Potential Biomarker for Peri-Implantitis: A Cross-Sectional Study of Type I Collagen Levels in the Sulcular Fluid in relation to Cross-Linked N-Telopeptide and Calprotectin
2 Associate Professor, Department of Prosthodontics, Vice President for Post Graduate & Scientific Research, Riyadh Elm University, Riyadh, Saudi Arabia, Email: drassery@riyadh.edu.sa
3 Lecturer, Department of Prosthodontics, College of Dentistry, Riyadh Elm University, Riyadh, Saudi Arabia
4 Intern, Alfarabi Private College, Jeddah, Saudi Arabia
Citation: Alotaibi DH, et al. Biomarker for Peri-Implantitis. Ann Med Health Sci Res. 2020;10:1158-1162
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Background: Peri-implantitis results after inflammation and bone resorption around implant site. The crevicular fluids present in that region are rich in N-Telopeptide (NTx) and Calprotectin, which are assumed as potential biomarkers. The aim was to assess and compare the calprotectin and NTx levels in the peri-implant sulcular fluid (PISF) from implant sites with or without peri-implantitis. Methodology: A total of fifty healthy patients with a single dental implant were recruited. The peri-implant clinical parameters, namely; probing pocket depth (PPD), bleeding upon probing (BOP), and gingival index (GI), was recorded for each patient. Radiographic images for each implant were obtained to assess the bone loss. PISF was collected using sterile paper strips for detection of calprotectin and NTx levels using the ELISA technique. The relationships between the peri-implant clinical parameters and the level of PISF calprotectin and NTx were analyzed. Results: The overall mean calprotectin level in PISF samples was 120.7 ± 0.78ng per µL and 235.4 ± 0.42ng per µL for healthy and diseased groups, and these values were statistically significant (p=0.003). The overall mean NTx level in PISF samples was 6.93 ± 0.52 ng per µL and 10.78 ± 0.33 ng per µL for healthy and diseased groups, respectively, and these values were statistically significant between the groups (p=0.002). The mean bone loss in healthy and disease sites was 48.8% and 20.2%, respectively. There was a positive correlation between GI scores and Calprotectin (r=0.57; p<0.001); PPD and Calprotectin (r=0.67; p<0.001). Similarly, a positive correlation was also observed between bone loss rate and NTx (r=0.61; p<0.001). Conclusion: The calprotectin and NTx levels in the PISF could be a promising biomarker for peri-implant inflammation and bone destruction in patients with peri-implantitis.
Keywords
Biomarkers; Calprotectin; N-telopeptides; Peri-implantitis
Introduction
Dental implants have been an effective treatment option with a predictable outcome to replace missing teeth from the past 20 years. This is well supported by a recent meta-analysis which demonstrated a 10-year implant survival rate of 96.4%. [1] However, in past years, there is an increased concern over the number of implant failures caused by peri-implantitis, which is found to affect 20-22% of the patients with implants. [2-4] Periimplantitis is clinically characterized by bleeding upon probing (BOP) and/or suppuration, and increased probing pocket depths (PPDs) compared to the initial depth at the time of implant placement. The clinical and radiographic parameters, such as probing pocket depth (PPD), bleeding upon probing (BOP), bone loss, recession, suppuration, and mobility, are routinely used for clinical diagnosis of peri-implantitis. [5] Such parameters are, however, highly sensitive and not precise enough to help the clinician to determine the onset, progression, and prognosis of the disease. Therefore, there is still a need for new methods or tools for early diagnosis of peri-implantitis and track the disease progression over time.
The diagnosis of peri-implant diseases using biomarkers in peri-implant sulcular fluid (PISF) is gaining considerable recognition. Biomarkers are considered to be more accurate than that of clinical indicators to assess inflammation and tissue deterioration around dental implants. They are widely used for a deeper understanding of peri-implantitis in conjunction with clinical parameters. Biomarkers can be measured through oral fluids such as saliva, gingival crevicular fluid (GCF), and PISF. [5]
The previous study has found an increased level of nuclear factor-κB (RANK), soluble RANK ligand (sRANKL), cathepsin-K, osteoprotegerin (OPG), and sclerostin in patients with peri-implantits. [6] Similarly, Ata-Ali et al., [7] demonstrated a significantly increased level of pro-inflammatory cytokines such as IL-1β, IL-6 IL-10, and TNF-α at the peri-implantitis sites compared to healthy peri-implant tissue. These factors and proteins in GCF and PISF are considered as diagnostic biomarkers for peri-implant diseases. This was well supported in a recent systematic review, which concluded that biomarkers and enzymes in PISF have a promising outcome in differentiating peri-implant disease from the healthy state. [8]
Calprotectin is an inflammation-related protein and has been demonstrated to predict periodontal disease activity. Previous studies have shown significantly high levels of calprotectin in periodontal disease relative to healthy non-diseased sites as estimated by their GCF levels. [9-13] Cross-linked N-telopeptide of type I collagen (NTx) is a common biomarker of bone resorption and is used as a diagnostic tool for several bone metabolism diseases including hyperparathyroidism, osteoporosis, and bone metastasis of cancer. [14,15] However, the role of a biomarker for bone loss in peri-implantits has found varied outcomes. [16]
Therefore, this study aimed to assess and compare the calprotectin and NTx levels in the peri-implant sulcular fluid (PISF) from implant sites with or without peri-implantitis.
Methodology
Ethical aspects and patient recruitment
The present study was conducted at the outpatient department clinics, outpatient department clinics, College of Dentistry, Riyadh Elm University. The institutional review board at the university approved the study with the registration number (FRP/2019/175/54). The study was in accordance with the ethical principles of the World Medical Association Declaration of Helsinki (2013). Patients were informed about the study protocol, and written consent was obtained before they participated in the study. Fifty patients who had received a single dental implant in the previous 5-10 years with the healthy or peri-implant diseased site were recruited. The patients had visited the OPD clinic for routine implant care or with the chief complaint related to the implant.
The patients were in the age group of 26 to 50 years with no underlying systemic conditions and no history of antibiotic intake in the previous three months. The recruited patients either belonged to the healthy or diseased group with 50:50 allocation based on the clinical examination. The patients’ details are presented in Table 1.
| Participants | |
|---|---|
| Gender | |
| Male (%)Â Female (%) |
30 (60%) 20 (40%) |
| Age (years) | |
| Healthy implant sites Diseased implant sites |
36 ± 8.1 42 ± 7.3 |
Table 1: Demographic details of the participants.
Collection of Peri-Implant Sulcular Fluid (PISF) and analysis
The diagnosis of peri-implantitis was based on the presence of perceptible inflammatory changes surrounding the periimplant soft tissues combined with bleeding on probing and/ or suppuration, increased probing pocket depths (PPDs), and progressive bone loss. If the baseline measurements for PPD and bone level were missing, probing pocket depth ≥6 mm with profuse bleeding and bone loss ≥ 3 mm was taken into consideration. [17]
Samples of PISF were collected from both the peri-implantitis and healthy peri-implant sites using sterile paper strips (Periopaper, Oralflow Inc., NY, USA). The sample collection site was isolated using cotton rolls, and the external soft tissue surface was air-dried gently for 5s. Any visible dental plaque was removed using a manual scaler. This was followed by inserting a paper strip into the gingival sulcus up to a maximal depth of 1 mm and held in the position for 30 s. Any paper strips contaminated with blood or pus was discarded, and care was taken to avoid any mechanical irritation of the sulcus. The samples were collected from mesial, distal, buccal, and lingual sites. The volume of the collected PISF was measured with the aid of Periotron 8000 (Pro-Flow Inc., NY, USA). The paper strips were then extracted into 200 μL sterile phosphatebuffered saline (PBS) by centrifugation, and the obtained fluid was stored at −80°C until further analysis.
The collected fluid samples were then sent to the laboratory for the detection of biochemical parameters. Calprotectin level was detected using a ready-to-use solid-phase enzyme-linked immunosorbent assay (ELISA) (Life Technologies (India) Pvt Ltd., India). The NTx level was detected using a competitive ELISA (Ostex, osteomark, Seattle, WA, USA). The detection of both the parameters was performed according to the instruction manual accompanied by the ELISA kit.
Recording of clinical parameters
Following PISF collection, the clinical parameters, namely PPD, BOP, and GI, were recorded. All the measurements were taken using a standard manual periodontal probe (PCP-UNC 15, Hu-Friedy, Leimen, Germany).
The PPD, expressed in millimeters, was measured as the distance between the margin of the peri-implant mucosa and the bottom of the sulcus. The measurement was performed at six aspects (mesio-buccal, disto-buccal, mesio-lingual, disto-lingual, midbuccal, and mid-lingual) around the dental implant. BOP was elicited after the insertion of a probe into the sulcus and gently probing of gingival tissue with the application of light pressure. The presence or absence of bleeding on probing was recorded as (BOP+) or (BOP-), respectively. [18] The GI scores were recorded using the modified Löe and Silness index. [19] The rate of alveolar bone loss was determined using the modified criteria, as described by Schie et al., [20] This technique determines the bone loss as a percentage of the original bone level, starting 1 mm below Cemento Enamel Junction (CEJ). [21] For the same purpose, an intraoral periapical radiograph was obtained using a standard parallel cone technique. [22]
Statistical analysis
All the data were analyzed using the Statistical Package for Social Sciences software (SPSS, version 16.0; SPSS Inc., Chicago, IL, USA). Descriptive data were presented as mean and standard deviations. Tukey post-hoc test was used for calculating the significant difference between healthy and diseased sites for each clinical and biochemical parameters. Spearman’s correlation coefficient was applied to determine the relationship between the clinical parameters and biochemical parameters (calprotectin and NTx). A p-value of less than 0.05 was considered significant.
Results
Comparison of biochemical parameters
Mean calprotectin levels in PISF samples from the healthy and diseased peri-implant site were 46.8 ± 0.37 and 180.8 ± 0.54 ng/ site, respectively [Table 2]. A significant difference in the calprotectin was observed between the healthy and diseased groups (p=0.001). The calprotectin level in the diseased group was approximately four-fold higher compared to the diseased group. The overall mean calprotectin level in PISF samples was 120.7 ± 0.78 ng per μL and 235.4 ± 0.42 ng per μL for healthy and diseased groups, and these values were statistically significant between the groups (p=0.003).
| Biochemical parameters | Healthy sites | Peri-implantitis sites | p value |
|---|---|---|---|
| Calprotectin levels (ng/site) Calprotectin concentration (ng/μL) |
46.8 ± 0.37 120.7 ± 0.78 |
180.8 ± 0.54 235.4 ± 0.42 |
0.001* 0.003* |
| NTx levels (ng/site) NTx concentration (ng/μL) |
3.02 ± 0.23 6.93 ± 0.52 |
7.69 ± 0.87 10.78 ± 0.33 |
0.001* 0.002* |
Table 2: Mean calprotectin and NTx levels in peri-implant sulcular fluid of healthy and diseased implant sites.
Mean NTx levels in PISF samples from the healthy and diseased peri-implant site were 3.02 ± 0.23 and 7.69 ± 0.87 ng/ site, respectively [Table 2]. A significant difference in the NTx level was observed between the healthy and diseased groups (p=0.001). The calprotectin level in the diseased group was approximately 2.5 fold higher compared to the diseased group. The overall mean NTx level in PISF samples was 6.93 ± 0.52 ng per μL, and 10.78 ± 0.33 ng per μL for healthy and diseased groups, respectively and these values were statistically significant (p=0.002).
Comparison of clinical parameters
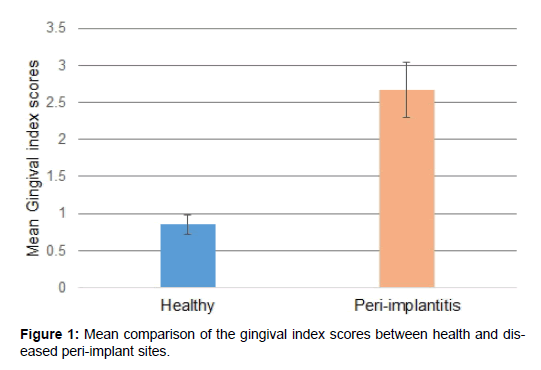
Mean gingival index scores was 0.85 ± 0.13 and 2.67 ± 0.37 for the healthy and diseased implant sites, respectively. The GI scores were significantly different between the groups (p< 0.05) [Figure 1].
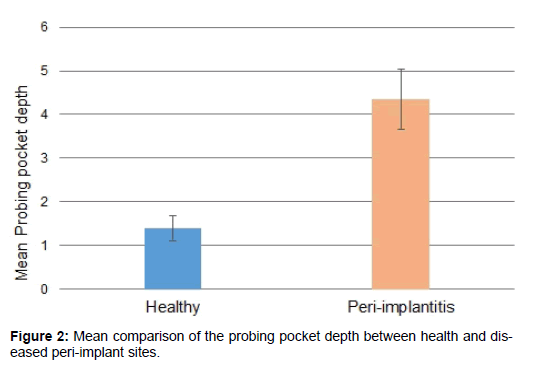
Mean probing pocket depth was 1.39 ± 0.29 mm and 4.36 ± 0.69 mm for the healthy and diseased implant sites, respectively. The PPD values between the healthy and diseased groups varied significantly (p< 0.05) [Figure 2].
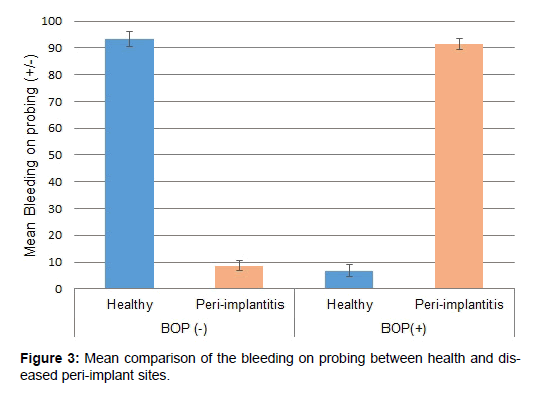
Figure 3 presents the bleeding on probing scores of the healthy and diseased groups. Mean negative bleeding sites were 93.2 ± 2.86 and 8.7 ± 1.79 for the healthy and diseased implant sites, respectively. Similarly, mean positive bleeding sites were 6.8 ± 2.13 and 91.3 ± 2.09 for the healthy and diseased implant sites, respectively.
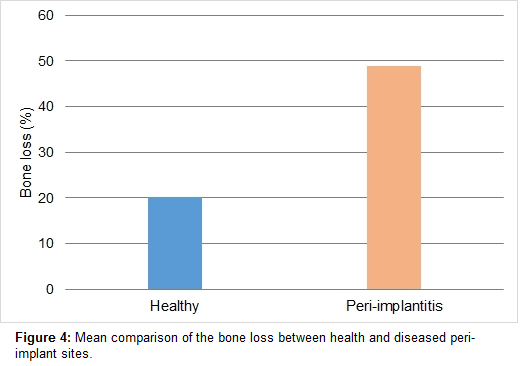
The radiographic analysis to assess the rate of bone loss demonstrated a significant difference in bone loss between the healthy and diseased groups (p<0.05). The mean percentage of bone loss in healthy and disease sites was 48.8% and 20.2%, respectively [Figure 4].
There was a positive correlation between GI scores and Calprotectin (r=0.57; P<0.001); PPD and Calprotectin (r=0.67; P<0.001). Similarly, a positive correlation was also observed between bone loss rate and NTx (r=0.61; P<0.001).
Discussion
Replacement of missing teeth using dental implants has become a routine dental therapeutic procedure and is practiced worldwide with a high success rate. However, local inflammation and bone destruction around the dental implants is a concern, especially if it result in implant failure.[1]
Previous studies have assessed the role of PISF biomarkers in the assessment of peri-implant diseases as clinical parameters alone would not provide an accurate appraisal of peri-implantitis. [6,8,23] Accordingly, in the present study, we assessed the role of calprotectin and NTx as a potential biomarker for the diagnosis of peri-implant disease and to correlate them with the clinical parameters.
Calprotectin is an inflammation-associated protein that is produced in macrophages/monocytes, leukocytes, and epithelial cells. Patients with inflammatory disorders such as ulcerative colitis, cystic fibrosis, and rheumatoid arthritis have demonstrated elevated levels of calprotectin. [24] Furthermore, they have also been detected in GCF and was shown to predict periodontitis. [11,12] Furthermore, GCF calprotectin levels have demonstrated to be well correlated with clinical parameters (GI, PD, and BOP). [12,13] All these results confirm the utility of calprotectin as an essential inflammatory biomarker for various periodontal pathologies. However, there is paucity in the literature regarding the comparison of PISF calprotectin levels of healthy and diseased peri-implant sites. The overall mean calprotectin in this study was found to be significantly higher (p= 0.003), approximately 2.5 fold higher in the diseased sites compared to the healthy sites.
In osteoclasts, degradation of bone type I collagen by cathepsin K leads to the formation of NTx as a product, which is soon released into blood and urine and is a precise biomarker of bone resorption. In patients with different bone pathologies like osteoporosis, hyperparathyroidism, and bone malignancies, NTx level is elevated in the blood and urine of the subjects. Hence, it can be used as a biomarker in these osseous metabolic pathologies. [15,25,26] Similar to calprotectin levels, the present study also demonstrated an increased level of overall mean NTx levels in peri-implantitis sites compared to the peri-implant healthy sites (p= 0.002). A positive correlation was observed between calprotectin levels and NTx levels in the PISF and the mean clinical periodontal parameters. Contrasting results have been found in the literature, where authors, after analyzing NTx levels in the GCF samples among periodontitis patients, showed the absence of NTx in the GCF of healthy subjects. [27] Recently Sakamoto et al., [16] analyzed and compared the calprotectin and NTx levels in PICF from dental implants with or without the peri-implant disease. The authors observed that in periimplant disease sites, PISF calprotectin and NTx levels were significantly higher in comparison to healthy sites. Furthermore, a significant correlation between PICF calprotectin levels and clinical parameters was observed, thus concluding that both calprotectin and NTx are potential biomarkers for periimplantitis diagnosis.
Although the sample size was relatively small and no attempt was done to correlate the calprotectin and NTx levels with the severity of peri-implantitis, yet the data presented in this study showed a relation between these markers and the periimplantitis. Therefore, further studies should be aimed at increasing the sample size and to correlate the calprotectin and NTx levels with the severity of peri-implantitis.
Clinical Significance
Calprotectin and NTx highly contribute to the diagnosis of peri-implantitis. Both biomarkers could be a useful clinical indicator to improve the clinician experience in the diagnosis, progression, and prognosis of peri-implantitis.
Conclusion
Calprotectin and NTx levels of the PISF were significantly increased in peri-implantitis sites compared to healthy implant sites. The calprotectin and NTx levels could be a potential biomarker for peri-implant inflammation and bone destruction in patients with peri-implantitis.
Competing Interests
The authors declare that they have no competing interests.
REFERENCES
- Howe MS, Keys W, Richards D. Long-term (10-year) dental implant survival: A systematic review and sensitivity meta-analysis. J Dent. 2019;84:9-21.
- Mombelli A, Muller N, Cionca N. The epidemiology of peri-implantitis. Clin Oral Implants Res. 2012;23 Suppl 6:67-76.
- Rosen P, Clem D, Cochran D, Froum S, McAllister B, Renvert S, et al. Peri-implant mucositis and peri-implantitis: a current understanding of their diagnoses and clinical implications. J Periodontol. 2013; 84:436-443.
- Derks J, Tomasi C. Peri-implant health and disease. A systematic review of current epidemiology. J Clin Periodontol. 2015;42 Suppl 16:S158-71.
- Heitz-Mayfield LJ. Peri-implant diseases: diagnosis and risk indicators. J Clin Periodontol. 2008;35:292-304
- Rakic M, Struillou X, Petkovic-Curcin A, Matic S, Canullo L, Sanz M, et al. Estimation of bone loss biomarkers as a diagnostic tool for peri-implantitis. J Periodontol. 2014;85:1566-1574.
- Ata-Ali J, Flichy-Fernandez AJ, Alegre-Domingo T, Ata-Ali F, Palacio J, Penarrocha-Diago M. Clinical, microbiological, and immunological aspects of healthy versus peri-implantitis tissue in full arch reconstruction patients: a prospective cross-sectional study. BMC Oral Health. 2015;15:43.
- Dursun E, Tozum TF. Peri-Implant Crevicular Fluid Analysis, Enzymes and Biomarkers: a Systemetic Review. J Oral Maxillofac Res. 2016;7:e9.
- Kaner D, Bernimoulin JP, Dietrich T, Kleber BM, Friedmann A. Calprotectin levels in gingival crevicular fluid predict disease activity in patients treated for generalized aggressive periodontitis. J Periodontal Res. 2011;46:417-426.
- Kido J, Kido R, Suryono, Kataoka M, Fagerhol MK, Nagata T. Calprotectin release from human neutrophils is induced by Porphyromonas gingivalis lipopolysaccharide via the CD-14-Toll-like receptor-nuclear factor kappaB pathway. J Periodontal Res. 2003;38:557-563.
- Kido J, Nakamura T, Kido R, Ohishi K, Yamauchi N, Kataoka M, et al. Calprotectin, a leukocyte protein related to inflammation, in gingival crevicular fluid. J Periodontal Res. 1998;33:434-437
- Kido J, Nakamura T, Kido R, Ohishi K, Yamauchi N, Kataoka M, et al. Calprotectin in gingival crevicular fluid correlates with clinical and biochemical markers of periodontal disease. J Clin Periodontol. 1999;26:653-657
- Nakamura T, Kido J, Kido R, Ohishi K, Yamauchi N, Kataoka M, et al. The association of calprotectin level in gingival crevicular fluid with gingival index and the activities of collagenase and aspartate aminotransferase in adult periodontitis patients. J Periodontol. 2000;71:361-367.
- Wilson AN, Schmid MJ, Marx DB, Reinhardt RA. Bone turnover markers in serum and periodontal microenvironments. J Periodontal Res. 2003;38:355-361.
- Herrmann M, Seibel MJ. The amino- and carboxyterminal cross-linked telopeptides of collagen type I, NTX-I and CTX-I: a comparative review. Clin Chim Acta. 2008;393:57-75
- Sakamoto E, Kido R, Tomotake Y, Naitou Y, Ishida Y, Kido J-I. Calprotectin and cross-linked N-telopeptides of type I collagen levels in crevicular fluid from implant sites with peri-implant diseases: a pilot study. Int J Implant Dent. 2018;4:26.
- Renvert S, Persson GR, Pirih FQ, Camargo PM. Peri-implant health, peri-implant mucositis, and peri-implantitis: Case definitions and diagnostic considerations. J Periodontol. 2018;89 Suppl 1:S304-S312
- Ainamo J, Bay I. Problems and proposals for recording gingivitis and plaque. Int Dent J. 1975;25:229-235.
- Loe H, Silness J. Periodontal Disease in Pregnancy. I. Prevalence and Severity. Acta Odontol Scand. 1963;21:533-551.
- Schei O, Waerhaug J, Lovdal A, Arno A. Alveolar Bone Loss as Related to Oral Hygiene and Age. J Periodontol. 1959;30:7-16.
- Lira-Júnior R, Freires IdA, de Oliveira IL, da Silva ESC, da Silva S, de Brito RL. Comparative study between two techniques for alveolar bone loss assessment: A pilot study. J Indian Soc Periodontol. 2013;17:87-90.
- Updegrave WJ. The paralleling extension-cone technique in intraoral dental radiography. Oral Surg Oral Med Oral Pathol. 1951;4:1250-1261.
- Faot F, Nascimento GG, Bielemann AM, Campao TD, Leite FR, Quirynen M. Can peri-implant crevicular fluid assist in the diagnosis of peri-implantitis? A systematic review and meta-analysis. J Periodontol. 2015;86:631-645.
- Striz I, Trebichavsky I. Calprotectin - A pleiotropic molecule in acute and chronic inflammation. Physiol Res. 2004;53:245-253.
- Hanson DA, Weis MA, Bollen AM, Maslan SL, Singer FR, Eyre DR. A specific immunoassay for monitoring human bone resorption: quantitation of type I collagen cross-linked N-telopeptides in urine. J Bone Miner Res. 1992;7:1251-1258
- Joerger M, Huober J. Diagnostic and prognostic use of bone turnover markers. Recent Results Cancer Res. 2012;192:197-223.
- Becerik S, Afacan B, Ozturk VO, Atmaca H, Emingil G. Gingival crevicular fluid calprotectin, osteocalcin and cross-linked N-terminal telopeptid levels in health and different periodontal diseases. Dis Markers. 2011;31:343-352.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.