Pediatric Precursor B-cell Lymphoblastic Lymphoma Presenting with Extensive Skeletal Lesions
- *Corresponding Author:
- Dr. Anupama Borker
Consultant Pediatric Hematologist and Oncologist, Bombay Hospital and Medical Research Centre, Mumbai, India.
E-mail: dranupamasb@gmail.com
Abstract
Lymphoblastic lymphoma (LL) of the B cell type is uncommon, and extranodal presentation is even rarer. It is difficult to suspect this diagnosis without clinically obvious lymph nodal mass or bone marrow involvement. A 3-year-old girl presented with progressive pain and swelling of the right knee joint of 3 months duration. Radiograph revealed expansile lytic lesion at the supracondylar area of the right femur; with pathological fracture and multiple lytic areas in both femora. She neither had lymphadenopathy nor organomegaly. Her blood counts, peripheral smear examination and bone marrow examination were normal. Right supracondylar biopsy revealed diagnosis of a precursor B cell LL. Computerized tomography scan revealed a hypodense, poorly enhancing lesion in the left adnexal region. Although rare, precursor B-cell LL may present with extensive bone lesions. Early and accurate diagnosis of this entity is very important due to its high cure rates.
Keywords
Lymphoblastic lymphoma, Precursor-B cell, Skeletal involvement at presentation
Introduction
Lymphoblastic lymphoma (LL) of the B cell type is uncommon, and extranodal presentation is even rarer. The skin is the most common site of extranodal involvement, followed by bone. It is difficult to suspect this diagnosis without clinically obvious lymph nodal mass or bone marrow involvement. We report a case of precursor B-cell LL who presented with extensive bony lesions and without clinically obvious lymph nodal mass or bone marrow involvement.
Case Report
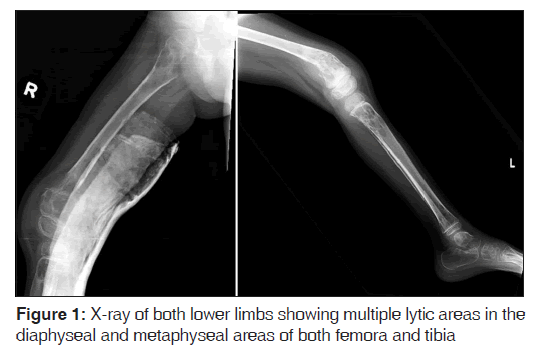
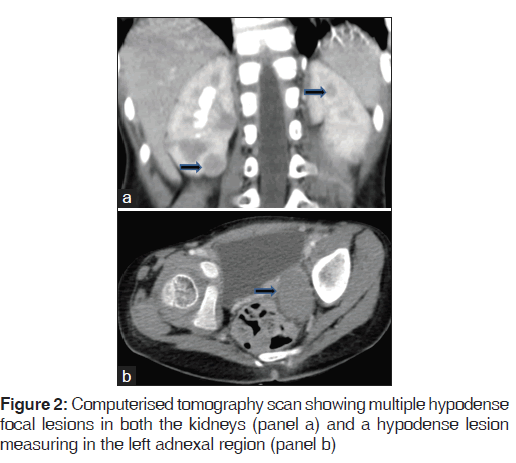
A 3-year-old girl presented with right knee pain and swelling of 3 months duration. She had a fracture of the left femoral diaphysis 6 weeks prior to presentation. There was no history of any constitutional symptoms. Radiograph revealed expansile lytic lesion at the right supracondylar region and multiple lytic areas in the diaphyseal and metaphyseal regions of both femora and tibia [Figure 1]. She had been treated with antibiotics for suspected osteomyelitis. In view of non-response to antibiotics, possibility of malignancy was considered. Biopsy revealed a small round cell tumor. When she presented to us, she had mild pallor, but no icterus, rash, hemorrhagic manifestations, edema, lymphadenopathy or organomegaly. Her investigations revealed hemoglobin 105 gm/L, white blood cell count 7.4 × 109/L, platelet count 445 × 109/L, neutrophils 52%, lymphocytes 33%, monocytes 5% and bands 10%. Peripheral smear examination did not reveal blasts. Serum uric acid level was 350 μmol/L and lactate dehydrogenase level was 368 U/L. Urinary vanillylmandelic acid was not increased. Bone scan revealed foci of patchy radiotracer concentration in the proximal shaft of the bilateral tibia, distal shaft of the bilateral femur, proximal shaft and head of the right femur and proximal shaft of the left femur. Increased radiotracer concentration was seen in the right superior pubic ramus. Computerized tomography scan revealed multiple, well-defined expansile lytic lesions of varying sizes without sclerotic rim involving the bilateral femoral heads and femoral shafts, bilateral iliac blades, bilateral ischial tuberosity, right ilio-pubic ramus, vertebral body of D9 vertebrae, bilateral scapulae, bilateral humeral head and humeral shafts. Multiple hypodense focal lesions were seen in both the kidneys, the largest measuring 15 × 16 mm in size in the right lower pole [Figure 2; panel A]. A hypodense, poorly enhancing lesion measuring 3.1 cm × 3.0 cm × 2.2 cm was seen in the left adnexal region [Figure 2; panel B]. Bone marrow aspiration revealed occasional abnormal cells. Bilateral bone marrow biopsy could not reveal any abnormal cells.
Right supracondylar biopsy review from an apex oncology institute revealed a high-grade hematolymphoid malignancy with blastic morphology. On immunohistochemistry, tumor cells were positive for leucocyte common antigen, CD79a, CD10 and terminal deoxynucleotidyl transferase, while being negative for CD20, CD3, CD5, c-kit and desmin, suggestive of precursor B-cell lymphoblastic lymphoma. Unfortunately, the parents refused treatment for socioeconomic reasons. The rate of refusal of treatment is about 20% in our unit.
Discussion
LLs are mainly of the T-cell type, with only 10-20% being of the B-cell type.[1] There are approximately 100 immunophenotyped cases of pre-B LL reported in the literature, most as single case reports or very small series.[2] Unlike T-LL, pre-B LL predominantly presents with extranodal masses, especially those involving the skin with low propensity for leukemic involvement.[2] Bone is the second most common site of extranodal involvement. There are only few individual case reports and small series of pre B-LL presenting as primary bone lesions. Skeletal radiographic changes associated with and/or suggestive of acute leukemia/lymphoma include diffuse osteopenia, metaphyseal bands, periosteal new bone formation, geographic lytic lesions, sclerosis, mixed sclerosis/lysis and permeative destruction.[3] Bony lesions in pre-B LL are usually osteolytic, although few cases with osteoblastic lesions have been reported.[4] The most frequent osseous sites are the femur, followed by the tibia. Other sites included the humerus, vertebrae, scapula, foot and rib.[4] Our patient had multiple osteolytic lesions in the femur, tibia, pelvis and vertebrae. Parathormone-related protein (PTHrP) produced by lymphoblast is a possible factor causing multiple osteolytic lesions and hypercalcemia in these patients.[5] Despite multiple lesions, our patient did not have hypercalcemia.
As bone marrow involvement is uncommon, biopsy from the soft tissue or bony lesion is useful. Positive immunohistochemical staining of the neoplastic cells for terminal deoxynucleotidyl transferase (TdT), CD10, CD79a, CD34, CD43, CD45/ CD45RB, CD20 and CD99 (MIC2) is useful for diagnosis.[6] CD20 may be negative in pre B-LL, as seen in the present case. CD99 is also expressed by Ewing’s sarcoma cell lines, which is a close differential diagnosis for solitary bone lesion, but TdT can reliably differentiate Ewing’s sarcoma from pre B-LL. Furthermore, characteristic t (11;22) (q24;q12) translocation can distinguish Ewing’s sarcoma from pre-B-LL.
Pre-B-LLs have been treated both with short intensive and with acute lymphoblastic leukemia (ALL)-like chemotherapy, but outcomes were better with ALL protocols.[2] A retrospective analysis of 83 children with ALL suggests that children with few (one to four) bone lesions have an indolent form of leukemia with short duration and good prognosis. On the other hand, more than four bony lesions suggest longer duration of disease and poor prognosis.[3] Patients without radiographic skeletal abnormalities have an “aggressive” form of leukemia.[3] Multiple lesions in our patient can be explained by longer duration of symptoms. Despite multiple lesions, inappropriately low levels of uric acid and lactate dehydrogenase in our patient further support the indolent nature of the lesions.
Conclusion
Precursor B-cell LL presenting with extensive skeletal involvement is rare. Clinically and histologically, it may be confused with other small round cell tumors. The curability of LL in contrast to other metastatic malignancies makes early and accurate diagnosis of paramount importance. Hence, LL must be considered in the differential diagnosis of extensive bony lesions, and adequate immunohistochemical markers must be used to differentiate this entity.
Source of Support
Nil.
Conflict of Interest
None declared.
References
- Knowles DM. Lymphoblastic lymphoma. In Knowles DM, editor. Neoplastic Hematopathology. 2nd ed. Philadelphia: Lippincott Williams & Wilkins; 2001. p. 915-52.
- Belgaumi AF, Al-Kofide A, Sabbah R, Shalaby L. Precursor B-cell lymphoblastic lymphoma (PBLL) in children: Pattern of presentation and outcome. J Egypt Natl Canc Inst 2005;17:15-9.
- Heinrich SD, Gallagher D, Warrior R, Phelan K, George VT, MacEwen GD. The prognostic significance of the skeletal manifestations of acute lymphoblastic leukemia of childhood. J Pediatr Orthop 1994;14:105-11.
- Recine M, Castellano-Sanchez AA, Sheldon J, Schwartz M, Cabello-Inchausti B. Precursor B-cell lymphoblastic lymphoma/ leukemia presenting as osteoblastic bone lesions. Ann Diagn Pathol 2002;6:236-43.
- Shimonodan H, Nagayama J, Nagatoshi Y, Hatanaka M, Takada A, Iguchi H, et al. Acute lymphocytic leukemia in adolescence with multiple osteolytic lesions and hypercalcemia mediated by lymphoblast-producing parathyroid hormone-related peptide: A case report and review of the literature. Pediatr Blood Cancer 2005;45:333-9.
- Iravani S, Singleton TP, Ross CW, Schnitzer B. Precursor B lymphoblastic lymphoma presenting as lytic bone lesions. Am J Clin Pathol 1999;112:836-43.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.