Evaluation of Antiulcer Properties of Ethanolic and Hot Aqueous Stem Extracts of Synclisia scabrida on Experimentally Induced Ulcer Models in Albino Mice
- *Corresponding Author:
- Dr. Ughachukwu PO
Department of Pharmacology and Therapeutics, College of Medicine, Anambra State University, Awka Campus, Anambra State, Nigeria.
E-mail: ughapieces@yahoo.com
Citation: Onwudiwe TC, Ughachukwu PO, Unekwe PC, Ogamba JO. Evaluation of antiulcer properties of ethanolic and hot aqueous stem extracts of Synclisia scabrida on experimentally induced ulcer models in albino mice. Ann Med Health Sci Res 2012;2:134-9.
Abstract
Background: The treatment of peptic ulcer disease poses therapeutic challenges to both patients and physicians alike because of the tendency of ulcers to relapse. Drugs used in the treatment of this disease are either costly or are associated with high incidence of adverse effects.Synclisia scabrida is a plant used in ethnomedicine for the treatment of various forms of stomach disorders and menstrual pains. The medicinal properties of the plants are claimed to reside in the roots, stems, and the leaves. Aim: This study, therefore, is to verify this claim and elucidate the probable mechanism of action by using crude stem extracts of this plant on drug‑ and stress‑induced ulcer models in albino mice. Materials and Methods: Crude ethanol and hot water extracts, EE and HWE respectively, of the stem were prepared. These extracts were fractionated and separated by chromatographic methods and the fractions pooled together as fractions (PF‑1, PF‑2, PF‑3 respectively) based on their chromatographic mobility and color reactions. Phytochemical analysis was done on the extracts. Ulcer models were induced in albino mice by means of indomethacin, histamine, and stress after prior cytoprotection with orally administered crude extracts and control (cimetidine). Results: Phytochemical analysis of the crude extracts and their fractions revealed the presence of cardiac glycosides (+++), tannins (+++), saponins (+), flavonoids (++), carbohydrates (++) and alkaloids (+++). Acute toxicity study on the crude extracts and their fractions revealed relative safety at the dose of 5000 mg/ kg. The crude extracts (EE and HWE) and their fractions (PF‑1, PF‑2, PF‑3) significantly (P = 0.001) protected against indomethacin‑, histamine‑ and stress‑induced ulcers. The decrease in GIT motility produced by these extracts was comparable to that produced by atropine sulfate. Conclusion: The findings suggest that these extracts of Synclisia scabrida possess antiulcer and antispasmodic properties, which justify the claims for its use in the treatment of various forms of stomach disorders.
Keywords
Albino mice, Drug induced ulcers, Synclisia scabrida extracts
Introduction
Peptic ulcer is defined as mucosal erosion of the gastrointestinal tract (GIT) which is ≥0.5 cm. It is among the commonest causes of ill-health, especially with increased sophistication and adoption of western lifestyle. This western lifestyle with attendant stress, consumption of low-fiber junk food, and smoking are important contributory factors.[1] Helicobacter pylori infection of upper GIT is implicated in the pathogenesis of peptic ulcer and many other GIT disorders like gastritis and gastric cancers.[2,3] It has been demonstrated that eradication of H. pylori infection was followed by a reduction in the incidence of peptic ulcer disease.[4] Thus, the pathogenesis of peptic ulcer largely revolves around low-fiber diet, Helicobacter pylori infection of upper GIT, nonsteroidal anti-inflammatory drugs use, stress, and probably cigarette smoking.
Gastric erosion and subsequent ulceration is associated with oral consumption of NSAIDs. Use of these anti-inflammatory agents is very common among the elderly who usually suffer from arthritis and other inflammatory joint diseases that require treatment with NSAIDs. For these elderly NSAID users, fatal complications occur in as high as 1 per 1000 users.[5] NSAIDs generate oxygen free radicals that play important roles in the pathogenesis of gastrointestinal mucosal injury.[6] They also inhibit cyclo-oxygenases, which leads to reduced synthesis of prostaglandins; chemical mediators that increase production of cytoprotective mucus in epithelial surfaces.
The role of stress in the pathogenesis of peptic ulcer is not clearly understood. It is probably related to decreased enteric blood flow as a result of stress-induced sympathetic discharge. This results in shock-like circulating disturbance of enteric and gastric blood supply, culminating in necrosis of apical mucosal cells.[7]
Poor efficacy occasioned by drug resistance and adulteration coupled with high cost and relative inaccessibility of orthodox drugs have heightened interest in herbal medication. Diseases like HIV/AIDS, malaria, tuberculosis, cancer, diabetes mellitus, and hypertension have continued to defy orthodox medical solutions. In particular, the treatment of peptic ulcer disease poses therapeutic challenges to both patients and physicians alike because of the tendency of ulcers to relapse. The more efficacious antiulcer drugs such as omeprazole and misoprostol are very expensive and not readily available in rural areas. Others, such as cimetidine, have unacceptably high incidence of adverse effects, especially in male patients.
There is therefore the need for continued search for antiucler drugs with more favorable pharmacokinetic and economic profiles. Medicinal plants with known ulcer-healing properties include Piper betel and Terminalia chebula.[5] Traditional healers in Southeast Nigeria use the root, stem, and leaf decoctions of Synclisias cabrida in the treatment of upper abdominal pains, dyspepsia, menstrual pains, and sexually transmitted diseases. The objectives of this study, therefore, include:
1. To determine the antiulcer properties of the stem extract of this plant so as to justify its use in tradomedicine for treatment of stomach ulcers.
2. To determine the possible mechanisms of action this plant’s extracts as an antiulcer agent.
Materials and Methods
Drug source
1. Cimetidine tablets (Sigma Ltd, London)
2. Indomethacin tablets (British Drug House)
3. Histamine (Sigma Ltd, London)
4. Atropine sulfate (British Drug House)
Animals
Adult male and female albino mice weighing between 16 and 18 g. Animals were maintained and utilized in accordance with the standard guide for the care and use of laboratory animals. Approval for this study was obtained from the Ethics Committee, Abia State University.
Collection and identification of the plant
The plant, Synclisia scabrida (uziza in Igbo), was collected from a farm land in Owerri, Imo State of Nigeria, and was identified by Mr. JMC Ekekwe of Department of Botany, University of Nigeria, Nsukka, and a voucher specimen was deposited there.
Preparation of plant extracts
Fresh stems of the plant were obtained, washed, air-dried, cut into small sizes, and pounded in a mortar into a coarse powder. For the ethanol extraction, 50 g of the powdered material was soaked in 200 mL of 80% ethanol and left to stand at room temperature for 36 h, with occasional shaking.[8] For hot water extraction, 50 g of the powdered material was soaked in 200 mL of hot water (100°C) and left to stand for 4 h with occasional shaking.[8] After standing for appropriate hours, each portion was filtered through Whatman No. 1 filter papers and the filtrates were collected.The filtrates were evaporated to dryness in a steady air current and the residues collected after which the percentage yield of the extract was calculated.
Determination of pH of crude extracts
1.0 g of each extract was mixed with 10 mL de-ionized water and the pH of the resulting solution was measured using a pH meter.
Fractionation of the crude extracts
This was done using column chromatography with chloroform/ ethylacetate/hexane solvent system Sufficient quantity of silica gel (particle size 0.063-0.1 mm) was wet packed using the chloroform/ethylacetate/hexane (7:2:1, v/v/v) solvent system. This was followed by the addition of 5.0 g of each extract in chloroform. The extract was placed on the column and then continuously eluted with the mobile phase solvent (chloroform/ ethylacetate/hexane, 7:2:1, v/v/v) and fractions/aliquots were collected in the test tubes. The fractions were compared by TLC mobility and color reaction and similar fractions in separate evaporating dishes and evaporated under a steady air-current in a tune chamber. The percentage yield and pH of the pooled fraction were also determined. Phytochemical analysis was carried out on the crude extract and pooled fractions.
Labeling and storage extracts
The crude extract and fractions were put in clean sterile containers, labeled, and stored in a refrigerator until use.
Acute toxicity test
Using the modified method of Lorke, the LD50 of the crude extract was determined.[9]
Evaluation of antiulcer properties of crude and pooled fraction extract on different ulcer models
The antiulcer activity of the extracts was evaluated on three ulcer models in albino mice. These models included:
1. Indomethacin-induced ulcer model
2. Histamine-induced ulcer model
3. Stress-induced ulcer model
The albino mice in each case were fasted for 24 h and allowed free access to water until 2 h prior to the experiment.
Preliminary tests
These tests were done to determine the least effective doses of indomethacin and histamine that would produce 100% gastric ulcers in test animals in 4 h and to determine the minimum dose of the crude hot water extract that has highest antiulcer activity. For indomethacin, this was done by administering various oral doses of indomethacin (40, 60, 80, and 100 mg/ kg body weight) respectively to four groups of animals, consisting of three animals in each group. For histamine, this was done by administering various doses of histamine (0.2, 0.3, 0.4, and 0.5 mg/kg body weight) respectively to four groups of animals, consisting of three animals in each group. 100 mg/ kg indomethacin and 0.5 mg/kg histamine were respectively found to produce 100% gastric ulceration in all the animals in 4 h.[10] The animals were sacrificed with chloroform, their stomach dissected out and opened along the greater curvature, rinsed under a stream of clean water, and pinned flat on a corkboard. The stomachs were examined with hand lens (×10 magnification) for gastric ulcers.
To determine the minimum dose of the crude hot water extract (HWE) that would possess greater antiulcer activity, 50, 100, 200, and 400 mg/kg body weight of the crude HWE was administered to another four groups of test animals consisting of three animals per group before oral administration of indomethacin and histamine as above, and it was found that 400 mg/kg body weight HWE produced the greatest antiulcer activity. This dose was also adopted for other extracts (ethanol, PF-1, PF-2, and PF-3).
Using optimal doses of drugs and extracts from the preliminary tests, the following tests were done using another set of albino mice.
Indomethacin-induced ulcer
28 albino mice of both sexes, weighing between 16 and 18 g, were randomized into seven groups of 4 animals per group and labeled A-G. The animals were fasted for 24 h and allowed free access to water until 2 h prior to the experiment.
The animals received the following drugs and control orally:
Group A (positive control) received 100 mg/kg cimetidine (1.6 g in 0.5 mL distilled water)
Group B received 400 mg/kg ethanol extract (EE) (6.4 g in 0.5 mL distilled water)
Group C received 400 mg/kg HWE (6.4 g in 0.5 mL distilled water)
Group D received 400 mg/kg PF-1 (6.4 g in 0.5 mL distilled water)
Group E received 400 mg/kg PF-2 (6.4 g in 0.5 mL distilled water)
Group F received 400 mg/kg PF-3 (6.4 g in 0.5 mL distilled water)
Group G (negative control) received 5 mL/kg 3% v/v Tween 80 orally.
Thirty minutes following the respective treatment, ulcer was induced with indomethacin 100 mg/kg (1.6 g in 0.5 mL water) orally as determined in the preliminary test. Six hours after this, the animals were sacrificed and their stomachs examined for ulcers as described above. The gastric ulcers were rated as shown below.[11]
| Severity rating | Ulcer diameter |
|---|---|
| 0 | No ulcer |
| 0.5 | Pinpoint ulcer |
| 1.0 | Ulcer ≤2 mm |
| 2.0 | Ulcer ≥2 mm ≤3 mm |
| 3.0 | Ulcer ≥3 mm |
Ulcer index was calculated by multiplying the number of ulcers whose diameters fall within a designated ulcer diameter range with the corresponding severity rating and dividing the mean of the product by a factor 10.[11]
Histamine-induced ulcer
The procedure was repeated as for indomethacin-induced ulcers using another set of animals; except that ulcers were induced with 0.5 mg/kg histamine (80 mcg in 0.5 mL distilled water) orally instead of indomethacin. The ulcer index was equally scored according to the guide provided above.
Stress-induced ulcer
The method adopted by Suzuki et al. 1976 and Okwuosa et al. 2006 was used.[12,13] The doses of controls and extracts were repeated as for the two models above, but stress was used to induce ulcer in another set of animals. The doses of drugs/extracts were repeated thrice a day. After the third dose, the animals were challenged to cold-induced stress by putting them in restraint cages containing water and kept in a refrigerator at 15°C for 18 h. Thereafter, the animals were sacrificed, and gastric ulcers were counted and rated as above. Percentage of ulcer protection was calculated as follows.[11]
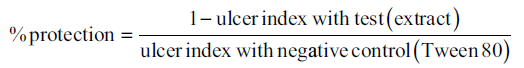
Gastrointestinal motility (charcoal meal) test
84 albino mice of both sexes, weighing 16-18 g, were randomized into seven groups of 12 animals each and the groups labeled 1-7. The animals were fasted for 24 h as well as allowed free access to water until 2 h prior to experiment. Thereafter, the following were given to the animals orally:
Group 1 (Negative control): 5 mL/kg of 3% v/v Tween 80
Group 2 (Positive control): 5 mg/kg A - atropine sulfate
Group 3: 400 mg/kg of crude EE
Group 4: 400 mg/kg of crude HWE
Group 5: 400 mg/kg of PF-1
Group 6: 400 mg/kg of PF-2
Group 7: 400 mg/kg of PF-3.
Five minutes after administration of drugs, 1.0 mL of charcoal meal (5% deactivated charcoal suspended in 10% aqueous solution of tragacanth) was administrated orally to each animal. After 30 min, 60 min, 120 min, and 240 min, three animals in each group were sacrificed using chloroform, and the stomach, small intestine, and large intestine were dissected out. The intestinal distance in centimeters moved by the charcoal meal from the pylorus was measured using a meter rule and the values obtained were expressed as percentage of distance from pylorus to ileocecal junction.[10]
Statistical analysis
The figures obtained were expressed as mean values. These values were further tested for statistical significance (students t test) using GraphPad Prism 5 (USA).
Results
Yield of the extracts
The yield of both EE and HWE are shown in Table 1.
| Extract | % yield | pH |
|---|---|---|
| Ethanol (80%) | 17.36 | 6.4 |
| Hot water (100°C) | 13.44 | 5.7 |
Table 1: The yield of both ethanol and hot water extracts with their respective pH values
Phytochemical analysis
Phytochemical analysis of the crude extracts and their fractions revealed the presence of cardiac glycosides (+++), tannins (+++), saponins (+), flavonoids (++), carbohydrates (++) and alkaloids (+++).
Acute toxicity test
No death was recorded among the experimental animals within the 48-h observation period after oral administration of up to 5000 mg/kg of the crude extracts and their fractions.
Results of antiulcer activity test
The effects of the crude extracts (EE ×HWE), pooled fractions (PF-1, PF-2, and PF-3) and standard drug (cimetidine) on indomethacin-induced ulcer models in mice were compared with those produced by control (3% v/v Tween 80).All the crude extracts and pooled fractions offered significant (P = 0.001) protection against indomethacin-induced ulcer in adult albino mice of both sexes as shown in Table 2. The same was done for histamine- and stress-induced ulcer models, and the results are presented in Tables 3 and 4, respectively. Both the crude and the fractional extracts at oral doses of 400 mg/kg reduced gastric motility significantly (P = 0.001) when compared with negative control (3% v/v Tween 80) as shown in Table 5.
| Animal group | Drug given | Dose (mg/kg/oral) | Ulcer index | % protection |
|---|---|---|---|---|
| A (positive control) | Cimetidine | 100 | 0.50 (0.14) | 70.62* |
| B | EE | 400 | 1.03 (0.60) | 41.81* |
| C | HWE | 400 | 1.17 (0.19) | 33.90* |
| D | PF-1 | 400 | 0.89 (0.11) | 49.72* |
| E | PF-2 | 400 | 1.06 (0.16) | 40.12* |
| F | PF-3 | 400 | 1.16 (0.21) | 33.84* |
| G (negative control) | Tween 80 (3% v/v) | 5 mL/kg/oral | 1.77 (0.11) | 0.00 |
Table 2: Effect of crude extracts and their fractions on indomethacin-induced ulcer models in adult albino mice
| Animal group | Drug given | Dose (mg/kg/oral) | Ulcer index | % protection |
|---|---|---|---|---|
| A (positive control) | Cimetidine | 100 | 0.20 (0.13) | 91.74* |
| B | EE | 400 | 1.12 (0.10) | 53.72* |
| C | HWE | 400 | 2.18 (0.79) | 9.92 |
| D | PF-1 | 400 | 1.09 (0.16) | 54.96* |
| E | PF-2 | 400 | 2.16 (0.44) | 10.74 |
| F | PF-3 | 400 | 2.20 (0.35) | 9.86 |
| G (negative control) | Tween 80 (3%v/v) | 5 mL/kg/oral | 2.42 (1.10) | 0.00 |
Table 3: Effect of crude extracts and their fractions on histamine-induced ulcer models in adult albino mice
| Animal group | Drug given | Dose (mg/kg/oral) | Ulcer index | % protection |
|---|---|---|---|---|
| A (positive control) | Cimetidine | 100 | 0.85 (0.58) | 79.59* |
| B | EE | 400 | 0.85 (0.75) | 73.27* |
| C | HWE | 400 | 1.86 (0.93) | 41.51* |
| D | PF-1 | 400 | 1.40 (0.23) | 55.97* |
| E | PF-2 | 400 | 2.77 (0.37) | 12.89 |
| F | PF-3 | 400 | 1.86 (0.93) | 41.51* |
| G (negative control) | Tween 80 (3% v/v) | 5 ml/kg/oral | 3.18 (1.31) | 0.00 |
Table 4: Effect of the crude extracts and their fractions on stress-induced ulcer models in adult albino mice
| Animal group | Drug given | Dose/oral | 30 min | 60 min | 120 min | 240 min | |
|---|---|---|---|---|---|---|---|
| 1 | (negative control) | Tween 80 (3% v/v) | 5 ml/kg | 5.90 (0.64) | 22.70 (1.20) | 48.40 (2.50) | 10.40 (0.57) |
| 2 | (positive control) | Atropine sulfate | 5 mcg/kg | 2.80 (0.14) | 4.90 (0.82) | 10.30 (0.94) | 21.60 (1.10) |
| 3 | EE | 400 mg/kg | 4.30 (0.52) | 8.60 (0.78) | 14.10 (1.10) | 35.5 (2.30) | |
| 4 | HWE | 400 mg/kg | 4.60 (0.22) | 7.10 (0.66) | 15.30 (0.66) | 34.70 (1.80) | |
| 5 | PF-1 | 400 mg/kg | 4.20 (0.22) | 8.50 (0.64) | 15.60 (1.30) | 30.40 (1.20) | |
| 6 | PF-2 | 400 mg/kg | 4.20 (0.82) | 8.60 (0.51) | 15,10 (1.20) | 32.10 (1.40) | |
| 7 | PF-3 | 400 mg/kg | 4.60 (0.22) | 7.00 (0.40) | 15.10 (0.72) | 34.90 (1.14) | |
Table 5: Effect of the crude extracts and their fractions on gastrointestinal motility of adult albino mice
The figures represent distances (in cm) traveled by charcoal meal. Differences between values are significant (P = 0.001) relative to negative control group. Values are ±SEM.
Discussion
In the study, the crude extracts of S. scarida and their fractions exhibited significant antiulcer properties in different ulcer models of adult albino mice.
The percentage yield of the extract using ethanol as extraction solvent was higher than that using hot water [Table 1]. The higher yield of extract obtained with EE compared to HWE could be explained by higher proportion of plant chemical constituents being soluble in ethanol.[14] This is in keeping with earlier findings that EE solvents generally yield more crude extract than aqueous extraction solvents.[15,16] However, extraction solvent alone does not determine the yield of crude extracts. Other factors like extraction time, temperature, sample to solvent ratio, as well as chemical and physical characteristics of the sample also affect the yield of crude extracts.[17]
The LD50 of the crude extracts and their fractions was greater than 5000 mg/kg body weight. This implied that the crude extracts and their fractions have low potential for toxicity because Lorke (1983) had earlier demonstrated that chemical compounds with LD50 of up to 5000 mg/kg body weight have very low toxic potentials.[9] The National Academy of Science (1975) also suggested compounds that do not show adverse effects when given in doses of 3000-5000 mg/kg body weight are essentially non-toxic.[18]
Peptic ulcers develop as a result of imbalance between aggressive action of acid-pepsin secretion and maintenance of mucosal integrity.[19] Apart from excessive and secretion, NSAIDS induce gastric ulceration by inhibition of cytoprotective endogenous prostaglandins synthesis as well as increased production of leukotrienes, which may induce mucosal vasoconstrictions, there by reducing local blood flow and enhancing mucosal injury.[20,21]
Therefore, the ability of the crude and pooled fractions of the extract to significantly (P = 0.001) protect against indomethacin-induced ulcers [Table 2] may indicate prostaglandins-mediated cytoprotective mechanism of action. This finding is similar to that obtained with unripe plantain and banana (Musa species) on aspirin-induced ulcerogenesis in rats.[22]
Histamine, a potent stimulator of parietal cells, has an essential role in the pathogenesis of peptic ulcer disease. Apart from stimulation of gastric acid secretion, it also produces mucosal microcirculatory changes, which contributes to gastric ulceration.[23,24] Therefore, the ability of the extracts to protect against histamine-induced ulcers [Table 3] suggests histamine receptor blockade as a possible mechanism of action mechanism.
Stress induces activation of the sympathetic system with release of vasoactive amines that generate ischemia by reducing blood flow to the gastric mucosa. This causes mucosal damage as well as generates oxidative stress, which decreases prostaglandin synthesis that will farther cause mucosal damage.[25]
Gastric ulcer, like other irritants, increases gastric emptying and most antiulcer drugs decrease gastric emptying. [26] Studies have shown that antispasmodic activity and decreased gastric motility leading to mucosal flattening is associated with gastric cytoprotection.[19,27] Decreased gastric motility increases absorption of orally administered antiulcer agents promoting healing. The ability of the crude extracts and pooled fractions to significantly reduce GIT motility [Table 5] may be due to anticholinergic activity. This inherent antidiarrheal activity of the extracts is a plus in the treatment of ulcer, bearing in mind that some antiulcer agents such as pantoprazole and tripotassium misoprostol have diarrhea as one of their adverse effects.[28-30] Further studies are needed to isolate and study the effects of the active principles of this plant.
Conclusion
The stem extracts of Synclisias cabrida at doses used in this study exhibited statistically significant antiulcer activities in indomethacin-, histamine- and stress-induced gastric ulcers in albino mice. The use of the extracts of this plant in tradomedicine for treatment of abdominal pains is therefore justified. The mechanism of antiulcer activity appears to be cytoprotective and antispasmodic in nature. These extracts, may, therefore, be of value in development of novel agents in the treatment of peptic ulcers.
References
- Ryan-Harshman M, Aldoori W. How diet and lifestyle affect duodena ulcers. Can Fam Physician 2004;50:727-32.
- Smith KI, Oyedeji KS, Arigbabu HO, Coker AO, Bayerdoffer E, Miehlke S. Prevalence of Helicobacter pylori vacA, cagA, and iceA genotypes in Nigerian patients with duodenal ulcer disease. J Med Microbiol 2002;51:851-4.
- Makola D, Peura DA, Crowe SE. Helicobacter pylori infection and related gastrointestinal diseases. J Clin Gastroenterol 2007;41:548-58.
- Goh KL, Chan WK, Shiota S, Yamaoka Y. Epidemidogy of Helicobacter pylori infection and public health implications.Helicobacter 2011;16:1-9.
- Bhattacharya S, Chaudhuric SR, Chattopadhyay S, Bandyopadhyay SK. Healing properties of some Indian medicinal plants against indomethacin-induced gastric ulceration of rats. J Clin Biochem Nutr 2007;41:106:14.
- Biswas K, Bandyopadhyway U, Chattopadhyay I, Varadoraj A, Aji E, Banerjee RK. A novel antioxidant and antiapoptotic role of omeprazole to block gastric ulcer through scavenging of hydroxyl radical. J Biol Chem 2003;278:10993-1001.
- Berndt V, Gotz E, Schonlenben K, Langhans P. Stress-induced peptic ulcer: pathogenesis, clinical features, prevention and treatment (author’s transl). Pract Anaesth 1978;13:108-22.
- Okoli AS, Okeke MI, Iroegbu CU, Ebo PU. Antibacterial activity of Harunganamadagascariensis leaf extracts. Phytother Res 2002;16:174-9.
- Lorke D. A new approach to practical acute toxicity. Arch Toxicol 1983;54:275-87.
- Nwafor PA, Okwuasaba FR, Binda LG. Antidiarrhoeal and antiulcerogenic effects of methanolic extract of Asparagus pubescens root in rats. J Ethapharmacol 2000;72:421-7.
- Evbuonwa MI, Baleruipwa AF. Effects of diet on indomethacin-induced ulceration in pregnant rat. Niger J Physiol Sci 1990;6:189-91.
- Suzuki Y, Hayachi M, Itom M, Vamagani T. Anti-ulcer effects of 4’-(2-carboxyetyl) phenyl trans-4-aminomethyl 1976;26:471-80.
- Okwuosa C, Unekwe PC, Nwobodo E, Chilaka K. Antiulcer properties of leaf extract of Combretumracemosun. J Biomed Invest 2006;4:8-14.
- Ibrahim MB, Owonubi MO, Onaolapu JA. Antimicrobial effects of extract of leaf, stem and root bark of Anoglassusleicarpus on Staphylococcus aurous (NCTC 9198), E. Coli (NCTC10418) and Proteus vulgaris (NCTC 4636). J Pharm Res Drug Dev 1998;2:20-6.
- Parekh J, Chanda SV. Antibacterial activity of aqueous and alcoholic extracts of 34 Indian medicinal plants against some staphylococcus species. Turk J Biol 2008;32:63-71.
- Mohammedi Z, Atik F. Impact of solvent extraction type on total phenols content and biological activity from Tamarixaphylla (L). International Journal of Pharma and Bio Sciences 2011;2:609-12.
- Dai J, Munper RJ. Plant phenolics: extraction, analysis and their antioxidant and anticancer properties. Molecules 2010;15:7313-52.
- National Academy of Science. Principles for evaluating chemical in the environment. Washington DC: National Academy of Science; 1975.
- Venkataranganna MV, Gopumadhaun S, Sunderam R, Mitra SK. Evaluation of possible mechanism of antiulcerogenic activity of UL-90, a herbal prepration. J Ethnopharmacol 1998;6:225-61.
- Hawkey CJ. Prostaglandins: mucosal protection of peptic ulceration. Methods Find Exp Clin Pharmacol 1989;11 Suppl 1:45-51.
- Dajani EZ, Agrawal NM. Prevention and treatment of ulcers induced by NSAIDS: an update. J Physiol Pharmacol 2005;45:3-16.
- Best R, Lewis DA, Nasser N. The anti-ulcerogenic activity of the unripe plantain banana. (Musa species). Br J Pharmacol 1984;82:107-16.
- Ibu JO, Ezemuzie IC, Nwankwoala RN. Pathophysiology and management of peptic ulcer in West Africa: G review. J Exp Clin Sci 1994;2:1-11.
- Al-Mashhadani WM, Karin K, Al-Taie RI, Al-Zahawi HM. NiFedipine versus cimetidine on prevention of stress-induced gastric ulcer in rats. Eur J Pharmacol 1997;192:128-40.
- Barndyopadhyay U, Das D, Barndyipadhyay D, Bhattaacharejee M, Banerjee RK. Antisecretory therapy for bleeding peptic ulcer. Curr Sci 1999;76:55-63.
- Bertaccini G, de Cadliglione R, Scarpignato C. Effect of substance P and its natural analogues on gastric emptying of the conscious rat. Br J Pharmacol 1981;72:221-3.
- Akah PA, Orisakwe OE, Gamaniel SK, Shittu A. Evaluation of Nigerian traditional medicines: II. Effects of some Nigerian folk remedies on peptic ulcer. J Ethnopharmacol 1998;62:123-7.
- Lam SK. Use of cytoprotective agents in the treatment of gastric ulcers. Med J Aust1985;142:S21-3.
- Armold R. Safety of proton pump inhibitors-an overview. Aliment Pharmacol Ther 1994;8:65-70.
- Mohammed AH, Hunt RH. The rational of acid suppression in the treatment of acid-related disease. Aliment Pharmacol Ther 1994;8:3-10.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.