Predictors of Loss to Follow‑up Among HIV‑infected Patients in a Rural South‑Eastern Nigeria Hospital: A 5‑year Retrospective Cohort Study
- *Corresponding Author:
- Dr. Eguzo KN
Department of Academic Family Medicine, University of Saskatchewan, 172-1621 Albert Street, Regina, SK S4P 2S5, Saskatchewan, Canada.
E-mail: kelechi.eguzo@usask.ca
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
Abstract
Background: Patient attrition has been a challenge in managing HIV programs in resource‑limited settings. Aim: This study reviews the predictors of loss to follow‑up (LTFU) in our hospital and suggests the best practices for dealing with the issue. Subjects and Methods: A 5‑year retrospective cohort study of 1256 HIV‑infected patients. Baseline CD4 counts, age, gender, year of enrolment, and antiretroviral therapy combination regimen were considered in this study. Kaplan–Meier models were used to estimate the univariate time‑to‑LTFU and Cox proportional hazards models to identify the multivariate predictors of LTFU. Results: Twenty‑four percent (23.9% [301/1256]) of patients were lost to follow‑up. Baseline CD4 count, year of enrolment, and drug combination were significant predictors of LTFU. Patients enrolled earlier (2008/2009) were twice as likely to be LTFU compared with those enrolled later (2010–2013). Gender and age did not significantly predict LTFU nor confound other predictors. Conclusion: The program showed higher LTFU rates than most studies in Nigeria and Africa, maybe due to difficulties with the access to the hospital and possible treatment fatigue. This study recommends the provision of transportation subsidies and proactive patient follow‑up with “peer‑tracking” to reduce LTFU among HIV infected patients, especially in resource‑limited settings.
Keywords
Cohort studies, HIV infections, Loss to-follow-up, Nigeria, Transportation
Introduction
In the last decade, Nigeria has scaled up the antiretroviral program for people living with HIV. The number of people accessing antiretroviral therapies (ARTs) rose from 10,000 (2002) to 300,000 (2010), especially through the U.S President’s Emergency Plan for AIDS Relief.[1] Our hospital was activated in August 2008 to provide ART services in southeastern Nigeria. Despite this free treatment, it is surprising that many patients still dropped out of the various programs at this facility. Rates of the loss to follow-up (LTFU) in resource-limited settings such as ours reported in the literature range from 16% to 40%.[2-4] Onoka et al. studied HIV programs in South-eastern Nigeria and reported LTFU rate between 11% and 32.8%.[5]
Researchers have shown that LTFU among HIV-infected patients in resource-limited settings is significantly associated with being male, baseline CD4 count <200/μL, age <35 years and with stavudine containing first line regimen.[2] Few studies have been conducted to research into the predictors of LTFU in this region of Nigeria. Onoka et al. reported that LTFU for patients in South-eastern Nigeria was more likely for males, patients with CD4 count ≤200/μL and enrolees of programmes in public hospitals.[5] The follow-up period for this particular study was rather short (median 14 months), and did not consider the differences, based on treatment regimens throughout the follow-up time.
LTFU has been shown to contribute to poorer health outcome for patients and constitutes resource wastage, as well as the promotion of HIV drug resistance.[2,5] Research, however, has not differentiated LTFU rates between patients on ART and non-ART. Also, the rate of LTFU could be utilized as an index of program effectiveness with respect to patient tracking.[6] This study aims to identify the predictors of LTFU in a rural hospital, and to highlight best practices in improving patient retention in similar settings.
Subjects and Methods
Study setting and participants
This study was conducted at Nigerian Christian Hospital (NCH), Aba-Nigeria; a rural 110-bed faith-based hospital affiliated with the Churches of Christ and International Health Care Foundation. Since August 2008, more than 3000 HIV-infected people have been enrolled in its program. This program offers the voluntary counselling and testing, prevention-of-mother-to-child-transmission, pediatric and non-pediatric ARTs, as well as immunology services. Currently, there are about 2000 clients on treatment, and about 40 clients are seen each day in clinic. The majority of patients enrolled in the program were recruited through antenatal care, voluntary testing, referral and in-patient hospitalization. A team of doctors, nurses, community health officers as well as monitoring and evaluation staff ensured patient follow-up and tracking of appointments. The hospital also offers specialist services in surgery, ophthalmology, oncology, internal medicine as well as obstetrics and gynaecology.[7]
According to the NCH treatment protocol, patients were considered eligible for ART if they were in the WHO clinical Stage 3 or 4 irrespective of CD4 cell count; if they were pregnant irrespective of CD4 count, and if they had a CD4+ cell count ≤350 cells/μL. Details of other treatment criteria were per the National and WHO guidelines.[1,8] Follow-up appointments ranged from fortnightly to quarterly, depending on the progression of the clinical disease, adherence profile, and prevailing circumstance. They were monitored for adherence and retention through home visits, frequent phone calls as well as attendance at support group meetings. Monitoring was conducted through the IQ Care software (Futures Group, Washington D.C, USA).
This research is a retrospective cohort study of HIV patients aged 15 years or older who were enrolled at the NCH between August 1, 2008, and October 25, 2013. The inclusion criteria were; (1) subjects must be HIV positive, ≥15 years of age and (2) enrolled at NCH. It excluded the patients who were transferred to other treatment facilities during the study period. Approval for the study was obtained from the Board of NCH. Patient confidentiality was maintained by using anonymized data extracted from the database.
Treatment, monitoring and endpoints
NCH program uses Truvada and Combivir based Highly Active Antiretroviral Therapy. First line treatment includes a combination of lamivudine-3TC and zidovudine (ZDV) (Combivir), with either nevirapine (NVP), or efavirenz (EFV). Truvada® (emtricitabine [FTC]/tenofovir [TDV]) with either NVP or EFV is also a first line. Regimen choice was based on baseline CD4 count, pregnancy status, tuberculosis co-infection, and previous ARTs exposure. Co-trimoxazole prophylaxis (960 mg q.d.) was provided to patients with CD4 ≤400 cells/μL. Following ART initiation, patients were followed up for 2 weeks to monitor for drug reactions and assess adherence. Subsequently, ARTs were dispensed on a monthly basis. With consistent adherence and follow-up, patients who were considered stable were given 2-monthly appointment schedules. Second line therapy was provided to clients who had clinical, or virologic failure on fi rstline therapy. The 2nd line therapy included a combination of lopinavir/ritonavir (Kaletra®, Aluvia®-ALV) with either Truvada® or Combivir®. For the purpose of this research, these combinations were categorized as Combivir-based first line (3TC/ZDV with EFV or NVP); Truvada-based first line (TDV/FTC with EFV or NVP); Combivir-based second line (3TC/ZDV with ALV), and Truvada-based second line (TDV/FTC with ALV).
This study considered the immunologic classification of the disease using baseline CD4 count, based on WHO criteria.[9] Patients were classified as follows: CD4 ≥ 500/μL- “not significant disease;” 350–499/μL- “mild disease;” 200–349/μL- “advanced disease” and <200/μL- “severe disease.” Age at enrolment was further classified as 15–24 years; 35–44 years, and ≥45 years; in line with the classification used in peer-reviewed literature.[10-12] Table 1 shows the baseline characteristics of patients included in the study.
| Characteristics | Number of patients | Number LTFU (%) |
|---|---|---|
| LTFU | 301 (23.9%) | |
| Gender | ||
| Male | 398 | 107(35.6) |
| Female | 858 | 194(64.5) |
| Enrolment year | ||
| 2008 | 156 | 46(15.3) |
| 2009 | 341 | 99(32.9) |
| 2010 | 173 | 38(12.6) |
| 2011 | 255 | 52(17.3) |
| 2012 | 216 | 56(18.6) |
| 2013 | 115 | 10(3.3) |
| Care program | ||
| ART | 1045 | 157(52.2) |
| Non-ART | 190 | 133(44.2) |
| Age group (years) | ||
| 15-24 | 51 | 17(5.65) |
| 25-34 | 422 | 115(38.2) |
| 35-44 | 508 | 109(36.2) |
| 45+ | 275 | 60(19.9) |
| Baseline CD4 staging (cells/µL) | ||
| >500 | 183 | 73(24.3) |
| 350-499 | 174 | 40(13.3) |
| 200-349 | 325 | 54(17.9) |
| <200 | 594 | 134(44.5) |
| 200-349 | 156 | 10(3.3) |
| <200 | 594 | 253(84.1) |
| Drug regimen | ||
| Combivir-based first line | 383 | 58(19.3) |
| Truvada-based first line | 547 | 88(29.2) |
| Combivir-based second line | 66 | 6 (2) |
| Truvada-based second line | 53 | 4(1.3) |
LTFU: Loss to follow-up, NCH: Nigerian Christian Hospital, ART: Antiretroviral therapy
Table 1: Baseline characteristics and the proportion of LTFU among 1256 HIV patients in NCH
LTFU, the main endpoint in this study, was defined as a patient who missed an appointment for more than 6 months after the last visit and could not be traced until the end of the study period.[13] The date of the last visit was taken as the date of the event. Patients were considered censored, if dead (based on reported date or last visit date) or if they were alive at the end of the study period (October 25, 2013).
Statistical analysis
The dependent variable in this study is the patient’s status, categorized as either dead, alive, transferred, or LTFU. Univariable Kaplan–Meier analysis was performed at α =0.05 level to determine the independent predictors of LTFU using sex, year of enrolment, age group, baseline WHO CD4 category, treatment program, and drug combination. Significant baseline variables in the univariable analysis were included in the final multivariable model. All other analyses were two-sided, and level of significance was set at P < 0.05. Statistical analysis was done using SAS version 9.3. (SAS Institute Inc., Cary, NC, USA). Kaplan–Meier method was used to estimate the retention rates after ART initiation, and log-rank tests to compare the survival curves. Cox proportional hazards models were used to identify the predictors of LTFU and calculate the hazard ratios (HRs). The survival time was calculated in months using the time interval between the date of enrolment and (1) the date of the event (LTFU) or (2) date of censoring.
Results
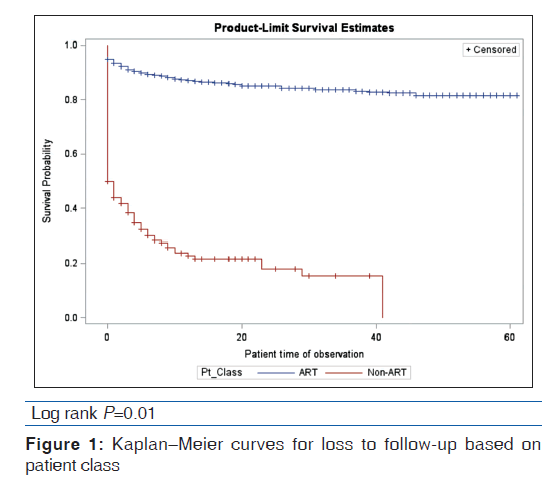
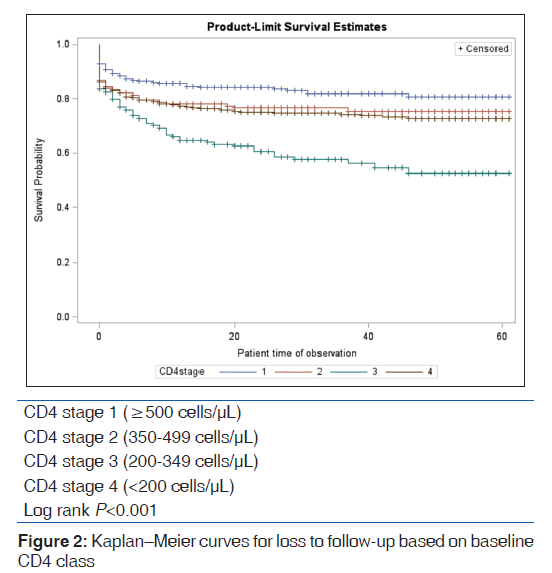
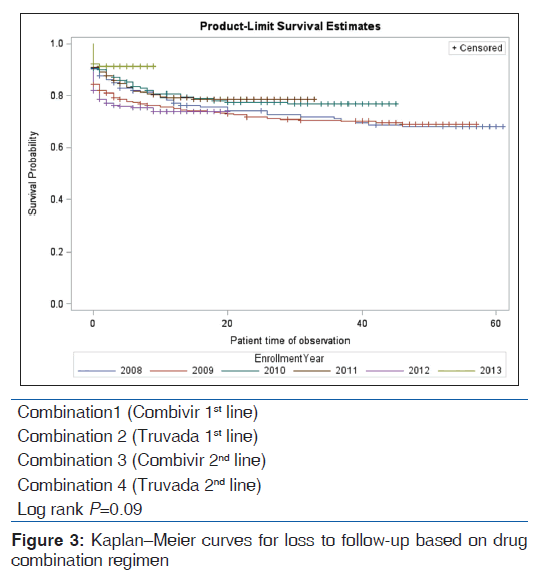
A total of 1256 patients were eligible for this study, comprising 68.3% (858/1256) females and 31.7% (398/1256) males. The median age in the study was 37 years; males 41 years and females 35 years, respectively. 23.9% (307/1256) patients were lost to follow-up during the 5-year period after enrolment in the program while 76% (955/1256) were censored. Table 2 shows the baseline characteristics of study population. The median LTFU time for non-ART clients was 1-month while those on ART could be not estimated as the majority of the observations were censored. In total, those LTFU comprised 36.8% (107/307) males and 64.5% (194/307) females. Based on age, those LTFU comprised 5.6% (17/307) aged 15–24; 38.2% (115/307) aged 25–34; 36.2% (109/307) aged 25–44, and 19.9% (60/307) aged above 45 years. In terms of the year of enrolment, the highest proportion of patients LTFU (32.9% [99/307]) was recorded in 2009. LTFU was most common among the patients with baseline CD4 < 200/μL (82.4% [253/307]) and lowest among those with baseline CD4 counts of 200–349/ml, 3.3% (10/307). LTFU was more prominent among patients taking Truvada-based first line agents 28.7% (88/307) compared with 18.9% (58/307) for those on the Combivir-based first line. At the end of the study period, 955 patients were censored comprising 10.2% (97/955) deaths and 89.8% (858/955) alive.
| Last known status | Number (%) | |
|---|---|---|
| Dead | 97 | (7.7%) |
| Alive | 858 | (68.3%) |
NCH: Nigerian Christian Hospital
Table 2: Last know status of HIV patients NCH included in the study
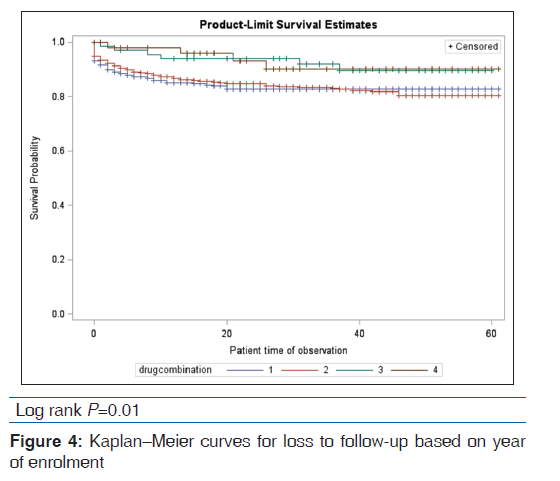
Based on the univariable analysis, baseline CD4 count [Figure 1], year of enrolment [Figure 2], drug combination [Figure 3], and treatment program (ART, non-ART) [Figure 4] were significant predictors of LTFU. Gender and age did not significantly predict LTFU and did not interact/confound the other predictors. Table 3 shows the univariable and multivariable analyses.
| Variable | Unadjusted (univariable) | Adjusted (multivariable) | ||
|---|---|---|---|---|
| HR (95% CI) | P | HR (95% CI) | P | |
| Baseline CD4 staging (vs. >500 cells) | ||||
| <200 | 0.60 (0.45-0.79) | <0.001 | 1.00 (0.62-1.62) | 0.99 |
| 200-349 | 0.54 (0.37-0.80) | <0.001 | 0.42 (0.21-0.85) | 0.02 |
| 350-499 | 0.40 (0.27-0.55) | <0.001 | 0.75 (0.44-1.27) | 0.28 |
| Drug combination | ||||
| Truvada versus Combivir first line | 0.95 (0.68-1.33) | 0.78 | 0.82 (0.58-1.16) | 0.26 |
| Truvada versus Combivir second line | 0.90 (0.25-3.20) | 0.88 | 0.95 (0.27-3.34) | 0.94 |
| Enrolment year (vs. 2008) | ||||
| 2009 | 1.02 (0.72-1.45) | 0.89 | 0.87 (0.56-1.36) | 0.55 |
| 2010 | 0.76 (0.50-1.17) | 0.30 | 0.53 (0.30-0.96) | 0.03 |
| 2011 | 0.74 (0.50-1.11) | 0.14 | 0.41 (0.23-0.73) | <0.001 |
| 2012 | 1.06 (0.71-1.57) | 0.76 | 0.66 (0.58-1.15) | 0.14 |
| 2013 | 0.44 (0.22-0.87) | 0.02 | 0.33 (0.12-0.86) | 0.02 |
NCH: Nigerian Christian Hospital, LTFU: Loss to follow-up, HRs: Hazard ratios, CI: Confidence interval
Table 3: HRs of LTFU according to predictors for HIV patients in NCH
Discussion
This 5-year retrospective cohort study of people living with HIV/AIDS sheds light on the concept of LTFU and its determinants in a program administered in a rural Nigerian hospital. Previous studies found that the male patients were more likely to suffer attrition; our study did not find any significant difference in LTFU rates based on age and gender, and these were neither confounding nor interacting with any other predictors considered in the study. However, we found that the patients who were enrolled earlier in the program (2008 and 2009) were at least twice more likely than those enrolled later (2010–2013) to be lost to follow-up. Hazard of LTFU for patients enrolled in 2008 was 3 times higher than 2013 enrolees (HR 3.1 95% confidence interval [CI] 1.16–8.17; P = 0.02), while the hazard for those enrolled in 2009 was 2 times higher (HR 2.69 95% CI 1.05–6.88, P = 0.04). Baseline CD4 counts and drug combinations were not found to be significant predictors of LTFU in this study.
Overall, our LTFU rate was more than twice the value reported by Onoka et al., who compared patient attrition between public and private institutions.[5] Several factors could be responsible for this; the functioning of the hospital was adversely affected by insecurity between 2009 and 2012 due to kidnapping that was rampant in our community. In addition, the access road to the hospital has been in a state of disrepair for more than 3 years. The difficulty in accessing treatment due to transportation difficulties could have discouraged a good number of our patients.
Although the adjusted hazards for patients on ART compared to non-ART patient was not significant, the univariable analysis may help to draw our attention to the concept of “treatment fatigue” among HIV patients. Treatment fatigue describes the psychological exhaustion of patients with chronic diseases, who have to take numerous pills over a long time. Further research should be conducted to explore this concept and its impact on rates of LTFU among HIV patients.
Toward reduction of LTFU among HIV patients: Review of best practices.
Understanding the causes of patient attrition and improving retention in care is challenging in resource-limited-settings.[14] Evidence shows that improved documentation can impact patient attrition. Some patients have been known to be lost to follow-up in one facility, only to end up in another facility as clients.[14] We think it is necessary to improve local documentation and share medical records (to some extent) for patients receiving ART. This can be achieved through greater collaboration among treatment providers in a geographic area. Nigeria has moved toward this by streamlining the coordination of treatment providers under two broad programs; this makes records sharing easy.
Also, the model of treatment programs is crucial in improving patient retention. Although Geng et al. found that LTFU was about 8-fold higher in larger, centralized treatment centers compared to smaller centers. This may not be unconnected with the difficulty in monitoring a large pool of patients, especially in resource-limited settings. Although there is no definition of “large centers,” our experience in NCH suggests that the programs with more than 2000 clients may be more prone to higher LTFU rates. It may be important to limit the size of individual treatment programs to below 2000. This will lead to the activation of more treatment sites that will be closer to the clients’ communities, thus improving tracking. It may also handle the challenge of tracking non-ART patients, who are historically more likely to be lost to follow-up, even in NCH.[14] Retention is also higher for programs with peer group support (such as the support group meetings at NCH) and outreach services (such as home visits).[14]
Meanwhile, many patients experience the transportation difficulties, maybe due to the poor condition of roads (like in NCH), or poverty. Travel times longer than 2 hours have been associated with double LTFU rates.[15] Emenyonu et al. found that the patients who received transportation subsidy were significantly less likely to be LTFU.[16] This suggests that the programs may benefit from some form of transportation subsidy. Evidence also suggests that the patients with strong social support are less likely to abandon treatment.[14] NCH has a vibrant support group for our clients, and this improves “peer-tracking.” Etienne et al. reviewed the treatment programs in 27 low-income countries, and found that sites with “proactive follow-up” models, such as home visits, had lower rates of LTFU.[17] These suggestions are not exhaustive, but only go to show some approaches that are successful. With the rapid penetration of mobile technology in Africa, we encourage program managers to explore its use in patient tracking and retention.
Study limitations
This study is limited because of the sampling strategy. Including all eligible patients provided a skewed sample that made it difficult to explore some statistical quantities. We did not explore other variables that could influence patient retention such as the effect of travel time, social support, and home visits on LTFU at NCH and inability to capture the washout period before switching to second-line antiretroviral agents made it difficult to draw comparisons between levels of therapy.
Conclusion
The study was conducted to identify the determinants of LTFU in a retrospective cohort of HIV patients followed for 60 months. We have shown that people enrolled earlier in the program are more likely to drop out of treatment, thus helping program managers to better focus their interventions on LTFU. Although an observed difference was identified for patients on ART compared to non-ART patients, this difference was not significant when other factors were considered in the adjusted model. Further research is needed to explore the concept of “treatment fatigue” as a factor in LTFU for patients receiving HIV treatment. HIV program managers are also encouraged to intensify the follow-up strategies for patients on ART medications in their programs.
Acknowledgement
The authors acknowledge the support of the staff at the Monitoring and Evaluation Unit of Nigerian Christian Hospital as well as the hospital board towards to the success of this work.
Financial support and sponsorship
Private.
Conflicts of interest
There are no conflicts of interest.
References
- Federal Ministry of Health: National Guidelines for HIV and AIDS Treatment and Care in Adolescents and Adults; 2010. Available from: http://www.who.int/hiv/pub/guidelines/ nigeria_art.pdf. [Last accessed on 2014 Jun 21].
- Charurat M, Oyegunle M, Benjamin R, Habib A, Eze E, Ele P, et al. Patient retention and adherence to antiretrovirals in a large antiretroviral therapy program in Nigeria: A longitudinal analysis for risk factors. PLoS One 2010;5:e10584.
- Messou E, Kouakou M, Gabillard D, Gouessé P, Koné M, Tchehy A, et al. Medication possession ratio: Predicting and decreasing loss to follow-up in antiretroviral treatment programs in Côte d’Ivoire. J Acquir Immune Defic Syndr 2011;57 Suppl 1:S34-9.
- Hønge BL, Jespersen S, Nordentoft PB, Medina C, da Silva D, da Silva ZJ, et al. Loss to follow-up occurs at all stages in the diagnostic and follow-up period among HIV-infected patients in Guinea-Bissau: A 7-year retrospective cohort study. BMJ Open 2013;3:e003499.
- Onoka CA, Uzochukwu BS, Onwujekwe OE, Chukwuka C, Ilozumba J, Onyedum C, et al. Retention and loss to follow-up in antiretroviral treatment programmes in southeast Nigeria. Pathog Glob Health 2012;106:46-54.
- Ware NC, Wyatt MA, Geng EH, Kaaya SF, Agbaji OO, Muyindike WR, et al. Toward an understanding of disengagement from HIV treatment and care in sub-Saharan Africa: A qualitative study. PLoS Med 2013;10:e1001369.
- Eguzo KN, Lawal AK, Eseigbe CE, Umezurike CC. Determinants of mortality among adult HIV-infected patients on antiretroviral therapy in a rural hospital in Southeastern Nigeria: A 5-year cohort study. AIDS Res Treat 2014;2014:867827.
- World Health Organization. Antiretroviral Therapy for HIV Infection in Adults and Adolescents: Recommendations for a Public Health Approach; 2010 Revision.
- WHO. WHO Case Definitions of HIV for Surveillance and Revised Clinical Staging and Immunological Classification of HIV-Related Disease in Adults and Children. Geneva: WHO; 2007. Available from: http://www.who.int/hiv/ pub/guidelines/HIVstaging150307.pdf. [Last accessed on 2014 Nov 02].
- Ogoina D, Obiako RO, Muktar HM, Adeiza M, Babadoko A, Hassan A, et al. Morbidity and mortality patterns of hospitalised adult HIV/AIDS patients in the era of highly active antiretroviral therapy: A 4-year retrospective review from Zaria, Northern Nigeria. AIDS Res Treat 2012;2012:940580.
- Alemu AW, Sebastián MS. Determinants of survival in adult HIV patients on antiretroviral therapy in Oromiyaa, Ethiopia. Glob Health Action 2010;3:5398
- Johannessen A, Naman E, Ngowi BJ, Sandvik L, Matee MI, Aglen HE, et al. Predictors of mortality in HIV-infected patients starting antiretroviral therapy in a rural hospital in Tanzania. BMC Infect Dis 2008;8:52.
- Chi BH, Yiannoutsos CT, Westfall AO, Newman JE, Zhou J, Cesar C, et al. Universal definition of loss to follow-up in HIV treatment programs: A statistical analysis of 111 facilities in Africa, Asia, and Latin America. PLoS Med 2011;8:e1001111.
- Geng EH, Nash D, Kambugu A, Zhang Y, Braitstein P, Christopoulos KA, et al. Retention in care among HIV-infected patients in resource-limited settings: Emerging insights and new directions. Curr HIV/AIDS Rep 2010;7:234-44.
- Rabkin M, Austin J, Nash D. High Patient Retention Rates in a Multinational HIV/AIDS Treatment Program: The Columbia University Mother-to-Child-Plus Experience. In: 17th Conference on Retroviruses and Opportunistic Infections San Francisco, CA; 2010.
- Emenyonu N, Muyindike W, Habyarimana J, Pops-Eleches C, Thirumurthy H. Cash Transfers to Cover Clinic Transportation Costs Improve Adherence and Retention in Care in a HIV Treatment Program in Rural Uganda. In: 17th Conference on Retroviruses and Opportunistic Infections San Francisco, CA; 2010. p. 16-9.
- Etienne M, Burrows L, Osotimehin B, Macharia T, Hossain B, Redfield RR, et al. Situational analysis of varying models of adherence support and loss to follow up rates; findings from 27 treatment facilities in eight resource limited countries. Trop Med Int Health 2010;15 Suppl 1:76-81.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.