Hydroxycut‑induced Liver Toxicity
- *Corresponding Author:
- Dr. Dharmesh H Kaswala
Department of Medicine, The University Hospital, New Jersey Medical School-UMDNJ, Newark, NJ 07103, USA.
E-mail: dr.dharmesh@yahoo.com
Abstract
In the recent era, use of various nutritional supplements is highly encouraged amongst the people of United States. Weight loss supplements are major part of the nutritional supplements and their usage is unregulated in the US. Obesity is a major health concern in the US and Americans spend around $30 billion a year for weight loss supplements. At times, these supplements can be responsible for documented or undocumented adverse drug effects. The health consequences related to these supplements are often overlooked by the general public, even though FDA issues advisories regarding them. One common supplement used for weight loss was Hydroxycut (Iovate Health Sciences Research, Oakville, Ontario, Canada). Hydroxycut was recalled from the market after a FDA warning in May 2009 because of 23 reports of serious health problems ranging from jaundice and elevated liver enzymes to liver damage. 1 This case report adds evidence for Hydroxycut ‑ induced hepatotoxicity. A 27 year old man with right upper quadrant pain and jaundice was found to have elevated liver enzymes and was taking Hydroxycut along with other supplements. Liver biopsy showed drug induced hepatotoxicity. Discontinuation of Hydroxycut dramatically improved liver functions and related symptoms.
http://www.oajournal.org/
http://www.journalsres.org/
http://www.journalsres.com/
http://www.journalsoa.org/
http://www.journalsoa.com/
http://www.journalsci.org/
http://www.journalres.org/
http://www.journalres.com/
http://www.journaloa.org/
http://www.journalinsights.org/
http://www.jpeerreview.org/
http://www.imedresearch.com/
http://www.imedpubjournals.com/
http://www.imedpubjournal.org/
http://www.imedjournals.org/
http://www.peerreviewedjournal.org/
http://www.peerjournals.org/
http://www.peerjournals.com/
http://www.sciencesinsight.org/
http://www.scholarresearch.com/
http://www.scholarres.org/
http://www.nutritionres.com/
http://www.gastroinsights.org/
http://www.pathologyinsights.org/
http://www.echemistry.org/
http://www.echemcentral.com/
http://www.chemistryres.com/
http://www.biochemresearch.org/
http://www.biochemjournals.com/
http://www.ebusinessjournals.org/
http://www.businessjournals.org/
http://www.peerjournal.org/
http://www.oajournalres.com/
http://www.alliedres.org/
http://www.alliedjournals.org/
http://www.alliedjournal.org/
http://www.scientificres.org/
http://www.scientificres.com/
,
https://www.mongoliannutrition.com/
https://www.nsbmb.com/
https://www.arabspp.org/
https://www.arabianmultidisciplinary.com/
https://www.italystemcell.com/
https://www.traditional-medicine.org/
https://www.episportsmedicine.org/
https://www.worldmedicalassociation.org/
https://www.silaeitaly.com/
https://www.ceramicsmedicine.org/
https://www.isaddictionmedicine.org/
https://www.europeanbionetwork.com/
https://www.aarsecp.com/
https://www.edycseg.org/
https://www.europeanneurology.org/
https://www.clinicaldermepi.com/
https://www.cardiac-society.com/
https://www.psychologicalassociation.org/
https://www.indian-psychology.com/
https://www.mongoliancardiology.org/
https://www.pediatricssociety.com/
https://www.cocrt.org/
https://www.european-aesthetic.com/
https://www.sohnsb.org/
Keywords
Hydroxycut, Liver toxicity, Nutritional supplements
Introduction
In US, nutritional supplements are largely unregulated; frequently, health advisories are issued regarding these products, but these may not be heeded by general public that may not be aware of the consequences of taking an unregulated substance promoted for its health benefits. This case demonstrates the possible harm that patients may experience with the use of weight loss herbal supplement, Hydroxycut.
Case Report
27-year-old man with no significant past medical history presented as a referral from a community hospital for evaluation of hepatitis, initially diagnosed as choledocholithiasis. The patient complained of fever, chills, and abdominal pain (achy in the Right Upper Quadrant and epigastrium) for the past 2 weeks, nausea, and vomiting of clear liquids without blood for a 1 week, and jaundice for 2 days. Records from referring facility indicated that patient had MRCP-Magnetic Resonant Cholangio Pancreatography that demonstrated cholelithiasis, but it did not show any signs of cholecystitis. An abdominal ultrasound revealed sludge and gallstones in the gallbladder.
Further questioning revealed that patient had been studying to be a physical trainer at a local college. He exercised vigorously at least 3 times/week. Patient stated that he was “prescribed” a set of supplements (Hydroxycut, Black powder, mitotropin, xenadrine, arson, and L-glutamine powder 23) by his local nutrition center.
After admission in the hospital, hepatology consult was called. When patient was admitted, liver enzymes panel showed: AST-Aspartate Aminotransferase: 3800 U/L, ALT-Alanine aminotransferase: 5100 U/L, total bilirubin: 14.7 mg/dl, direct bilirubin: 10.0 mg/dl and alkaline phosphatase: 99 mg/dl, all correlated with the degree of jaundice seen. Additional tests included a urine toxicology screen, immunological studies for liver (anti-nuclear antibodies, anti-smooth muscle antibodies, and anti A liver/kidney microsomal antibodies), serum acetaminophen level, serum ceruloplasmin level, and genetic tests for hemochromatosis; all of them were negative. Liver biopsy showed signs of acute liver failure.
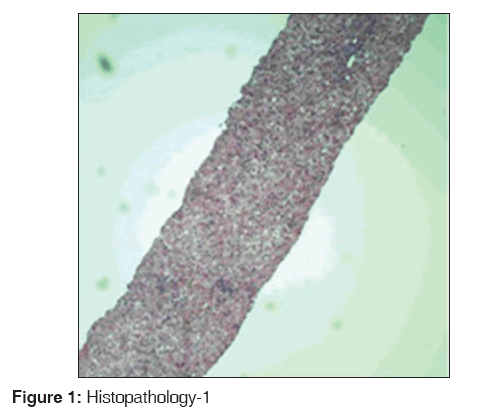
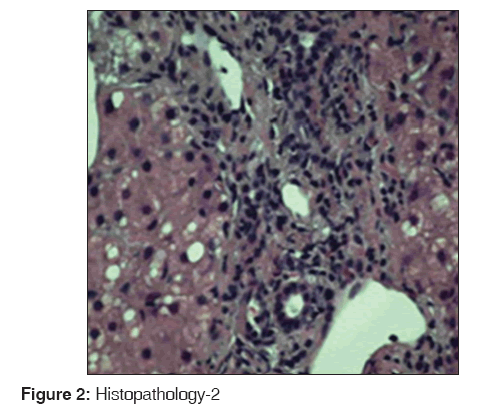
Hematoxylin and eosin stain of the liver biopsy specimen is shown in Figures 1 and 2. Figure 1 shows extensive patchy areas of necrosis as in acute fulminant hepatitis with sparing of biliary tract. Figure 2 shows lymphocytic infiltration of the portal tracts and lobular parenchyma.
Patient was asymptomatic during the hospital stay and was planned to discharge with trending down of liver enzymes by discontinuation of Hydroxycut and conservative management. The patient was advised to follow-up with Hepatology. On follow-up visit after 2 weeks of discharge, Labs: AST: 669 U/L, ALT: 1965 U/L, Total Bilirubin: 4.5 mg/dl, Alkaline phosphatase: 128 mg/dl, PT: 1.1.
Discussion
Use of alternative and herbal medication as a dietary supplement is becoming increasing popular in recent years owing to toxicity associated with most prescription medication and the presumed notion of safety with over the counter and so called natural weight loss therapies in the setting of our epidemic of obesity in the United States. Drugs are a significant cause of liver injury. More than 900 drugs, toxins, and herbs have been reported to cause liver injury, and drugs account for 20-40 % of all instances of fulminant hepatic failure. Approximately, 75% of the idiosyncratic drug reactions result in liver failure or death. Drug-induced hepatic injury is the most common reason for withdrawal of approved drugs. The manifestations of drug-induced hepatotoxicity are highly variable, ranging from asymptomatic elevation of liver enzymes to fulminant hepatic failure.
Hydroxycut had recently issued a public recall and a health advisory connecting the use of Hydroxycut and severe fulminant Hepatitis was issued in July, 2009.[1] There are several previous case reports suggesting hepatotoxicity associated with Hydroxycut.[2-7] This case illustrates the importance of obtaining a full history of both prescribed medications, non-prescription medications and additional supplements that a patient may be taking and the potential harm that unregulated nutritional supplements may pose to patients. The fulminant hepatitis that this patient experienced is not rare; however, without a complete history inclusive of patient’s nutritional supplements, the diagnosis may have continued to be elusive.
Following are the components of the Hydroxycut product, which is still available online.[8] Serving size: 2 Caplets
• Calcium: 156 mg
• Chromium: 133 mcg
• Potassium: 218 mg
• Hydroxagen plus: 1.32 g*
• Garcinia Cambogia Extract (Fruit and Rind) standardized for 60%- hydroxycitric acid (HCA)
• Gymnema Sylvestre Extract (Leaf) standardized for 25% gymnemic acids
• Soy phospholipids
• Rhodiola Rosea Extract (Root) standardized for 5% rosavins
• Withania somnifera Extract Root standardized for 1.5% withanolides
• Hydroxy tea: 414 mg*
• Green tea Extract as Camellia Sinensis leaf: 91 mg*
• Caffeine anhydrous: 200 mg*
• White Tea Extract as Camellia Sinensis Leaf
• Oolong Tea Extract as Camellia Sinensis Leaf.
From all of these components chromium, Garcinia Cambogia Extract and Camellia Sinensis of tea extracts are found to have an association with the hepatotoxicity related to Hydroxycut. There have been several cases reported in the past linking these components to cause hepatotoxicity. In all of those cases, when the product containing the component is withdrawn, improvement in liver enzymes was seen. Garcinia Cambogia has HCA as the main component, which is an inhibitor of the ATP-Adenosine Triphosphate citrate lyase so it blocks de novo synthesis of fatty acids and helps in weight reduction. However, two recent reports show its association with hepatotoxicity. One report shows the synergistic effect of HCA with montelukast in causing liver damage.[9] Other one states that the increase in liver enzymes is seen with HCA containing product use.[10] Chromium is used in Hydroxycut because of its effects such as increasing basal metabolic rate and decreasing total body fat. However, there have been some cases reporting its dangerous side-effects such as acute hepatitis, thrombocytopenia, and renal failure.[11-13] Green tea, which is widely used because of its ant-oxidant, weight-reducing and anti-cancer properties, contains Camellia Sinensis. There have been several cases that show an association of Camellia Sinensis with liver toxicity ranging from acute hepatitis to liver failure.[14-17]
Although, it is difficult to prove a causal relationship between Hydroxycut and hepatotoxicity, in our case liver damage was seen after starting Hydroxycut use and improvement in liver enzymes was seen after stopping it. In addition, absence of any other etiologies proven by extensive lab test suggests that Hydroxycut was most probable cause of the hepatotoxicity. Our patient did not have any existing liver disease or any comorbid disease. While causation is difficult to prove in any drug induced injury, the temporal relationship after Hydroxycut exposure and gradual improvement after withdrawing the involved medication plus absence of any other etiologies despite comprehensive testing would point to Hydroxycut being the most likely possible cause for hepatotoxicity. Earlier reports from Dara et al., with case series of 2 patients have shown a similar temporal relationship, also present histological data for possible association with drug toxicity.[2] A report of 2 cases from Stevens et al., showed one with hepatocellular and the other with cholestatic liver toxicity secondary to Hydroxycut use.[3]
In May, 2009, FDA-Food and Drug Administration issued a warning regarding Hydroxycut related hepatotoxicity because of 23 case reports suggesting evidence of such consequences and so the product was removed from the market by the manufacturer.
Conclusion
Our case report emphasizes on the importance of post-marketing surveillance of nutritional supplements and over the counter medications. Though Hydroxycut is off the market, there are several other supplements in the market, and we do not know, which health consequences they carry. Physicians should always be alert about non-prescription drug usage by patients and should always try to inquire about that and report any unusual side-effects to designated authorities. More stringent policy and regulations are required for stopping such harmful products from coming to the market.
Source of Support
Nil.
Conflict of Interest
None declared.
References
- Commissioner of the Consumer Updates-Warning on Hydroxycut Products. Available from: http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm152152.
- Dara L, Hewett J, Lim JK. Hydroxycut hepatotoxicity: A case series and review of liver toxicity from herbal weight loss supplements. World J Gastroenterol 2008;14:6999-7004.
- Stevens T, Qadri A, Zein NN. Two patients with acute liver injury associated with use of the herbal weight-loss supplement hydroxycut. Ann Intern Med 2005;142:477-8.
- Jones FJ, Andrews AH. Acute liver injury associated with the herbal supplement hydroxycut in a soldier deployed to Iraq. Am J Gastroenterol 2007;102:2357-8.
- Shim M, Saab S. Severe hepatotoxicity due to Hydroxycut: A case report. Dig Dis Sci 2009;54:406-8.
- Lobb A. Hepatoxicity associated with weight-loss supplements: A case for better post-marketing surveillance. World J Gastroenterol 2009;15:1786-7.
- Sharma T, Wong L, Tsai N, Wong RD. Hydroxycut(®) (herbal weight loss supplement) induced hepatotoxicity: A case report and review of literature. Hawaii Med J 2010;69:188-90.
- Online store selling Hydroxycut, 2012. Available from: http:// www.Hydroxycutonline.com. [Accessed on 2012 Jun 26].
- Actis GC, Bugianesi E, Ottobrelli A, Rizzetto M. Fatal liver failure following food supplements during chronic treatment with montelukast. Dig Liver Dis 2007;39:953-5.
- Pittler MH, Ernst E. Dietary supplements for body-weight reduction: A systematic review. Am J Clin Nutr 2004;79:529-36.
- Fristedt B, Lindqvist B, Schuetz A, Ovrum P. Survival in a case of acute oral chromic acid poisoning with acute renal failure treated by haemodialysis. Acta Med Scand 1965;177:153-9.
- van Heerden PV, Jenkins IR, Woods WP, Rossi E, Cameron PD. Death by tanning – A case of fatal basic chromium sulphate poisoning. Intensive Care Med 1994;20:145-7.
- Cerulli J, Grabe DW, Gauthier I, Malone M, McGoldrick MD. Chromium picolinate toxicity. Ann Pharmacother 1998;32:428-31.
- Pedrós C, Cereza G, García N, Laporte JR. Liver toxicity of Camellia sinensis dried etanolic extract. Med Clin (Barc) 2003;121:598-9.
- García-Morán S, Sáez-Royuela F, Gento E, López Morante A, Arias L. Acute hepatitis associated with Camellia thea and Orthosiphon stamineus ingestion. Gastroenterol Hepatol 2004;27:559-60.
- Bonkovsky HL. Hepatotoxicity associated with supplements containing Chinese green tea (Camellia sinensis). Ann Intern Med 2006;144:68-71.
- Richard L Cleland, Walter C Gross, Laura D Koss, Matthew Daynard and Karen M Muoio. Federal Trade Commission Staff Report Sept 2002: Weight-loss advertising: An analysis of current trends. Available from: http://www.ftc.gov/bcp/ reports/weightloss.pdf. [Last access on 2012 July].




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.