First Case Report of Costello Syndrome in the Azeri Turkish Population
2 Genetic Counseling Center of Allameh Amini, State Welfare Organization of Tabriz, Iran
3 Department of Medical Genetics, Faculty of Medicine, Tabriz University of Medical Sciences, Tabriz, Iran
4 Division of Medical Genetics, Tabriz Children’s Hospital, University of Medical Sciences, Tabriz, Iran, Email: Rezazadeh.mary@gmail.com
Maryam Rezazadeh, Division of Medical Genetics, Tabriz Children’s Hospital, University of Medical Sciences, Golghasht St,Tabriz, Iran, Tel: +9833376308, Email: Rezazadeh.mary@gmail.com
Citation: Vahedi L, et al. First Case Report of Costello Syndrome in the Azeri Turkish Population. Ann Med Health Sci Res. 2017; 7: 435-437
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Background: Costello syndrome as a complex multi organ system anomalies caused by heterozygous de novo missense mutations in the proto oncogene HRAS which its product involved in controlling cell division and growth. Typical features that characterize this phenotype include distinctive coarse facial features, craniofacial anomalies, macrocephaly, low body mass gain, delayed development, short stature, skeletal and cardiac abnormalities with tumor predisposition, especially rhabdomyosarcoma. Aim: We here report one pediatric patients with Costello syndrome and umbilical ligament rhabdomyosarcoma. In this article, we report a case study of an 8-year-old boy born of a non-consanguineous marriage and presenting with mental retardation and delayed development. Physical examination revealed loss of speech, repetitive hand wringing movement, short stature (120 cm), low-set ears, strabismus in eyes, microcephaly [head circumference (HC) = 49 cm] and autistic behaviour. The clinical findings prompted a suspicion of CS. Genetic testing provided a proof with G12S mutation in the exon 2 of HRAS gene. The current work aim was to report the first case of Costello syndrome in the Azeri Turkish population.
Keywords
Costello syndrome; HRAS; Facio-Cutaneous-Skeletal (FCS) syndrome
Introduction
Costello syndrome (CS) (OMIM #218040), also known as faciocutaneoskeletal syndrome or FCS syndrome, is a rare mental retardation and multiple congenital abnormality syndrome [1] was described by by Dr Jack Costello in 1971 and 1977 years at the first time [2] with an estimated more than 300- 400 medical cases worldwide [3] with prevalence range from 1 in 300,000 to 1 in 1.25 million. [2,4]
It mostly reported by severe feeding difficulties with postnatal growth failure, macrocephaly, a typical coarse face, short neck, developmental delay, psychomotor retardation, sparse curly hair, dark skin, neurologic abnormalities, cardiac defects (congenital heart disease, cardiac arrhythmia, heart abnormalities, hypertrophic cardiomyopathy) and cutaneous anomalies and increased risk of tumors such as nasal papillomas. [1,5,6]
It has an autosomal dominant inheritance pattern caused by mutations in Harvey rat sarcoma viral oncogene homolog (HRAS), located on chromosome 11p15.5 [7,8]. RAS as a proto ontogenesis part of a pathway that participates in normal cell growth and division. [9,10] In addition to, mutations have been identified in the KRAS and BRA genes in the number of patients. [11] Different inheritance pattern such as sporadic autosomal dominant in people with no history of the disorder in their family, autosomal recessive inheritance and gonadal mosaiesm have been reported. [12-14] Detected mutations show a gain-of-function manner in the abnormal expressed protein and cause to hyper activation of the mitogen activated protein kinase (MAPK) pathway. [15]
Here, we report an 8 year old male clinically diagnosed as Costello syndrome and discussed the management of cardiac arrhythmia, one of its life threatening complications. Also, we had a brief review of the literature about this clinical condition.
Case Presentation
History
An 8-year-old Iranian boy, an only child born of a nonconsanguineous marriage, was referred to our Medical Genetic Centre (Tabriz, Iran) in 2011 due to manifestation of developmental and growth delay. The parents were healthy unrelated Azeri Turkish. He was born to a 20 year old mother after a pregnancy complicated by polyhydroamnios and cloudy amniotic fluid at 28 weeks and due to vaginal delivery. Her father was 34 years old at his birth. The parents were healthy and alert. Birth weight was 4300 gr and Apgar score was low because of that he had been admitted with respiratory distress over 1 week in hospital. His parents mentioned mental retardation in cousin of mother. The pedigree chart using Progeny 10 is shown in Figure 1.
At one age, because of developmental and growth delay, he was screened by a pediatric specialist. The Brain-CT-Scan was reported mild cerebral atrophy with greater intensity in the right hemisphere. Electromyogram (EMG) and nerve conduction velocity (NCV) tests were normal so cerebral palsy (CP) was detected. With increasing age and the presentation of coarse face appearance, Mucopolysaccharidoses (MPS) diseases kind of hunter syndrome was considered and he had been referred to the pediatric endocrinologist and the diagnostic tests of Heparan sulfate, Dermatan sulfate, Keratan sulfate was requested but was not done by the parents. Urine MPS Test was requested by pediatric specialist that was negative. X-Ray skeletal survey was not shown MPS and only coaxes valgus of neck and body femur was reported in X-ray of the wrist, disorder growth of epiphysis of hands, done growth delayed enough one age was reported so with findings and disorder movement prompted suspicions of CP. Finally, he had been referred to Medical Genetic Centre. At visiting, he was 8 years. Her mother seems good appearance, tall height and very alert. She answered carefully to questions. His father believed that he had not any problem but mild coarse face was showed in appearance.
He was quiet and too shy when have been visited by our group. He realized questions but he could not answer to our questions because of speech problems. Mathematic intelligent was very weak and he was studied in school for children with special problems because of the lack of progress in education. He had an emotional personality and delayed developmental retardation.
Clinical examination
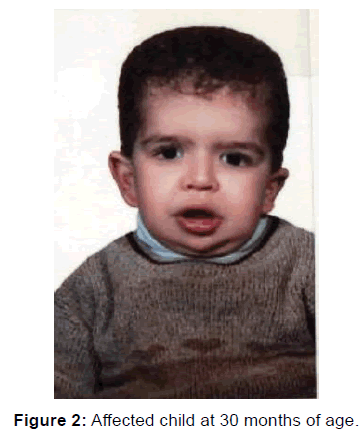
Clinical examinations forhead circumference (HC=40.5), height (92 cm) and weight (19 kg) remained below the 3rd centile for height and weight. Macrocephaly, coarse face, curly, sparse hair, thick eyebrows, eye (epicantal folds), nose (short bulbous nose, depressed nasal bridge), mouth (large mouth, thick lips, macroglossia, teeth abnormalities), ear (Low set ears), skin (Loose of skin), inguinal hernia bilateral, musculoskeletal (hyper extensible fingers, deep plantar, deep palmar, lesions of hyperkeratolic of palms and soles), hoarse voice, increased anteroposterior diameter of chest. Heart auscultation was normal but auscultation of the lungs includes hoarseness [Figure 2]. Given the history and clinical signs, CT-Scan was requested again and hydrocephalus was reported. He suspected that he might be Costello syndrome. The molecular analysis has been done in German-lab and Costello syndrome was confirmed. After several years, his parents decided to get pregnant again. During pregnancy, amniocentesis and Chorionic Villus Sampling (CVS) were negative and ultimately a normal girl was born. At the last visit, being 13 years old, physical examination had microcephaly (HC = 45 cm), short stature (139 cm), low body weight (34 kg). Their daughter was normal child (3 years old).
Method
Genetic testing
Genomic DNA was isolated from peripheral blood in collection tubes contained EDTA by salting out method [16] and stored at –80°C. HRAS is located in 11p15.5 location. CS is caused by any of at least five different mutations in the HRAS. We have analyzed sequence in the institute Zentrum für Medizinische Genetik in Osnabrück, Germany. Direct sequencing analysis of HRAS exons revealed the mutation Gly12-Ser in the exon 2 that have been reported previously as a causative missense mutation for the CS. [7]
Discussion
Costello syndrome is a rare congenital abnormality syndrome [2,17] estimated prevalence for this syndrome is 1: 1,290,000 individuals in eastern Asia and at least 1:500,000 in the United Kingdom. [18] CS is an autosomal dominant inherited phenotype with frequent de novo mutation in germ line. [11] It have been previously reported that RAS/MAPK pathway signaling dysregulationhas major impact on skeletal development, bone homeostasis and modeling. [19,20] Affected patients with RASopathies present an overlapping musculoskeletal phenotype such as spine curvature (scoliosis, kyphosis), tight tendons, deformity in chest (pectuscarenatus/excavatum), contractures (elbow, wrist, hip, knee, ankle) and different abnormalities in feet (pesplanus/cavus, toe crowding). [18,21,22] Clinically, similar to what is observed in other RASopathies, individuals affected by CS causative HRAS mutations promote increased signal flow through the RAF–MEK–ERK and PI3K–AKT signaling cascades. While we can detect decreased bone mineralization in other RASopathies, such as neurofibromatosis type 1 and Noonan syndrome, systematic studies investigating bone mineral density (BMD) are not recognizable in CS. [23]
This syndrome is characterized by two phases: the first phase with polyhydramnios and increased birth weight [13] and the second phase with the presence or absence of malignancy, and appearance difficulties and other disorders. [2] Postnatal failure to thrive and short stature happened in many infants with CS despite this characters may be large or normal at birth and natural history. [17,24,25]
Conclusion
Therefore, CS is a multisystem disorder caused by germline mutations in the HRAS gene. Clinical manifestations of affected patients by syndrome compose of feeding difficulties, insufficient weight gain, poor suction power and infants may be born either at full-term with normal and high weight. Macrocephaly, coarse face, large mouth with thick lips or macroglossia, frizzy and sparse hair, strabismus, epicanthic fold, thick eyebrows, low nasal bridge and low set ears, loose of skin, hoarse voice, short stature, unusually flexible joints and inguinal hernia bilateral frequent were reported in these children. Heart and respiratory system can involve in CS. furthermore, clinical signs may be highly variable such as neuropsychomotor development delay and most patients have significant mental retardation with intellectual impairment. CS is considered in the differential diagnosis with other disease because of Overlapping clinical manifestations. We focused on neurodevelopmental disorders that have been refereed to our research centers and this work has been done in continuous of previous work. [26]
Acknowledgement
We would like to thank State Welfare Organization and Liver & Gastrointestinal Disease Research Center in Tabriz, Dr. Yousef Shafeghati and institute Zentrum für Medizinische Genetik in Osnabrück in Germany.
Ethical standards
Ethical aspects were considered while obtaining permission and informed consent form the parents of the child for the study and the name of the child was kept anonymous.
Conflict of Interest
All authors disclose that there was no conflict of interest.
REFERENCES
- Kerr B, Mucchielli ML, Sigaudy S, Fabre M, Saunier P, Voelckel MA, et al. Is the locus for Costello syndrome on 11p? J Med Genet 2003; 40: 469-471.
- Kerr B, Delrue MA, Sigaudy S, Perveen R, Marche M, Burgelin I, et al. Genotype-phenotype correlation in Costello syndrome: HRAS mutation analysis in 43 cases. J Med Genet 2006; 43: 401-405.
- Beukers W, Hercegovac A, Zwarthoff EC. HRAS mutations in bladder cancer at an early age and the possible association with the Costello Syndrome. Eur J Hum Genet 2014; 22: 837-839.
- Dileone M, Profice P, Pilato F, Alfieri P, Cesarini L, Mercuri E, et al. Enhanced human brain associative plasticity in Costello syndrome. J Physiol 2010; 588: 3445-3456.
- Say B, Güçsavas M, Morgan H, York C. The Costello syndrome. Am J Med Genet A 1993; 47: 163-165.
- Kerr B, Eden OB, Dandamudi R, Shannon N, Quarrell O, Emmerson A, et al. Costello syndrome: two cases with embryonal rhabdomyosarcoma. J Med Genet 1998; 35: 1036-1039.
- Zampino G, Pantaleoni F, Carta C, Cobellis G, Vasta I, Neri C, et al. Diversity, parental germline origin, and phenotypic spectrum of de novo HRAS missense changes in Costello syndrome. Hum Mutat 1997; 28: 265-272.
- Da Rosa Pereira AP, Grunewald STF. Costello syndrome: A case report and review of diagnostic approach. Residência Pediátrica 2016; 6: 141-144.
- Rauen K. HRAS and the Costello syndrome. Clinical genetics 2007; 71: 101-108.
- Gripp KW, Lin AE. Costello syndrome: a Ras/mitogen activated protein kinase pathway syndrome (rasopathy) resulting from HRAS germline mutations. Genet Med 2012; 14: 285-292.
- Sol-Church K, Stabley DL, Demmer LA, Agbulos A, Lin AE, Smoot L, et al. Male-to-male transmission of Costello syndrome: G12S HRAS germline mutation inherited from a father with somatic mosaicism. Am J Med Genet A 2009; 149: 315-321.
- Johnson JP, Golabi M, Norton ME, Rosenblatt RM, Feldman GM, Yang SP, et al. Costello syndrome: Phenotype, natural history, differential diagnosis, and possible cause. J Pediatr 1998; 133: 441-448.
- Hinek A, Smith AC, Cutiongco EM, Callahan JW, Gripp KW, Weksberg R. Decreased elastin deposition and high proliferation of fibroblasts from Costello syndrome are related to functional deficiency in the 67-kD elastin-binding protein. Am J Hum Genet 2000; 66: 859-872.
- Hennekam RC. Costello syndrome: An overview. Am J Med Genet C Semin Med Genet 2203; 117: 42-48.
- Nava C, Hanna N, Michot C, Pereira S, Pouvreau N, Niihori T, et al. Cardio-facio-cutaneous and Noonan syndromes due to mutations in the RAS/MAPK signalling pathway: genotype–phenotype relationships and overlap with Costello syndrome. J Med Genet 2007; 44: 763-7 71.
- Morata P, Queipo-Ortuño MI, De Dios Colmenero J. Strategy for optimizing DNA amplification in a peripheral blood PCR assay used for diagnosis of human brucellosis. J Clin Microbiol 1998; 36: 2443-2446.
- Gripp KW. Tumor predisposition in Costello syndrome. Am J Med Genet C Semin Med Genet 2005; 137: 72-79.
- Abe Y, Aoki Y, Kuriyama S, Kawame H, Okamoto N, Kurosawa K, et al. Costello and CFC syndrome study group in Japan. Prevalence and clinical features of Costello syndrome and cardio-facio-cutaneous syndrome in Japan: Findings from a nationwide epidemiological survey. Am J Med Genet A 2012; 158: 1083-1094.
- Yu X, Chen S, Potter OL, Murthy SM, Li J, Pulcini JM, et al. Neurofibromin and its inactivation of Ras are prerequisites for osteoblast functioning. Bone 2005; 36: 793-802.
- Stevenson DA, Schwarz EL, Viskochil DH, Moyer-Mileur LJ, Murray M, Firth SD, et al. Evidence of increased bone resorption in neurofibromatosis type 1 using urinary pyridinium crosslink analysis. Pediatr Res 2008; 63: 697-701.
- Stevenson DA, Schwarz EL, Carey JC, Viskochil DH, Hanson H, Bauer S, et al. Bone resorption in syndromes of the Ras/MAPK pathway. Clin Genet 2011; 80: 566-573.
- Yassir WK, Grottkau BE, Goldberg MJ. Costello syndrome: Orthopaedic manifestations and functional health. J Pediatr Orthop 2003; 23: 94-98.
- Rauen KA, Huson SM, Burkitt-Wright E, Evans DG, Farschtschi S, Ferner RE, et al. Recent developments in neurofibromatoses and RASopathies: management, diagnosis and current and future therapeutic avenues. Am J Med Genet A 2015; 167: 1-10.
- Gripp KW, Scott CI Jr, Nicholson L, McDonald-McGinn DM, Ozeran JD, Jones MC, et al. Five additional Costello syndrome patients with rhabdomyosarcoma: proposal for a tumor screening protocol. Am J Med Genet 2002; 108: 80-87.
- Philip N, Sigaudy S. Costello syndrome. J Med Genet 1998; 35: 238-240.
- Gharesouran J, Khalili AF, Azari NS, Vahedi L. First case report of Rett syndrome in the Azeri Turkish population and brief review of the literature. Epilepsy Behav Case Rep 2015; 3: 15-19.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.