect of Q10 on the level of AMH was evaluated in PCOS patients. Results showed that CoQ10 in patients with PCOS significantly decreases AMH and the mean AMH level after CoQ10 administration in patients is obviously reduced [Figure 1].
| Table 1: Demographic characteristics and measurement of AMH levels in patients. | |||
| Parameters Age (years old) | Mean ±SD | p-value | |
|---|---|---|---|
| 23.3 ±4.7 | - | ||
| Marriage status | Single | 55% | - - |
| Married | 45% | ||
| PCOS duration (Years) | 4.8 ±0.76 | - | |
| Serum AMH level | Before CoQ10 consumption | 10.36 ±2.2 | 0.02 |
| After CoQ10 consumption | 8.57 ±1.4 | ||
| Referral reason | Oligomenorrhea | 78% | - |
| Fertility problems | 22% | ||
Discussion
Managing and treating women with the polycystic ovarian syndrome (PCOS) is one of the major challenges in reproductive medicine. This syndrome is associated with a wide range of causes and clinical symptoms, including increased insulin levels, insulin resistance, and increased androgens. [15] The initial causes of this complication remain questionable. However, due to the proven role of oxidative stress in the pathogenesis of PCOS, many studies have been performed on the effects of antioxidants on biochemical parameters and on the course of the disease and clinical manifestations of these patients in order to improve and treat these patients along with other treatments using these compounds. [16] In this study, we also tried to investigate the role of antioxidant coenzyme Q10 in evaluating the effect of this coenzyme on the level of AMH as an indicator hormone in diagnosing patients with PCOS syndrome. The results of this study showed that CoQ10 consumption along with the treatment regimen of patients with PCOS significantly decreased AMH level and improved fertility.
Studies have shown that CoQ10 is the most potent endogenous antioxidant in that inhibits protein, DNA and fat oxidation. The amount of CoQ10 is reduced in the pathologic conditions associated with oxidative stress and the aging process. [17] The addition of CoQ10 supplement to the diet of patients with some disorders such as cardiovascular disease, metabolic disorders, and immune deficiency can be useful. [18-20]
The results of previous studies were almost consistent with our study, in a study by Molyneux et al. numerous studies were reviewed, and collectively they reported that CoQ10 deficiency leads to manifestations such as progressive muscle weakness, abnormal fatigue, and dysfunction of the nervous system. [21] Animal studies have also reported that COQ10 increases the reproductive life of mice to 30%, and animals that produce more CoQ10 can produce more eggs. [14] Several other trials have been conducted on the use of CoQ10 in various diseases and pathological conditions. One of the most important studies was performed on the effect of Q10 on the reduction of myopathic pain associated with statin use. A prospective observational study titled relationship between serum AMH level and the number of antral follicles in PCOS women performed by Park A et al. In that study, all patients with PCOS, had serum AMH levels ranged between 1.65 to 14.2 ng/ml (an average of 5.61 ± 0.9) and the number of antral follicles performed in the MRI was between 14-37 follicles (An average of 23.9 + - 1.7). Results of this study showed a significant relationship between serum levels of AMH and the number of antral follicles in patients with PCOS. [7]
In another study, Refaeey et al. surveyed the effects of clomiphene citrate and CoQ10 on ovulation induction in patients with clomipheneresistant PCOS. The results of their study were presented as primary and secondary outcomes. Primary outcomes include the number of follicles, serum estradiol and progesterone levels, endometrial thickness, and ovulation and secondary outcomes include pregnancy and abortion. The overall results indicate a significant effect of CoQ10 and clomiphene citrate on improving the rate of ovulation and pregnancy in patients with PCOS and suggested that these components could be considered as an effective option before treatment with gonadotrophin and Ovarian Drilling. [12]
Another study titled Protective Effects of CoQ10 on ovarian resistance against oxidative stress damage in 2016 performed by Özcan P and colleagues on female rats. Their results indicated that CoQ10 supplementation could have a protective role of ovarian reserve against the mitochondrial aging process and the planned physiological ovarian aging process. According to studies, oocytes have the highest number of mitochondria in each cell (about 2 × 105 copies). The mitochondrial function affects the quality of the eggs and plays an important role in the fertility and fetal development process. It has been shown that impairment of mitochondrial function may be the onset of a metabolic syndrome such as obesity, insulin resistance, abnormal lipid profile, and increased risk of postpartum cardiovascular disease. [17-21]
Conflict of Interest
The authors disclose that they have no conflicts of interest.
REFERENCES
- Umland EM, Weinstein LC, Buchanan EM. Menstruation-related disorders. DiPiro JT, Talbert RL, Yee GC, et al. (Eds) Pharmacotherapy: A Pathophysiologic Approach, (8th ed) New York: McGraw-Hill. 2011;1393.
- Aubuchon M, Legro RS. Polycystic ovary syndrome: Current infertility management. Clinical Obstetrics and Gynecology. 2011;54:675-684.
- Obstetricians ACo Ltd, Gynecologists. ACOG Practice Bulletin No. 41: Polycystic ovary syndrome. Obstet Gynecol. 2002;100:1389-1402.
- Moran L, Norman R. Understanding and managing disturbances in insulin metabolism and body weight in women with polycystic ovary syndrome. Best Practice & Research Clinical Obstetrics & Gynaecology. 2004;18:719-736.
- Turan V, Sezer ED, Zeybek B, Sendag F. Infertility and the presence of insulin resistance are associated with increased oxidative stress in young, non-obese Turkish women with polycystic ovary syndrome. Journal of pediatric and adolescent gynecology. 2015;28:119-123.
- Suresh S, Vijayakumar T. Correlations of insulin resistance and serum testosterone levels with LH: FSH ratio and oxidative stress in women with functional ovarian hyperandrogenism. Indian Journal of Clinical Biochemistry. 2015;30:345-350.
- Park A, Brown M, Rosencrantz M, Shayya R, Chuan S, Chang R. Serum AMH levels correlate with antral follicle count in PCOS adolescents. Fertility and Sterility. 2009;92:S99.
- Pellatt L, Rice S, Dilaver N, Heshri A, Galea R, Brincat M, et al. Anti-Müllerian hormone reduces follicle sensitivity to follicle-stimulating hormone in human granulosa cells. Fertility and sterility. 2011;96:1246-1251. e1.
- Nestler JE. Metformin in the treatment of infertility in polycystic ovarian syndrome: an alternative perspective. Elsevier; 2008.
- Neagu M, Cristescu C. Anti-Mullerian hormone–a prognostic marker for metformin therapy efficiency in the treatment of women with infertility and polycystic ovary syndrome. Journal of medicine and life. 2012;5:462.
- Ravn P, Haugen A, Glintborg D. Overweight in polycystic ovary syndrome. An update on evidence based advice on diet, exercise and metformin use for weight loss. Minerva Endocrinol. 2013;38:59-76.
- El Refaeey A, Selem A, Badawy A. Combined coenzyme Q10 and clomiphene citrate for ovulation induction in clomiphene-citrate-resistant polycystic ovary syndrome. Reproductive biomedicine online. 2014;29:119-124.
- Farhangi MA, Alipour B, Jafarvand E, Khoshbaten M. Oral coenzyme Q10 supplementation in patients with nonalcoholic fatty liver disease: effects on serum vaspin, chemerin, pentraxin 3, insulin resistance and oxidative stress. Archives of medical research. 2014;45:589-595.
- Janzadeh A, Nasirinezhad F, Jameie S. Effect of Coenzyme Q10 on neuropathic pain in adult CCI Rat model. Journal of Gorgan University of Medical Sciences. 2012;14:10-18.
- Speroff L, Fritz MA. Clinical gynecologic endocrinology and infertility: Lippincott Williams & wilkins; 2005.
- Zuo T, Zhu M, Xu W. Roles of oxidative stress in polycystic ovary syndrome and cancers. Oxidative Medicine and Cellular Longevity. 2016;2016.
- Özcan P, Fıçıcıoğlu C, Kizilkale O, Yesiladali M, Tok OE, Ozkan F, et al. Can Coenzyme Q10 supplementation protect the ovarian reserve against oxidative damage? Journal of Assisted Reproduction and Genetics. 2016;33:1223-1230.
- Sharma A, Fonarow GC, Butler J, Ezekowitz JA, Felker GM. Coenzyme Q10 and heart failure: A state-of-the-art review. Circulation: Heart Failure. 2016;9:e002639.
- Miles MV, Morrison JA, Horn PS, Tang PH, Pesce AJ. Coenzyme Q10 changes are associated with metabolic syndrome. Clinica Chimica Acta. 2004;344:173-179.
- Farough S, Karaa A, Walker M, Slate N, Dasu T, Verbsky J, et al. Coenzyme Q10 and immunity: a case report and new implications for treatment of recurrent infections in metabolic diseases. Clinical Immunology. 2014;155:209-212.
- Molyneux SL, Young JM, Florkowski CM, Lever M, George PM. Coenzyme Q10: is there a clinical role and a case for measurement? The Clinical Biochemist Reviews. 2008;29:71.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.