Metabolic Abnormalities, Cognitive Functions, and Bipolar Disorder: A Meta-Analysis
2 Medical Intern, Jouf University, Sakaka, Saudi Arabia
3 Medical Student, Alqassim University, Onizah, Saudi Arabia
4 Medical Intern, Umm Al- Qura University, Qunfudah City, Saudi Arabia, Email: mohammed16@gmail.com
5 General Practitioner, Ajyad Emergency Hospital, Makkah City, Saudi Arabia
Citation: Almasabi AA, et al. Metabolic Abnormalities, Cognitive Functions, and Bipolar Disorder: A Meta-Analysis. Ann Med Health Sci Res. 2020;10:1105-1110.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Background: In the general population, obesity and MetS and its components are important risk factors for incident cognitive impairment and dementia. Similarly, obesity and components of MetS negatively influence the integrity of brain structure. However, the relationship between obesity/MetS and neurocognition/ neuroimaging has received limited attention in patients with BD. Aim: This work aims to determine the prevalence of metabolic syndrome (MetS) components, in bipolar disorder (BD) patients. Materials and Methods: A systematic search was performed over different medical databases to identify Psychiatry studies, which studied the outcome of the BD group versus the Control group of bipolar disorder (BD) patients. Using the meta-analysis process, either with fixed or random-effects models, we conducted a meta-analysis on the prevalence of “≥1 MetS components” as a primary outcome, and on obesity, DM, HTN, Hyperlipidemia prevalence, as secondary outcomes. Results: Seven studies were identified involving 2773 patients, with 566 patients in the BD group, and 2207 patients in the Control group. The meta-analysis process revealed a highly significant increase in MetS prevalence in the BD group compared to the Control group (p<0.001), along with obesity, HTN, and hyperlipidemia (P<0.05) respectively. Conclusion: To conclude, the present study suggests that over a mean period of about 5 years, there is an increase in the prevalence of MetS among patients with BD. Accordingly, there is a need to periodically monitor the metabolic parameter of patients with BD and interventions must be started for the same at the earliest.
Keywords
Metabolic abnormalities; Bipolar disorder
Introduction
Bipolar disease (BD) is a chronic mental illness characterized by recurrent episodes of depression, mania, and hypomania. Global, the combined lifetime incidence of BD I and II are estimated among 1 and 3%. People with BD are more prone to seek care while they’re depressed than when experiencing mania or hypomania. The condition might consequently mistakenly be diagnosed as major depression and BD patients are frequently treated with antidepressants in primary care. That is trouble because monotherapy with antidepressants increase the hazard of the switch to mania in BD and may sometimes get worse symptoms. [1]
Bipolar disorder (BPD) is a complicated, recurrent, severe mental illness that affects over 350 million people globally. Individuals with BPD will often require long-term drug treatment with the aim of preventing relapse or recurrence. Much of the evidence for maintenance remedy comes from relatively shorttime period randomized controlled trials, which can be then extrapolated to longer-time period use. But, this fails to take into account the potential longer period adverse effects of the recommended medication. [2]
In recent years, metabolic syndrome has become an important issue in both psychiatry and public health. It is a complicated global epidemic that imposes substantial socio-economic costs on different societies. The disease is step by step becoming more prevalent amongst children and young adults, and accomplishing regarding levels around the world. Metabolic syndrome is of limited utility in diagnosis or treatment. Rather than a medical analysis, the syndrome is a premorbid condition from which recognized diabetic and cardiovascular patients must be excluded. [3]
In the general population, obesity and MetS and its components are important risk factors for incident cognitive impairment and dementia. Similarly, obesity and components of MetS negatively influence the integrity of brain structure. However, the relationship between obesity/MetS and neurocognition/ neuroimaging has received limited attention in patients with BD. [4]
This work aims to determine the prevalence of metabolic syndrome (MetS) components, in bipolar disorder (BD) patients.
Literature Review
Our review came following the (PRISMA) statement guidelines. [5]
Study eligibility
The included studies should be in English, a journal published article, and a human study describing bipolar disorder (BD) patients. The excluded studies were non-English or animal studies or describing other types of psychiatry patients (e.g. schizophrenic patients).
Study identification
Basic searching was done over the PubMed, Cochrane library, and Google scholar using the following keywords: Metabolic Abnormalities, Bipolar Disorder.
Data extraction and synthesis
RCTs, clinical trials, and comparative studies, which studied the outcome of the BD group versus the Control group of bipolar disorder (BD) patients, will be reviewed.
Outcome measures included the prevalence of “≥1 MetS components” as a primary outcome, and on obesity, DM, HTN, Hyperlipidemia prevalences, as secondary outcomes.
Study selection
We found 350 records, 290 excluded based on title and abstract review; 60 articles are searched for eligibility by full-text review; 24 articles cannot be accessed; 15 studies were reviews and case reports; 14 were not describing functional outcomes; leaving 7 studies that met all inclusion criteria.
Statistical methodology
The pooling of data, Proportions (%), Odds Ratios (ORs), Standard Mean Differences (SMDs), with 95% confidence intervals (CI) were done, using MedCalc ver. 18.11.3 (MedCalc, Belgium). According to heterogeneity across trials using the I2- statistics; a fixed-effects model or random-effects model were used in the meta-analysis process.
Results
The included studies were published between 2014 and 2020 [Table 1]. [6-12]
Table 1: Patients and study characteristics.
| N | Author | Number of patients | Age (average years) | Disease duration (average years) | ||
|---|---|---|---|---|---|---|
| Total | BD group | Control group | ||||
| 1 | Hajek et al. [6] | 48 | 11 | 50.6 | 27.7 | |
| 2 | Naiberg et al. [7] | 34 | 35 | 17.2 | -- | |
| 3 | Bond et al. [8] | 57 | 31 | 22.7 | -- | |
| 4 | Islam et al. [9] | 40 | 48 | 17 | -- | |
| 5 | Coello et al. [10] | 206 | 109 | 29 | 10 | |
| 6 | Moreira et al. [11] | 1023 | 41 | 982 | 23.5 | -- |
| 7 | Mohite et al. [12] | 140 | 991 | 15.1 | -- | |
#Studies arranged via publication year.
Regarding patients’ characteristics, the total number of patients in all the included studies was 2773 patients, with 566 patients in the BD group, and 2207 patients in the Control group, while their average disease duration was (18.8 years) [Table 1]. The mean age of all patients was (25 years), [Table 1]. [6-12]
A meta-analysis study was done on 7 studies that described and compared the 2 different groups of patients; with an overall number of patients (N=2773) [Table 2].
Table 2: Summary of outcome measures in all studies.
| N | Author | Primary outcome | Secondary outcomes | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| MetS prevalence | Obesity | DM | HTN | Hyperlipidemia | |||||||
| BD group | Control group | BD group | Control group | BD group | Control group | BD group | Control group | BD group | Control group | ||
| 1 | Hajek et al. [6] | -- | -- | -- | -- | -- | -- | 27 | 0 | 15 | 0 |
| 2 | Naiberg et al. [7] | 22 | 8 | 6 | 1 | 2 | 1 | 7 | 5 | 6 | 2 |
| 3 | Bond et al. [8] | -- | -- | 5 | 1 | -- | -- | -- | -- | -- | -- |
| 4 | Islam et al. [9] | -- | -- | 25 | 21 | -- | -- | -- | -- | -- | -- |
| 5 | Coello et al. [10] | 31 | 6 | -- | -- | -- | -- | -- | -- | -- | -- |
| 6 | Moreira et al. [11] | 19 | 230 | -- | -- | -- | -- | -- | -- | -- | -- |
| 7 | Mohite et al. [12] | 20 | 66 | 35 | 117 | 13 | 75 | 19 | 79 | 52 | 230 |
Each outcome was measured by:
Odds Ratio (OR)
• For the prevalence of “≥1 MetS components”
• Obesity prevalence.
• DM prevalence.
• HTN prevalence.
• Hyperlipidemia prevalence.
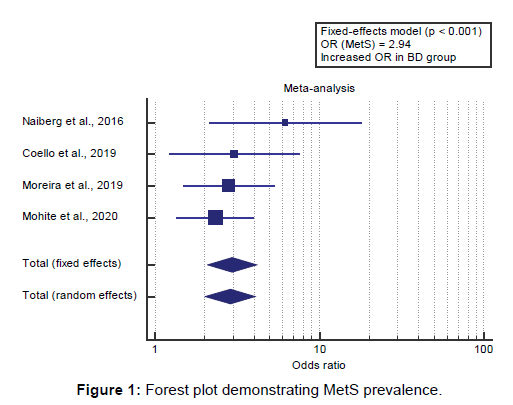
Concerning the primary outcome measure, we found 4 studies reported MetS prevalence.
I2 (inconsistency) was 0% with a non-significant Q test for heterogeneity (p>0.05), so fixed-effects model was carried out; with overall OR=2.94 (95% CI=2.075 to 4.174).
Using the fixed-effects model, the meta-analysis process revealed a highly significant increase in MetS prevalence in the BD group compared to the Control group (p<0.001) [Figure 1].
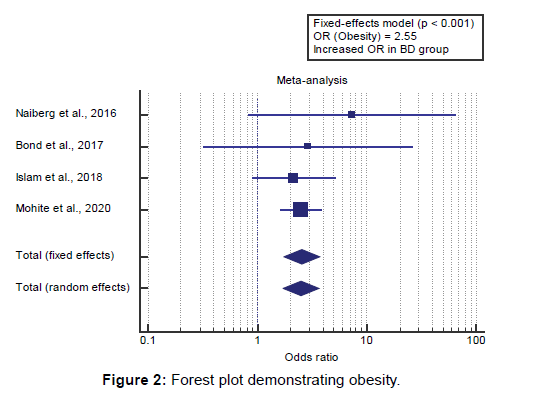
Concerning the secondary outcome measures, we found 4 studies reported obesity prevalence.
I2 (inconsistency) was 0% with a non-significant Q test for heterogeneity (p>0.05), so fixed-effects model was carried out; with overall OR=2.55 (95% CI=1.758 to 3.7). Using the fixed-effects model, the meta-analysis process revealed a highly significant increase in obesity prevalence in the BD group compared to the Control group (p<0.001) [Figure 2].
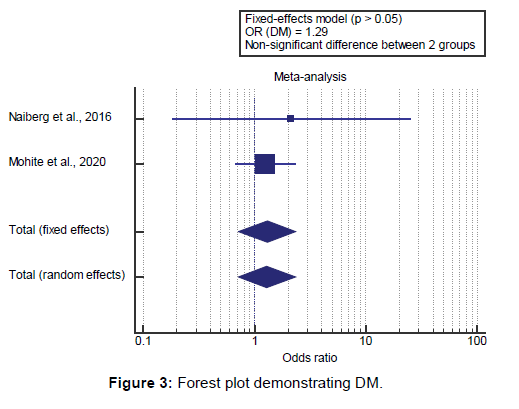
We found 2 studies reported DM prevalence. I2 (inconsistency) was 0% with a non-significant Q test for heterogeneity (p>0.05), so fixed-effects model was carried out; with overall OR=1.29 (95% CI=0.715 to 2.348). Using the fixed-effects model, the meta-analysis process revealed a non-significant difference in DM prevalence in the BD group compared to the Control group (p>0.05) [Figure 3].
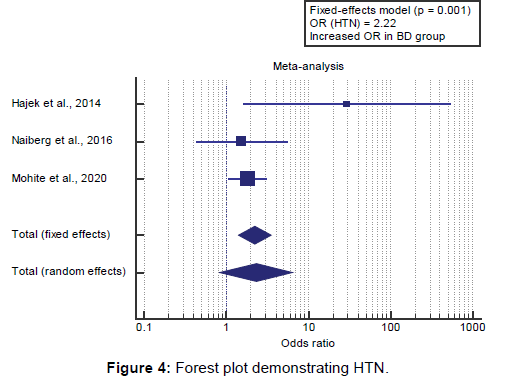
We found 3 studies reported HTN prevalence. I2 (inconsistency) was 49% with a non-significant Q test for heterogeneity (p>0.05), so fixed-effects model was carried out; with overall OR=2.22 (95% CI=1.404 to 3.525).
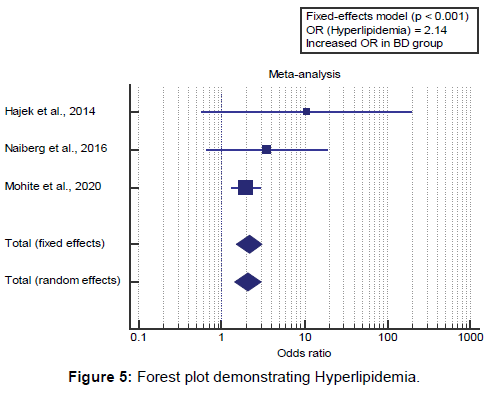
Using the fixed-effects model, the meta-analysis process revealed a highly significant increase in HTN prevalence in the BD group compared to the Control group (p=0.001) [Figure 4]. We found 3 studies reported the prevalence of hyperlipidemia.
I2 (inconsistency) was 49% with a non-significant Q test for heterogeneity (p>0.05), so fixed-effects model was carried out; with overall OR=2.14 (95% CI=1.505 to 3.068).
Using the fixed-effects model, the meta-analysis process revealed a highly significant increase in the prevalence of hyperlipidemia in the BD group compared to the Control group (p<0.001) [Figure 5].
Discussion
This work aims to determine the prevalence of metabolic syndrome (MetS) components, in bipolar disorder (BD) patients.
The included studies were published between 2014 and 2020. Regarding patients’ characteristics, the total number of patients in all the included studies was 2773 patients, with 566 patients in the BD group, and 2207 patients in the Control group, while their average disease duration was (18.8 years). The mean age of all patients was (25 years).
A meta-analysis study was done on 7 studies that described and compared the 2 different groups of patients; with an overall number of patients (N=2773).
Concerning the primary outcome measure, we found 4 studies reported MetS prevalence. Using the fixed-effects model, the meta-analysis process revealed a highly significant increase in MetS prevalence in BD group compared to Control group (p<0.001), which came in agreement with Grover et al., [13] Rosenbaum et al., [14] Vancampfort et al. [15] and SayuriYamagata et al. [16]
Grover et al. reported that, at the baseline, 54.2% (N=32) of patients fulfilled the criteria for MetS, and after the mean follow-up period of 62.1 (SD-55.3) months, the prevalence of MetS extended to 66.1% (N=39). [13]
Rosenbaum et al. reported that, overall, 9 research met the inclusion standards which include 9,673 individuals in midlife with PTSD and 6852 general population controls. The pooled MetS incidence was 38.7% (Q=52.1, p<0.001; N=9; n=9,673; age range=44–61 years). [14]
Vancampfort et al. reported that the primary goal of this systematic evaluation and meta-analysis become to evaluate the prevalence of MetS and its components in people with schizophrenia and related psychotic disorders, bipolar disorder, and major depressive sickness, comparing subjects with different issues and thinking of demographic variables and psychotropic medication use. The secondary aim was to compare the MetS incidence in individuals with any of the selected disorders as opposed to matched general population controls. The pooled MetS incidence in humans with severe mental illness was 32.6%. [15]
SayuriYamagata et al. reported that metabolic syndrome has been described by a cluster of metabolic risk factors for cardiovascular disease and insulin resistance, together with hypertension, hyperglycemia, dyslipidemia, and obesity. A persistent low-grade level of inflammation is common amongst numerous medical conditions, including metabolic syndrome (MetS). The higher incidence of MetS in BD has been welldocumented. [16]
Concerning the secondary outcome measures, we found 4 studies reported obesity prevalence. Using the fixed-effects model, the meta-analysis process revealed a highly significant increase in obesity prevalence in BD group compared to Control group (p<0.001), which came in agreement with Grover et al., [13] Agaba et al., [17] Rosenbaum et al., [14] Vancampfort et al., [15] Correll et al. [18] and Bora, McIntyre, and Ozerdem. [4]
Grover et al. reported that, in terms of absolute values, compared to the baseline, there was a significant increase in the weight (kg), waist circumference (cm), diastolic blood pressure (mm Hg) and mean score of the consensus criteria for metabolic. [13]
Agaba et al. reported that, of the 304 participants, 44.41% were male and 55.59% female with a mean age of 38.56 ±13.66 years. Almost half (46.38%) of the participants were either overweight or obese, 33.22% had abdominal obesity. [17]
Rosenbaum et al. reported that, overall, the proportion of people with PTSD with abdominal obesity by the ATP definitions was 49.3% (n=194). [14]
Vancampfort et al. reported that Sixty-five research suggested obesity frequency defined as waist circumference >102 cm in males and >88 cm in ladies (ATP-III or ATP-III-A), even as 14 research mentioned the obesity frequency following the ethnicity-specific IDF criteria. Overall, the proportion of patients with abdominal obesity become 50.3% by using the ATP definitions (Q51.6, p<0.001) and 63.2% according to IDF ( Q5480.9, p< 0.001). [15]
Correll et al. reported that people with SMI are, in comparison to the general population, at increased risk of being obese and overweight. The chance of being obese is extended 2.8-4.4 folds in patients with schizophrenia and 1.2-1.7 fold in those with major depression or bipolar disorder. [18]
Bora, McIntyre, and Ozerdem reported that Twenty-three studies were included in the current systematic review. Overweight/ obese patients were significantly associated with impaired neurocognition compared to normal-weight individuals with BD (d=0.37). [4]
We found 2 studies reported DM prevalence. Using the fixedeffects model, the meta-analysis process revealed a nonsignificant difference in DM prevalence in the BD group compared to the Control group (p>0.05), which came in agreement with Kittel-Schneider et al. [19] and Bora, McIntyre, and Ozerdem. [4]
Kittel-Schneider et al. reported that patients suffering from BD had a T2D incidence of 7%, which was no longer substantially different from the control group (6%). Fasting glucose and impaired glucose tolerance were, contrary to our hypothesis, more often pathological in controls than in BD patients. Nondiabetic and diabetic bipolar patients significantly differed in age, BMI, number of depressive episodes, and disorder period. [19]
Bora, McIntyre, and Ozerdem reported that, the relationship among dyslipidemia (or glucose degrees) and cognitive impairment in BD. there has been no evidence of a significant relationship between glucose levels (or DM) or cognitive impairment in two of those research. [4]
Our result came in disagreement with Rosenbaum et al. [14] Rosenbaum et al. reported that it was possible to pool data from 3 studies to establish that the prevalence of hyperglycemia was 36.1% (N=3, n=194). [14]
We found 3 studies reported HTN prevalence. Using the fixedeffects model, the meta-analysis process revealed a highly significant increase in HTN prevalence in BD group compared to Control group (p=0.001), which came in agreement with Grover et al., Vancampfort et al. [15] and Bora, McIntyre, and Ozerdem. [4]
Grover et al. reported that, while absolutely the values have been evaluated further, it changed into seen that majority of the patients experienced weight gain, had a significant increase in weight (i.e.,>five% increase or lower in weight), increase inside the waist circumference and rise in blood pressure. [13]
Vancampfort et al. reported that Hypertension (ATP-III, ATPIII- A, and IDF) was present in 39.3% (N588, n527,441; Q52.7, p<0.001). [15]
Bora, McIntyre, and Ozerdem reported that the relationship between hypertension (or blood pressure) and cognitive impairment in BD reported a significant relationship between more severe cognitive deficits and categorical or dimensional assessment of blood pressure. [4]
We found 3 studies reported the prevalence of hyperlipidemia. Using the fixed-effects model, the meta-analysis process revealed a highly significant increase in the prevalence of hyperlipidemia in BD group compared to Control group (p<0.001), which came in agreement with Agaba et al., [17] Grover et al., [13] Rosenbaum et al., [14] Vancampfort et al., [15] and Fjukstad et al. [20]
Agaba et al. reported that the proportion of participants with abnormal lipid profiles were as follows; 34.11% had low highdensity lipoproteins, 37.42% had hypertriglyceridemia and 34.77% had hypercholesterolemia. [17]
Grover et al. reported that, among the various abnormalities, raised waist circumference was the most common abnormality at both the evaluation. At baseline, the second one most common abnormality was abnormal triglyceride stages, and this was observed by way of low HDL levels. [13]
Rosenbaum et al. reported that, Hypertriglyceridemia was present in 45.9% (N=3, n=194; Q=55.1, p<0.001); low highdensity- lipoprotein (HDL)-cholesterol was present in 46.4% (N=3, n=194; Q=16.2, p<0.001). [14]
Vancampfort et al. reported that Hypertriglyceridemia was present in 36.2% (N587, n526,577; Q52.7, p<0.001). Low HDL cholesterol was present in 39.1% (N586, n526,193; Q51.9, p<0.001). [15]
Fjukstad et al. reported that, after adjusting for potential confounders, an SSRI serum concentration in the middle of the reference interval was associated with an increase of the total cholesterol level by 14.56 mg/dL (P=0.002), the LDL cholesterol level by 8.50 mg/dL (P=0.044), the triglyceride level by 46.49 mg/dL (P<0.001) and the occurrence of the metabolic syndrome by a factor of 2.10 (P=0.008). There were also significant associations between the SSRI dose and total cholesterol and LDL cholesterol levels. [20]
Conclusion
To conclude, the present study suggests that over a mean period of about 5 years, there is an increase in the prevalence of MetS among patients with BD. Accordingly, there is a need to periodically monitor the metabolic parameter of patients with BD and interventions must be started for the same at the earliest.
Competing Interests
The authors declare that they have no competing interests. All the listed authors contributed significantly to the conception and design of study, acquisition, analysis, and interpretation of data and drafting of the manuscript, to justify authorship.
REFERENCES
- Yoshimi N, Futamura T, Kakumoto K, Salehi AM, Sellgren CM, Holmén-Larsson J, et al. Blood metabolomics analysis identifies abnormalities in the citric acid cycle, urea cycle, and amino acid metabolism in bipolar disorder. BBA Clinical 2016;5:151-158.
- Hayes JF, Marston L, Walters K, Geddes JR, King M, Osborn DP. Adverse renal, endocrine, hepatic, and metabolic events during maintenance mood stabilizer treatment for bipolar disorder: a population-based cohort study. PLoS Medicine 2016;13:e1002058.
- Nayerifard R, Zahiroddin A, Namjoo M, Rajezi S. Comparison of metabolic syndrome prevalence in patients with schizophrenia and bipolar I disorder. Diabetes Metab Syndr. 2017;11:S411-S416.
- Bora E, McIntyre RS, Ozerdem A. Neurococognitive and neuroimaging correlates of obesity and components of metabolic syndrome in bipolar disorder: a systematic review. Psychol Med. 2019;49:738-749.
- Liberati A, Altman D, Tetzlaff J, Mulrow C, Gøtzsche P, Ioannidis J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions. BMJ 2009;339.
- Hajek T, Calkin C, Blagdon R, Slaney C, Uher R, Alda M. Insulin resistance, diabetes mellitus, and brain structure in bipolar disorders. Neuropsychopharmacol Rep. 2014;39:2910-2918.
- Naiberg MR, Newton DF, Collins JE, Dickstein DP, Bowie CR, Goldstein BI. Elevated triglycerides are associated with decreased executive function among adolescents with bipolar disorder. Acta Psychiatrica Scandinavica 2016;134:241-248.
- Bond DJ, Silveira LE, MacMillan EL, Torres IJ, Lang DJ, Su W, et al. Diagnosis and body mass index effects on hippocampal volumes and neurochemistry in bipolar disorder. Transl Psychiatry. 2017;7:e1071-e1071.
- Islam AH, Metcalfe AW, MacIntosh BJ, Korczak DJ, Goldstein BI. Greater body mass index is associated with reduced frontal cortical volumes among adolescents with bipolar disorder. J Psychiatry Neurosci. 2018;43:120.
- Coello K, Vinberg M, Knop FK, Pedersen BK, McIntyre RS, Kessing LV, et al. Metabolic profile in patients with newly diagnosed bipolar disorder and their unaffected first-degree relatives. Int J Bipolar Disord. 2019;7:8.
- Moreira FP, Jansen K, Cardoso T de A, Mondin TC, Magalhaes PV, Kapczinski F, et al. Metabolic syndrome and psychiatric disorders: a population-based study. Braz. J. Psychiatry. 2019;41:38-43.
- Mohite S, Wu H, Sharma S, Lavagnino L, Zeni CP, Currie TT, et al. Higher Prevalence of Metabolic Syndrome in Child-adolescent Patients with Bipolar Disorder. Clin Psychopharmacol Neurosci. 2020;18:279.
- Grover S, Mehra A, Chakravarty R, Jagota G, Sahoo S. Change in prevalence of metabolic syndrome in patients with bipolar disorder. Asian J Psychiatr. 2020;47:101876.
- Rosenbaum S, Stubbs B, Ward PB, Steel Z, Lederman O, Vancampfort D. The prevalence and risk of metabolic syndrome and its components among people with posttraumatic stress disorder: a systematic review and meta-analysis. Metabolism 2015;64:926-933.
- Vancampfort D, Sienaert P, Wyckaert S, De Hert M, Stubbs B, Probst M. Sitting time, physical fitness impairments and metabolic abnormalities in people with bipolar disorder: an exploratory study. Psychiatry Res. 2016;242:7-12.
- SayuriYamagata A, Brietzke E, Rosenblat JD, Kakar R, McIntyre RS. Medical comorbidity in bipolar disorder: the link with metabolic-inflammatory systems. J Affect Disord. 2017;211:99-106.
- Agaba DC, Migisha R, Katamba G, Ashaba S. Cardio-metabolic abnormalities among patients with severe mental illness at a Regional Referral Hospital in southwestern Uganda. Plos One 2020;15:e0235956.
- Correll CU, Detraux J, De Lepeleire J, De Hert M. Effects of antipsychotics, antidepressants and mood stabilizers on risk for physical diseases in people with schizophrenia, depression and bipolar disorder. World Psychiatry 2015;14:119-136.
- Kittel-Schneider S, Bury D, Leopold K, Haack S, Bauer M, Pfeiffer S, et al. Prevalence of Prediabetes and Diabetes Mellitus Type II in Bipolar Disorder. Front Psychiatry. 2020;11.
- Fjukstad KK, Engum A, Lydersen S, Dieset I, Steen NE, Andreassen OA, et al. Metabolic abnormalities related to treatment with selective serotonin reuptake inhibitors in patients with schizophrenia or bipolar disorder. J Clin Psychopharmacol. 2016;36:615.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.