Gradenigo’s Syndrome: A Review
Citation: Gore MR. Gradenigo’s Syndrome: A Review. Ann Med Health Sci Res. 2018;8:220-224
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Background: Gradenigo’s syndrome is an uncommon triad of retroorbital pain, diplopia due to 6th nerve palsy, and otorrhea due to otitis media and petrous apicitis. Although rarer in the antibiotic era, this syndrome may be life-threatening and highly morbid if not treated aggressively with IV antibiotics, and surgery in some cases. Methods and Findings: Herein we present a review of patients reported in the literature since 1990, with an exploration of medical vs. surgical treatment, causative organisms, and patient outcomes. Thirty-seven studies representing 38 patients were identified. The mortality rate was 2.6%, and the majority of patients were treated with either IV antibiotics alone (14) or IV antibiotics in addition to surgery (14). Conclusions: Prompt awareness of Gradenigo’s syndrome can help avoid morbidity or mortality related to this uncommon phenomenon. It should be kept in the differential of any patient with otitis media, retroorbital pain, and diplopia.
https://blogum.blogaaja.fi/
https://blogum-1.jimdosite.com/
https://blogummm.edublogs.org/
https://blogummm.websites.co.in/
https://blogum18.wordpress.com/
https://benim-blogum.jigsy.com/
https://fuiegs-symbeaurds-build.yolasite.com/
https://blogum-03.webselfsite.net/
https://blogummm.mystrikingly.com/
https://blogum.splashthat.com/
https://blogum3.webnode.com.tr/
https://blogum.odoo.com/
https://blogum.creatorlink.net/
https://whiteseotr1-s-site.thinkific.com/enrollments
https://blogum.estranky.cz/
https://653ba4fbb538c.site123.me/
https://blogum12m.blogspot.com/
https://blogum.hashnode.dev/
https://whiteseoturkey1.wixsite.com/blogum
https://sites.google.com/view/blogummm/
https://codepen.io/blogum
https://blogumm.livejournal.com/
https://wakelet.com/@blogum82816
https://www.homify.com/users/9538383/blogum
https://lessons.drawspace.com/profile/323613/blogum
https://my.desktopnexus.com/blogum/
https://writeupcafe.com/profile/BLOGUM/
https://www.pearltrees.com/blogum
https://www.easyfie.com/blogum
https://pharmahub.org/members/27615/profile
https://www.zupyak.com/u/blogum/posts
https://www.metroflog.co/blogum
https://www.fuzia.com/fz/blogum-blogum
https://tr.pinterest.com/blogum12/
https://my.getjealous.com/blogum
https://micro.blog/blogum
https://www.tumblr.com/blogummm
https://hub.docker.com/u/blogum
https://fire.blogfree.net/?act=Profile&MID=1342323
https://blogum.pixnet.net/blog
https://www.threadless.com/@blogum/activity
https://blogum.neocities.org/
https://blogum12.amebaownd.com/
https://teletype.in/@blogum
https://ubl.xml.org/users/blogum
https://educatorpages.com/site/blogum/
https://blogum.onlc.fr/
Keywords
Gradenigo’s syndrome; Systematic review; Petrositis; Abducens palsy
Introduction
Gradenigo first described the syndrome that bears his name in 1904 and later elaborated on the triad in 1907. [1,2] The triad classically consists of retroorbital or periauricular pain due to inflammation/irritation of the first (ophthalmic) division of the trigeminal nerve, diplopia due to inflammation of the abducens (6th cranial) nerve in Dorello’s canal, and a draining ear due to a florid otitis media causing petrous apicitis. In the pre-antibiotic era, this condition was highly morbid and carried a high rate of mortality. Even in the antibiotic era, there are occasional reports of this serious condition. Petrous apicitis traditionally warranted surgical exploration to drain the infected middle ear, mastoid, and petrous apex, but with advances in imaging and antibiotic therapy, many patients can be managed conservatively with IV antibiotics, especially if the constellation of symptoms is recognized early. This article summarizes 37 studies published since 1990, encompassing 38 patients, and summarizes the medical and surgical management of Gradenigo’s syndrome, comorbid conditions, patient demographics, and trends in management of this uncommon syndrome with a review of the literature.
Patients and Methods
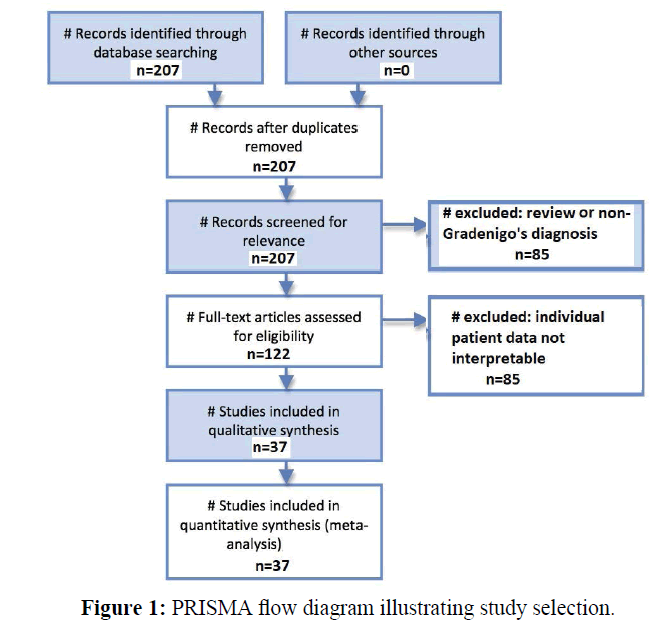
A PubMed database search was performed using the keywords “Gradenigo syndrome”. A total of 207 manuscripts were initially identified since 1990. After reviewing these articles, 85 were excluded as they were review articles or did not report data on patients with an actual diagnosis of Gradenigo’s syndrome with the classically described Gradenigo’s triad. An additional 85 of the remaining articles were excluded as they did not contain interpretable data or individual patient data that could be analyzed. Figure 1 illustrates the PRISMA flow chart for analysis and selection of the available articles. [3] The remaining 37 studies totaled 38 patients with individual data available. [4-40] Patient data was collected on age, sex, treatment modality (medical, surgical, or both), drugs used in treatment, sequelae of the disease, specific organisms isolated by culture, and any comorbidities present.
Results
Average patient age was 22.4 years (standard deviation=21 years). Of patients for who sex was reported there were 17 male patients and 17 female patients. Table 1 lists the data on patient age and sex. Four patients had neoplasms of the temporal bone that precipitated the Gradenigo’s: one solitary osseous plasmacytoma, one embryonal rhabdomyosarcoma, one diffuse giant B-cell non-Hodgkin’s lymphoma, and one well-differentiated nasopharyngeal squamous cell carcinoma. There was only one death reported, from brain abscess, giving a mortality rate of 2.6%. Two patients were reported to have bilateral Gradenigo’s syndrome, and there was one pregnant patient reported. There was one HIV-positive patient reported, one patient with cavernous sinus thrombosis, and one patient with temporary ipsilateral facial weakness/facial nerve dysfunction. Of the patients for whom culture results were reported, four had negative cultures; one grew Streptococcus intermedius, one Streptococcus pneumoniae, one Streptococcus acidominus, one Mycobacterium tuberculosis, and one aspergillus. Table 2 summarizes the patient data on treatment modality. Fourteen patients were treated with IV antibiotics only, while another 14 were treated with IV antibiotics in addition to some form of surgery. One patient with a neoplasm was treated with radiation treatment alone, while another patient with a neoplasm was treated with a craniotomy in addition to radiation therapy. Ceftriaxone was the most common IV antibiotic reported, with levofloxacin, metronidazole, ceftazidime, and clindamycin also utilized, typically in addition to ceftriaxone.
| Reference | Patient age (years) | Patient sex |
|---|---|---|
| Vitale | 8 | Male |
| Kazemi | 33 | Male |
| Jensen | 9 | Female |
| Ghani | 7 | Male |
| Karunakaran | 13 | Male |
| Janjua | 10 | Female |
| Lattanzi | 60 | Female |
| Valles | 36 | Female |
| Khalatbari | 45 | Male |
| Plodpai | 63 | Male |
| Choi | 8 | Female |
| Colpaert | 12 | Female |
| Bhatt | 72 | Male |
| Delgado | 28 | Female |
| Ricks | 5 | Male |
| Pedroso | 33 | Female |
| Fernández-Mayoralas | 8 | Female |
| Rossor | 11 | Female |
| Guedes | 6 | Male |
| Zengel | 12 | Male |
| Sethi | 11 | Unknown |
| Burston | 6 | Unknown |
| Burston | 70 | Male |
| Sherman | unknown | Male |
| Bloching | 13 | Female |
| Yozu | 6 | Female |
| Trimis | 12 | Female |
| Finkelstein | Unknown | Unknown |
| Penas-Prado | 53 | Male |
| Marianowski | 6 | Unknown |
| Minotti/Kountakis | Unknown | Female |
| Minotti/Kountakis | Unknown | Female |
| Hananya | 5 | Male |
| Dave' | 4 | Male |
| Morales | 44 | Male |
| Murakami | 8 | Female |
| Tutuncuoglu | 13 | Male |
| Hehl | 30 | Female |
Table 1: Summary of age and sex data in patients with Gradenigo’s syndrome.
| Treatment | Number of patients |
|---|---|
| IV antibiotics only | 14/37 |
| IV antibiotics + mastoidectomy | 3/37 |
| IV antibiotics + mastoidectomy + petrous apicectomy | 2/37 |
| Radiation therapy | 1/37 |
| IV antibiotics + mastoidectomy + craniotomy | 2/37 |
| IV antibiotics + tympanostomy tubes | 6/37 |
| Subtemporal/middle fossa craniotomy + radiation therapy | 1/37 |
Table 2: Summary of treatment modalities in patients with Gradenigo’s syndrome.
Discussion
In the post-antibiotic era Gradenigo’s syndrome is rare but can still prove life-threatening and potentially morbid. In their recent 40-year series of 44 patients with petrous apicitis Gadre and Chole [41] noted that only six patients presented with the full classical Gradenigo’s syndrome triad, and they noted a decreasing prevalence of patients needing surgery. They noted only one death in their series, consistent with the low mortality rate found in the present review. Kazemi [42] also recently reported on a young male adult patient with the classic Gradenigo’s triad who responded dramatically to IV clindamycin and ceftazidime with resolution of all Gradenigo’s symptoms by day four of antibiotic treatment.
Jensen [43] reported a retrospective series of four patients with Gradenigo’s syndrome. In three patients the syndrome presented as the classic acute triad, but interestingly one patient presented with a delayed abducens nerve palsy relapse 6 years after the onset of chronic suppurative otitis media. Govea- Camacho and coworkers [44] reported a series of 5 patients aged 17–52 with complicated otitis media. Although several developed complications such as brain abscess, cavernous sinus thrombosis, meningitis, or Bezold abscess, none of the five developed the classic Gradenigo’s triad.
Reddy and coworkers [45] published an in-depth exploration of the relationship between the work of Primo Dorello, Giuseppe Gradenigo, Wenzel Leopold Gruber, and Harris Holmes Vail and our current understanding of the relationship between Dorello’s canal and the abducens nerve palsy seen in Gradenigo’s syndrome. Gruber, an anatomist, was the first to describe Dorello’s canal in 1859. Dorello’s canal is a bony/ fibrous conduit made by a depression near the petrous tip. It is located behind the petrosphenoidal ligament and lies between the apex of the petrous portion of the temporal bone and the clivus. The 6th cranial nerve passes through Dorello’s canal while traveling through the petroclival region of the skull base. In 1904, Gradenigo first published descriptions of the triad of symptoms that bears his name: purulent and suppurative otitis media, retroorbital or periauricular pain due to trigeminal neuralgia, and palsy of the ipsilateral abducens nerve causing lateral rectus muscle paralysis. Gradenigo proposed that the mechanism of ipsilateral abducens nerve palsy was inward spread of the otitis media causing leptomeningeal inflammation that damaged the sixth cranial nerve, resulting in the abducens palsy. Primo Dorello published descriptions of the intracranial path of the abducens nerve. Dorello proposed that the abducens palsy seen in Gradenigo’s syndrome was actually due to passage of middle ear infection through tympanic veins to the petrosal sinus, causing inflammation and compression of the 6th cranial nerve at the narrow, anatomically vulnerable Dorello’s canal through which the abducens nerve passes on its way to innervate the lateral rectus muscle of the eye. This is consistent with the frequent resolution of the diplopia/lateral rectus paralysis once the concomitant apicitis/otitis media resolves and the localized compression of the abducens nerve is relieved. Later, in the 1920’s, American physician Harris Holmes reported his observations regarding the anatomy of Dorello’s canal in 8 specimens. He published the first English-language descriptions confirming Dorello’s descriptions of the petrous apex, petrosphenoidal ligament, and the path of the abducens nerve in relation to Dorello’s canal. His descriptions were the first widely recognized anatomical studies to use the term “Dorello’s canal” and Vail’s work popularized the eponym used today. He reiterated that the anatomical location of Dorello’s canal made the 6th nerve vulnerable to inflammatory swelling and compression in petrous apicitis/otitis media, producing the characteristic ipsilateral lateral rectus paralysis seen in Gradenigo’s syndrome.
The present study demonstrated that patients with Gradenigo’s syndrome tend to be young (average patient age=22.4 years in the present study), and male:female distribution is equal. The mortality rate was low, only 2.6%. Infection was the typical etiology but in rare cases malignancy of the temporal bone precipitated the infection leading to Gradenigo’s syndrome. The causative organisms tended to be Streptococcus species, but also included more rare microbes such as aspergillus or Mycobacterium tuberculosis. Treatment was roughly evenly distributed between antibiotic treatment alone, and antibiotic treatment combined with surgical intervention. Fourteen of 37 patients were treated with IV antibiotics alone, with one patient with a temporal bone malignancy being treated with radiation therapy alone. The surgical treatments in addition to IV antibiotic therapy in the study ranged from simple tympanostomy tubes is six patients, to mastoidectomy alone in three patients, mastoidectomy combined with removal of the petrous apex bone in two, mastoidectomy combined with temporal craniotomy in two, and combined sub-temporal and middle fossa craniotomy along with post-operative radiation therapy in one patient. This demonstrates that the aggressiveness of surgical treatment should be tailored to the clinical course, comorbid or precipitating factors, and radiographic findings in each case. Gradenigo’s syndrome should be kept in the differential for patients with otologic infections, particularly if 6th nerve palsy is present. Imaging with CT or MRI should be used to confirm the clinical findings and evaluate for associated malignancy or abscess, and aggressive IV antibiotic treatment combined with surgery when necessary should be implemented as soon as possible.
Conclusion
Though relatively rare in the post-antibiotic era, Gradenigo’s syndrome can be highly morbid and even fatal if not recognized. Antibiotics remain the primary treatment modality, but surgery may be necessary in cases failing to respond to antibiotics or cases with associated skull base or intracranial abscess. In the present review, about 50% of the patients recovered with IV antibiotics alone, and most patients requiring surgery underwent tympanostomy tube insertion or mastoidectomy. Mortality was low and most patients recovered without sequelae. Gradenigo’s should be kept in the differential for patients experiencing diplopia in the setting of facial/periauricular pain and otitis media.
Conflict of Interest
The authors disclose that they have no conflicts of interest.
REFERENCES
- Gradenigo G. Ueber Circumscripte Leptomeningitis mit spinallen Symptomen. Arch Ohrenhelik, 1904;51:60-62.
- Gradenigo G. Uber die Paralyse des Nervusabducens bei Otitis Gradenigo, G. Arch Ohrenheilkunde, 1907;774:149-187.
- Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA Statement. Open Med. 2009;3:e123-e130.
- Vitale M, Amrit M, Arora R, Lata J. Gradenigo's syndrome: A common infection with uncommon consequences. Am J Emerg Med. 2017;35:1388.e1-1388.e2.
- Karunakaran T, Kaneshamoorthy M, Harris R. Clivus erosions following Gradenigo's syndrome-mastoiditis causing VI nerve palsy. BMJ Case Rep. 2016.
- Janjua N, Bajalan M, Potter S, Whitney A, Sipaul F. Multidisciplinary care of a paediatric patient with Gradenigo's syndrome. BMJ Case Rep. 2016.
- Lattanzi S, Cagnetti C, Di Bella P, Provinciali L. Mystery case: Cholesterol granuloma of the petrous apex in Gradenigo syndrome. Neurology. 2015;84:e122-e123.
- Valles JM, Fekete R. Gradenigo syndrome: unusual consequence of otitis media. Case Rep Neurol. 2014;6:197-201.
- Khalatbari MR, Hamidi M, Moharamzad Y. Gradenigo's syndrome as first presentation of solitary osseous plasmacytoma of the petrous apex. Arch Iran Med. 2014;17:526-528.
- Plodpai Y, Hirunpat S, Kiddee W. Gradenigo's syndrome secondary to chronic otitis media on a background of previous radical mastoidectomy: A case report. J Med Case Rep. 2014;8:217.
- Choi KY, Park SK. Petrositis with bilateral abducens nerve palsies complicated by acute otitis media. Clin Exp Otorhinolaryngol. 2014;7:59-62.
- Colpaert C, Van Rompaey V, Vanderveken O, Venstermans C, Boudewyns A, Menovsky T, et al. Intracranial complications of acute otitis media and Gradenigo's syndrome. B-ENT. 2013;9:151-156.
- Bhatt YM, Pahade N, Nair B. Aspergillus petrous apicitis associated with cerebral and peritubular abscesses in an immunocompetent man. J Laryngol Otol. 2013;127:404-407.
- Delgado ME, Del Brutto OH. Teaching neuroimages: Gradenigo syndrome. Neurology. 2012;79:e141.
- Jacobsen CL, Bruhn MA, Yavarian Y, Gaihede ML. Mastoiditis and Gradenigo's Syndrome with anaerobic bacteria. BMC Ear Nose Throat Disord. 2012;12:10.
- Ricks RG, Hollis L. Embryonic rhabdomyosarcoma presenting as Gradenigo's syndrome. BMJ Case Rep. 2012.
- Pedroso JL, De Aquino CC, Abrahão A, De Oliveira RA, Pinto LF, Bezerra ML, et al. Gradenigo's Syndrome: Beyond the Classical Triad of Diplopia, Facial Pain and Otorrhea. Case Rep Neurol. 2011;3:45-47.
- Fernández-Mayoralas DM, Fernández-Jaén A, Bodegas Canora I, RodrÃÂguez Cuitino LA, Hernández CalvÃÂn FJ. Gradenigo's syndrome: The importance of general pediatric examination. An Pediatr (Barc). 2011;75:205-206.
- Rossor TE, Anderson YC, Steventon NB, Voss LM. Conservative management of Gradenigo's syndrome in a child. BMJ Case Rep. 2011.
- Kong SK, Lee IW, Goh EK, Park SE. Acute otitis media-induced petrous apicitis presenting as the Gradenigo syndrome: successfully treated by ventilation tube insertion. Am J Otolaryngol. 2011;32:445-447.
- Guedes V, Gallegos P, Ferrero A, GarcÃÂa Minúzzi M, Casanovas A, Georgetti B, et al. Gradenigo's syndrome: a case-report. Arch Argent Pediatr. 2010;108:e74-e75.
- Zengel P, Wiekström M, Jäger L, Matthias C. Isolated apical petrositis: An atypical case of Gradenigo's syndrome. HNO. 2007;55:206-210.
- Sethi A, Sabherwal A, Gulati A, Sareen D. Primary tuberculous petrositis. Acta Otolaryngol. 2005;125:1236-1239.
- Burston BJ, Pretorius PM, Ramsden JD. Gradenigo's syndrome: successful conservative treatment in adult and paediatric patients. J Laryngol Otol. 2005;119:325-329.
- Ghani S, Likeman M, Lyttle MD. New onset strabismus in association with ear pain. Arch Dis Child Educ Pract Ed. 2017;102:267-269.
- Karunakaran T, Kaneshamoorthy M, Harris R. Clivus erosions following Gradenigo's syndrome-mastoiditis causing VI nerve palsy. BMJ Case Rep. 2016.
- Sherman SC, Buchanan A. Gradenigo syndrome: A case report and review of a rare complication of otitis media. J Emerg Med. 2004;27:253-256.
- Bloching M, Heider C, Amm S, Kösling S. Gradenigo syndrome--still a threatening complication of otitis media. HNO. 2005;53:884-888.
- Yozu A, Suwa K, Mori M, Yamagata T, Mizuguchi M, Momoi MY. Magnetic resonance findings in a case of Gradenigo syndrome: widespread inflammation involving the paranasal sinuses and middle ear. No To Hattatsu. 2004;36:334-338.
- Trimis G, Mostrou G, Lourida A, Prodromou F, Syriopoulou V, Theodoridou M. Petrositis and cerebellar abscess complicating chronic otitis media. J Paediatr Child Health. 2003;39:635-636.
- Finkelstein Y, Marcus N, Mosseri R, Bar-Sever Z, Garty BZ. Streptococcus acidominimus infection in a child causing Gradenigo syndrome. Int J Pediatr Otorhinolaryngol. 2003;67:815-817.
- Penas-Prado M, DÃÂaz-Guzmán J, Jiménez-Huerta I, Juntas-Morales R, Villarejo-Galende A, DÃÂez-Torres I. Gradenigo syndrome as the form of presentation of nasopharyngeal carcinoma. Rev Neurol. 2001;32:638-640.
- Marianowski R, Rocton S, Ait-Amer JL, Morisseau-Durand MP, Manach Y. Conservative management of Gradenigo syndrome in a child. Int J Pediatr Otorhinolaryngol. 2001;57:79-83.
- Minotti AM, Kountakis SE. Management of abducens palsy in patients with petrositis. Ann Otol Rhinol Laryngol. 1999;108:897-902.
- Hananya S, Horowitz Y. Gradenigo syndrome and cavernous sinus thrombosis in fusobacterial acute otitis media. Harefuah. 1997;133:284-286.
- Davé AV, Diaz-Marchan PJ, Lee AG. Clinical and magnetic resonance imaging features of Gradenigo syndrome. Am J Ophthalmol. 1997;124:568-570.
- Morales C, Tachauer A. Gradenigo syndrome in a human immunodeficiency virus-positive patient. Arch Intern Med. 1997;157:2149.
- Murakami T, Tsubaki J, Tahara Y, Nagashima T. Gradenigo's syndrome: CT and MRI findings. Pediatr Radiol. 1996;26:684-685.
- Tutuncuoglu S, Uran N, Kavas I, Ozgur T. Gradenigo syndrome: A case report. Pediatr Radiol. 1993;23:556.
- Hehl K, Tisch M, Lampl L, Draf W, Stussak G, Frey G. Bilateral Gradenigo syndrome--on the value of an integrated therapy concept including hyperbaric oxygenation and chronic destructive inflammation of the skull base. HNO. 1992;40:318-321.
- Gadre AK, Chole RA. The changing face of petrous apicitis-A 40-year experience. Laryngoscope. 2018;128:195-201.
- Kazemi T. Acute otitis media-induced Gradenigo syndrome, a dramatic response to intravenous antibiotic. Iran J Otorhinolaryngol. 2017;29:165-169.
- Jensen PV, Hansen MS, Møller MN, Saunte JP. The forgotten syndrome? Four cases of Gradenigo's syndrome and a review of the literature. Strabismus, 2016;24:21-27.
- Govea-Camacho LH, Pérez-RamÃÂrez R, Cornejo-Suárez A, Fierro-Rizo R, Jiménez-Sala CJ, Rosales-Orozco CS. Diagnosis and treatment of the complications of otitis media in adults. Case series and literature review. Cir Cir. 2016;84:398-404.
- Reddy RK, Reddy RK, Jyung RW, Eloy JA, Liu JK. Gruber, Gradenigo, Dorello, and Vail: Key personalities in the historical evolution and modern-day understanding of Dorello's canal. J Neurosurg. 2016;124:224-233.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.