Prevalence, Seasonal Dynamics and Hematological Indices of Typhoid Infection in Eastern Uttar Pradesh, India
2 Department of Medicine, All India Institute of Medical Sciences (AIIMS), Raebareli, U.P., India
3 Department of Biotechnology, Dr. Ram Manohar Lohia Avadh University, Ayodhya, U.P., India
Citation: Gupta A, et al. Prevalence, Seasonal Dynamics and Hematological Indices of Typhoid Infection in Eastern Uttar Pradesh, India. Ann Med Health Sci Res. 2020;10:1077-1082.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Background: Typhoid fever is transmitted by contaminated food and water and highly endemic in developing countries including India. Widal agglutination test is widely accepted in the diagnosis of typhoid infection based on baseline titre. Objective: This study was aimed to investigate the prevalence and seasonal dynamics of typhoid infection with an exploration of hematological parameters as a diagnostic tool in Raebareli region. Research Methodology: More than nine hundred febrile patients over a period of one year were diagnosed in a tertiary care hospital for Widal agglutination test with dilution ranging from 1:40 to 1:320. In addition, various hematological parameters were also examined based on CBC and microscopy. Results: Typhoid infection was found prevalent in 22.72% of febrile patient’s particularly in females and peaked around the month of April and September during transient and monsoon period in Raebareli region. Low hemoglobin levels were also significantly detected in typhoid patients with normocytic anemia. Conclusion: Typhoid infection is highly prevalent particularly during monsoon and transition period favoring its transmission based on Widal agglutination test. Moreover typhoid patients were found more prone to normocytic hypochromic anemia than other forms.
Keywords
Typhoid; Widal agglutination test; Normocytic anemia; Hemoglobin; Seasonal dynamics
Introduction
Enteric fever includes febrile illness exclusively to humans caused by Salmonella enterica serovar typhi (S. typhi) causing typhoid fever and Salmonella enterica serovar paratyphi A, B and C (S. paratyphi) 1. Global burden of typhoid fever is estimated to be 11.9-26.9 million cases including 9.7% prevalence in India. [1,2] Clinical manifestations of typhoid include fever, headache, fatigue, loss of appetite, constipation etc. those are nonspecific and indistinguishable from other febrile diseases. Contaminated food and water, poor sanitation and hygiene are major risk factors for causing typhoid fever. Typhoid fever follows a seasonal pattern with a recurrence of peak incidence during same month across year. In Southeast Asian countries e.g. Nepal, Bangladesh and Cambodia typhoid incidence peaks around May to October. [3-5] Similarly, typhoid fever incidences were found on peak during monsoon season (July to October) in north India. [6]
Salmonella belongs to the family Enterobacteriaceae and has 2463 serotypes, strictly adapted to humans and higher primates. [7] Serotype Typhi may be isolated from blood, urine or stool depending on laboratory equipment and technical resources those are not available in most of primary health centres in developing countries. S. typhi have ‘O’ antigen on the cell wall and ‘H’ antigen on flagella against which host immune response produces antibodies. Agglutination is a standard serological reaction leads to the clumping of a cell suspension by a specific antibody against a specific antigen, O-somatic and H-flagellar. These antigens (O and H) are also shared by many other bacteria of same family therefore its accuracy and reliability is debatable regardless of its extensive use in serodiagnosis of typhoid fever in developing countries including India. [8] Widal agglutination test is almost 100 years old and has been widely used for serological diagnosis of enteric fever in most of the developing nations including India due to relatively cheaper, easy to perform, needs minimal training and require no sophisticated equipment.
Significant changes in the hematological parameters also aid in the clinical diagnosis of typhoid fever. The hematological changes like anemia, lower hematocrit, leucopenia, mild thrombocytopenia and elevated erythrocyte sedimentation rate (ESR) have been reported to be linked with typhoid fever. [9,10] Whereas, hepatic dysfunction has been reported to be linked with Typhoid up to some extent with rare cases of impaired renal functions. [11]
Typhoid fever is cured by using appropriate antibiotics those shorten the duration of fever and bacterial shedding. However, resistance to available antibiotics is more common and increasing rapidly. For example, multiple drug resistance (MDR) to ampicillin, chloramphenicol and Trimethoprimsulfamethoxazole has been emerged in Asia and Africa that limits the use of these traditional drugs. [12] Ceftriaxone, Cefixime, Ofloxacin and new class of broad-spectrum antibiotics called azalides e.g. azithromycin, are found useful against MDR typhoid fever. [13,14] The combination of antibiotics is found to be significantly superior over the use of single antibiotic.
A retrospective study has been conducted in eastern region of Uttar Pradesh, India to evaluate prevalence and seasonal dynamics of typhoid infection in reference to Widal antigen’s titre. In addition, alterations in hematological parameters were also emphasized with respect to sero-diagnosis of O and H antigens and combinatorial antibiotic therapy was used for treatment of typhoid in this region.
Materials and Methods
Study design and population
A hospital based cross-sectional study was carried out on the individuals living at Raebareli district of Uttar Pradesh, India. Widal tests were carried out on the patients suspected for typhoid fever in the laboratory, All India Institute of Medical Sciences (AIIMS), Raebareli. This hospital is a only tertiary care medical center available in this region and fully equipped with all medicare facilities. The patients visited at AIIMS, Raebareli outpatient department (OPD) during the period of one year (from March, 2019 to February, 2020) were considered for this study. A systematic random technique was employed on the febrile patients (n=977) with mean age of 29 ± 14 years, visited OPD and laboratory for Widal test during this period.
Sample collection and size determination
The fresh blood samples were collected in K2-EDTA and plain (without anticoagulant) vacutainers to run complete blood count and Widal test respectively. The minimum sample size of n=930 was determined using modified Fischer’s formula to conduct a study on the prevalence of typhoid fever in Uttar Pradesh, India. [15] A recent systematic review showed a typhoid burden around 9.7% with 95% confidence interval (Cl) in India. [16]
Inclusions and exclusions
Suspected cases of typhoid fever with clinical symptoms such as prolonged fever for a week or more and abdominal pain etc. were included in this study. Febrile patients with other infections e.g. malaria, dengue etc. and patients with the history of ongoing of antibiotic treatment lasts for 3 months before their visit at AIIMS hospital were excluded from the study.
Widal agglutination and tube titration
Fresh blood samples were collected without anticoagulant by vein puncture and centrifuged to separate serum. TYDAL kit (Tulip diagnostics) was used for performing Widal agglutination and titre determination against S. enterica serotype typhi antigens (O and H antigens) and paratyphi (AO, BO, AH, BH, CH, CO antigens). Widal agglutination was performed by mixing a drop of commercial antigens with a drop of serum on test card followed by orbital rotation for one minute. Positive Widal test was determined by the formation of visible agglutination reaction during mixing of antigen and patient’s serum. This procedure was also accompanied by positive and negative controls and final result was reported as reactive or nonreactive. [17]
Titre against reactive antigens (O and H) was determined by tube agglutination using eight tubes. First tube with normal saline as a control and rest of tubes (2-7) with serial dilution of patient’s serum (with normal saline) were taken as test followed by the addition of one drop of TYDAL antigen suspensions. These tubes were incubated at 37ºC for 2-4 h with proper mixing and agglutination was macroscopically observed to estimate the titre. Positive titration value is determined by agglutination at 1:160 or higher dilution for both O and H antigen to determine S. typhi infection in patients. [17]
Study variables and definitions
Anemia is defined as hemoglobin (Hb) less than 13 gm/dL and 12 gm/dL in males and females respectively as per WHO guidelines. [18] Complete blood count (CBC) test was performed on Sysmex XP-100 machine. In addition, microscopy on blood smear of patient’s sample was also performed to define types of anemia. [19] Mean corpuscular volume (MCV) value of 80-100 fl, less than 80 fl and more than 100 fl is defined as normocytic, microcytic and macrocytic anemia respectively. [20]
Statistical analysis
Student t-Test and Annova was used to determine significance for this analysis. A p-value < 0.01 was considered significant for the analysis of results.
Results
Prevalence of typhoid among local population in Raebareli
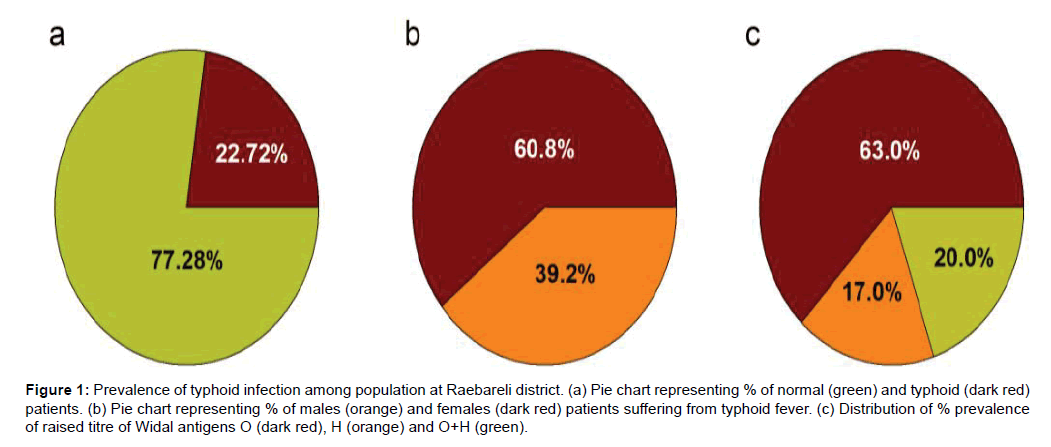
A total of 222 cases (mean age=28.84 ± 14.1 years) out of 977 febrile patients (mean age=36.84 ± 17.95 years) were diagnosed positive for Widal test at OPD, AIIMS, Raebareli. The prevalence of typhoid infection was ~22.72% in the population living at Raebareli district (Figure 1a). The typhoid infection was found more prevalent among females (mean age=28.44 ± 13.48 years) than males (mean age=29.47 ± 15.06 years) (Figure 1b). Furthermore, 63.06% of the patients were diagnosed for increased titre of O antigen (≥ 1:160 dilution) compared to H antigen or combined (O+H) antigens (Figure 1c).
Figure 1: Prevalence of typhoid infection among population at Raebareli district. (a) Pie chart representing % of normal (green) and typhoid (dark red) patients. (b) Pie chart representing % of males (orange) and females (dark red) patients suffering from typhoid fever. (c) Distribution of % prevalence of raised titre of Widal antigens O (dark red), H (orange) and O+H (green).
Seasonal dynamics of typhoid fever
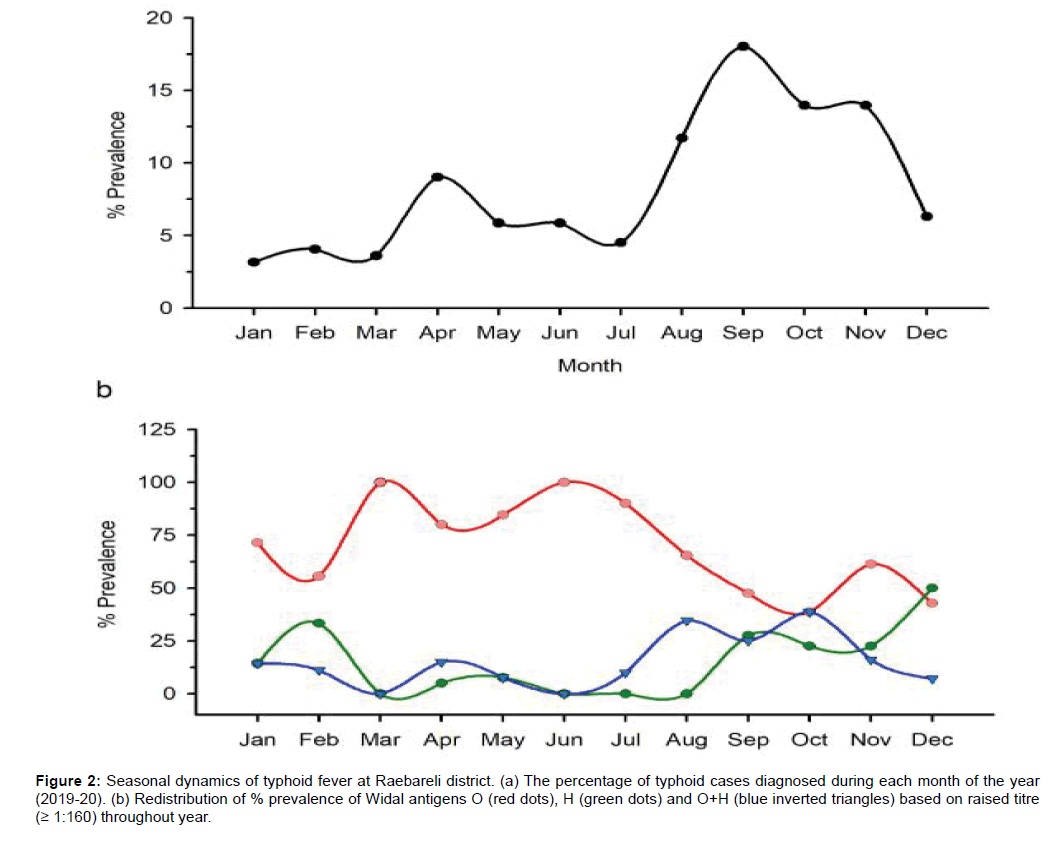
We have also looked for seasonal pattern of typhoid fever which is not fully understood in this region. We have noticed a distinct seasonal cycle of typhoid fever with a peak during month of April and September (Figure 2a). Interestingly, raised titre of O-antigen (≥ 1:160 dilution) was prevalent during the month of March and June (Figure 2b). Whereas, raised titre of H-antigen (≥ 1:160 dilution) was noticed during the month of February and September (Figure 2b). Both O and H-antigen titre were found simultaneously raised during August and October (Figure 2c).
Figure 2: Seasonal dynamics of typhoid fever at Raebareli district. (a) The percentage of typhoid cases diagnosed during each month of the year (2019-20). (b) Redistribution of % prevalence of Widal antigens O (red dots), H (green dots) and O+H (blue inverted triangles) based on raised titre (≥ 1:160) throughout year.
Normocytic hypochromic anemia was highly prevalent among typhoid patients
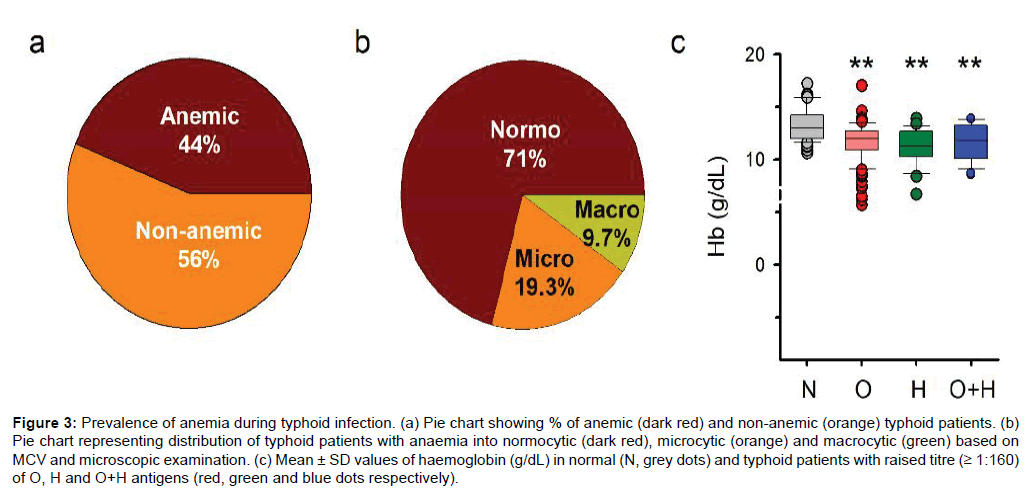
The hematological changes are common during typhoid infection including anemia. [21] Anemia was diagnosed in 44% of the total typhoid patients (Figure 3a). Interestingly, normocytic hypochromic anemia (~ 71%) was more prevalent in patients with typhoid fever and anemia compared to other forms e.g. microcytic and macrocytic (Figure 3b). Hemoglobin levels of typhoid patients were significantly lowered compared to normal healthy individuals with a p-value of 10-4 to 10-7 (n=50) (Figure 3c).
Figure 3: Prevalence of anemia during typhoid infection. (a) Pie chart showing % of anemic (dark red) and non-anemic (orange) typhoid patients. (b) Pie chart representing distribution of typhoid patients with anaemia into normocytic (dark red), microcytic (orange) and macrocytic (green) based on MCV and microscopic examination. (c) Mean ± SD values of haemoglobin (g/dL) in normal (N, grey dots) and typhoid patients with raised titre (≥ 1:160) of O, H and O+H antigens (red, green and blue dots respectively).
Alterations in hematological indices during typhoid infection
Hematological indices are also being reported to be closely associated with typhoid fever. [22] Therefore, we compared various CBC parameters of normal individuals with typhoid patients on the basis of raised titre (≥ 1:160 dilution) of O, H and both (O+H) Widal antigens. Hematocrit levels of patients with both positive O and O+H antigens were significantly lowered (p-value < 0.01, n=36-140) except H antigens as compared to normal healthy individuals (Figure 3c). However, other parameters such as erythrocyte (RBC), leucocyte (WBC), lymphocytes, and neutrophils were non-significantly different among normal and widal positive patients [Table 1]. Interestingly, eosinophil count in widal positive (both O and O+H antigen) patients were found significantly (p-value < 0.01, n=36-140) lower than normal control individuals whereas, platelets count in typhoid patients with positive O antigen was only significantly (p-value < 0.01, n=36-140) higher than normal patients [Table 1]. Cefixime (400 mg) with Ofloxacin (400 mg) or Azithromycin (500 mg) was prescribed to typhoid patients to take orally from 1-2 weeks. The patients were successfully recovered by using these antibiotic combinations in few days.
Table 1: Hematological indices of typhoid patients based positive widal antigen titre and their comparison with normal control patients (titre ≤ 1:80). Statistical analysis was performed between typhoid patients (O, H and O+H antigens) and normal control. (* p-value<0.01, ** p-value<0.00001).
| Parameters | Normal (control) | Serotype: Typhi (titre ≥ 1:160) | ||
|---|---|---|---|---|
| O antigen | H antigen | O+H antigen | ||
| n | 50 | 140 | 42 | 36 |
| Hb (g/dL) | 13.29 ± 1.5 | 11.64 ± 1.7** | 11.26 ± 1.7** | 11.65 ± 1.7** |
| HCT (%) | 39 ± 4.5 | 36.4 ± 5.4* | 37.5 ± 6.4 | 35.2 ± 7.7* |
| RBC (million cells/µl) | 4.4 ± 0.7 | 4.2 ± 0.7 | 4.3 ± 0.9 | 4.3 ± 0.7 |
| WBC (× 103/µl)) | 7.5 ± 2.3 | 6.9 ± 1.6 | 6.8 ± 1.7 | 6.8 ± 2.4 |
| Lymphocytes (%) | 29.4 ± 11.3 | 29.7 ± 10.8 | 28.1 ± 10.2 | 31.0 ± 11.3 |
| Neutrophils (%) | 58.9 ± 10.9 | 59.2 ± 12 | 61.5 ± 11.6 | 59.5 ± 12.2 |
| Eosinophils (%) | 7.2 ± 2.6 | 6.3 ± 2.2* | 6.5 ± 3.0 | 5.5 ± 2.0* |
| Platelets (Lakh/µl) | 1.8 ± 0.7 | 2.2 ± 1.0* | 2.0 ± 1.1 | 2.0 ± 1.0 |
Discussion
The Widal agglutination test is widely used for diagnosis of enteric fever in most of the developing countries including India due to limiting resources for blood culture method despite of being a gold standard for diagnosis of typhoid fever. The classical demonstration of rising titre of the antibodies in paired samples at the interval of two weeks is basically used for serological diagnosis of typhoid. But this rise is not consistently demonstrated due to high levels of background antibody in an endemic region. [23] Therefore, cut off titre for Widal agglutination in such areas depends on background antibody levels and typhoid vaccination. [24]
We have used a cut off titre of 1:80 for our study being widely accepted in most of the studies. This is the first ever study which has been conducted in eastern Uttar Pradesh including Raebareli district based on antibody titre (≥ 1:160) in individuals against serotype Typhi of Salmonella enterica. We have found a presence of high antibody titre in febrile patients with typhoid infection which is prevalent in 22.72% of the population living in this region [Figure 1a]. Other studies from other parts of India reported prevalence of typhoid ranging from 19.5% to 29.32%. [25,26] Interestingly, females (60.8%) were found more prone to typhoid infection in our study irrespective of published reports suggesting male dominance due to proinflammatory cytokine response and poor hygiene practices [Figure 1b]. [27,28] Our results support for pregnancy related issues in females with an average age of 29 years may contribute in high incidence of typhoid infection. [29] In addition, our results also indicated towards a high titre against S. typhi O antigen (≥ 1:160) which is more prevalent alone in 63% of diagnosed typhoid patients compared to H antigen [Figure 1c]. [30]
Ingestion of contaminated food and water is associated with transmission of typhoid fever due to limiting access to clean water and adequate sanitation those are susceptible to climatic events such as drought and monsoons. [31] However, a close association of typhoid infection with key environmental drivers e.g. rainfall and temperature is still lacking suggestive of other factors influencing the transmission. Here, we observed a distinct seasonal pattern for typhoid fever in eastern part of Uttar Pradesh [Figure 2a]. The peak period occurred during the month of April and September, which coincides with the monsoon and transition period. The monsoon is linked with floods which are identified as a risk factor for enteric fever due to contamination of drinking water with sewers containing fecal matter. [28] Whereas, temperature fluctuates during this transition period with mild rain favoring the growth of vectors e.g. flies those contaminate food and increase the risk of typhoid fever. Therefore, both monsoon and temperature fluctuation emerged as key environmental drivers influencing the seasonal dynamics of typhoid fever. We noticed that peak during April and September month coincidence with prevalence of O and H antigen respectively while comparing the peaks of typhoid incidence and raised titre of Widal antigens [Figure 2a and Figure 2b]. This result suggests that raised titre against particular Widal antigen is responsible for seasonal dynamics of typhoid infection.
Hematological parameters are also known to be linked with typhoid infection and therefore aid in its diagnosis. [32,33] We found 44% of typhoid patients were diagnosed for anemia as reported by other studies with a prevalence of 35%-42%. [34] Furthermore, hemoglobin levels were significantly dropped during typhoid infection with raised titre of O, H and combined (O+H) Widal antigens resulting into normocytic hypochromic anemia that was found more prevalent (71%) as compared to other forms e.g. micro and macrocytic anemia in the typhoid patients [Figures 3b and 3c]. In addition, a significant drop in hematocrit during positive O and O+H antigen was vigilant in our findings [Table 1]. Coming to value of differential count in diagnosis of fevers, we have surprisingly found a significant drop in eosinophil count during positive Widal O and O+H antigen [Table 1]. Interestingly, mild thrombocytosis was also significantly diagnosed in typhoid patients with positive O antigen [Table 1]. But other hematological parameters were remained unaltered in our findings.
Based on this study, health department should take special attention during monsoon and transition period e.g. April and September to combat typhoid infection by preventing its transmission and medication using effective antibiotics. CBC parameters should also be included during Widal agglutination test that will surely present a clear picture of typhoid infection. Proper nutrition with iron rich food may reduce the complications of resulting anemia during enteric fever.
Conclusion
Mostly young females are found more prone to the typhoid infection peaked around the month of April and September in Raebareli region. Typhoid fever is found strongly associated with normocytic hypochromic anemia with mild eosinopenia and thrombocytosis significantly vigilant during positive Widal O antigen (≥ 1:160).
Conflict of Interest
The authors declare that they have no conflict of interest
REFERENCES
- Buckle GC, Walker CLF, Black RE. Typhoid fever and paratyphoid fever: Systematic review to estimate global morbidity and mortality for 2010. J Glob Health. 2012;2:010401.
- John J, Van Aart CJC, Grassly NC. The Burden of Typhoid and Paratyphoid in India: Systematic Review and Meta-analysis. PLoS Negl Trop Dis. 2016;10:e0004616.
- Karkey A, Arjyal A, Anders KL, Boni MF, Dongol S, Koirala S, et al. The burden and characteristics of enteric fever at a healthcare facility in a densely populated area of Kathmandu. PLoS One. 2010;5:e13988.
- Das SK, Chisti MJ, Afrad MH, Malek MA, Ahmed S, Ferdous F, et al. Gastroenteritis due to typhoidal Salmonella: a decade of observation at an urban and a rural diarrheal disease hospital in Bangladesh. BMC Infect Dis. 2014;14:435.
- Pham Thanh D, Thompson CN, Rabaa MA, Sona S, Sopheary S, Kumar V, et al. The molecular and spatial epidemiology of typhoid fever in rural cambodia. PLoS Negl. Trop. Dis. 2016;10(6):e0004785.
- Singhal L, Gupta PK, Kale P, Gautam V, Ray P. Trends in antimicrobial susceptibility of Salmonella Typhi from North India (2001-2012). Indian J Med Microbiol. 2014;32:149-152.
- Brenner FW, Villar RG, Angulo FJ, Tauxe R, Swaminathan B. Salmonella nomenclature. J Clin Microbiol. 2000;38:2465-2467.
- Bhutta ZA. Current concepts in the diagnosis and treatment of typhoid fever. BMJ. 2006;333:78-82.
- Ifeanyi OE. Changes in some haematological parameters in typhoid patients attending University Health Services Department of Michael Okpara University of Agriculture, Nigeria. Int J Curr Microbio Appl Sci. 2014;31:670-674.
- Akindele ST, Awoderu AO, Bilesanmi-Awoderu JB. Prevalence of typhoid fever and anemia (low PCV) among patients attending University College Hospital Ibadan, Oyo, Nigeria.
- Nasrallah SM, Nassar VH. Enteric fever: a clinicopathologic study of 104 cases. Am J Gastroenterol. 1978;69:63-69.
- Mirza SH, Beeching NJ, Hart CA. Multi-drug resistant typhoid: a global problem. J Med Microbiol. 1996;44:317-319.
- Smith MD, Duong NM, Hoa NT, Wain J, Ha HD, Diep TS, et al. Comparison of ofloxacin and ceftriaxone for short-course treatment of enteric fever. Antimicrob Agents Chemother. 1994;38:1716-1720.
- Parry CM, Ho VA, Bay PV, Lanh MN, Tham NT, Wain J, et al. Randomized controlled comparison of ofloxacin, azithromycin, and an ofloxacin-azithromycin combination for treatment of multidrug-resistant and nalidixic acid-resistant typhoid fever. Antimicrob. Agents Chemother. 2007;51:819-825.
- Chukwuonye II, Ohagwu KA, Adelowo OO, Chuku A, Obi EC, Onwuchekwa U, et al. Prevalence and predictors of chronic kidney disease in a semiurban community in lagos. Int J Nephrol. 2019;2019:1625837.
- Balaji V, Kapil A, Shastri J, Pragasam AK, Gole G, Choudhari S, et al. Longitudinal typhoid fever trends in India from 2000 to 2015. Am J Trop Med Hyg. 2018;99:34-40.
- Andualem G, Abebe T, Kebede N, Gebre-Selassie S, Mihret A, Alemayehu H. A comparative study of Widal test with blood culture in the diagnosis of typhoid fever in febrile patients. BMC Res. Notes. 2014;7:653.
- Beutler E, Waalen J. The definition of anemia: what is the lower limit of normal of the blood hemoglobin concentration? Blood. 2006;107:1747-1750.
- Gupta A, Gani MU, Kumar P. Macrocytosis is highly prevalent in Raebareli district, Uttar Pradesh, India. International Journal of Research in Medical Sciences. 2020;8:1.
- Maner BS, Moosavi L. Mean corpuscular volume (MCV). StatPearls. 2019.
- Shrivastava K, Vahikar S, Mishra V. Hematological profile in typhoid fever. Trop J Pathol Microbiol. 2015.
- Ndako JA, Dojumo VT, Akinwumi JA, Fajobi VO. Changes in some haematological parameters in typhoid fever patients attending Landmark University Medical Center, Omuaran-Nigeria. Heliyon. 2020.
- Schroeder SA. Interpretation of serologic tests for typhoid fever. Jama. 1968.
- Clegg A, Passey M, Omena M, Karigifa K, Suve N. Re-evaluation of the Widal agglutination test in response to the changing pattern of typhoid fever in the highlands of Papua New Guinea. Acta tropica. 1994.
- Patki R, Lilani S, Lanjewar D. Baseline antibody titre against Salmonella enterica in healthy population of Mumbai, Maharashtra, India. International journal of microbiology. 2017.
- Gunjal SP, Gunjal PN, Patil NK, Vanaparthi N, Nalawade AV, Banerjee S, et al. Determination of baseline widal titres amongst apparently healthy blood donors in ahmednagar, maharashtra, India. J Clin Diagn Res. 2013;7:2709-2711.
- Khan M. A plausible explanation for male dominance in typhoid ileal perforation. Clin Exp Gastroenterol. 2012.
- Dewan AM, Corner R, Hashizume M, Ongee ET. Typhoid Fever and its association with environmental factors in the Dhaka Metropolitan Area of Bangladesh: a spatial and time-series approach. PLoS Negl. Trop. Dis. 2013;7:e1998.
- Sulaiman K, Sarwari AR. Culture-confirmed typhoid fever and pregnancy. Int J Infect Dis. 2007.
- Sreenath K, Sebastian S, Deepa R. Detection of baseline Widal titres among the blood donors: A population basedstudy. Int. J Curr Microbiol App Sci. 2014.3:428.
- Crump JA, Sjölund-Karlsson M, Gordon MA, Parry CM. Epidemiology, clinical presentation, laboratory diagnosis, antimicrobial resistance, and antimicrobial management of invasive salmonella infections. Clin. Microbiol. Rev. 2015;28:901-937.
- Uplaonkar SV, Kauser SH, Tengli MB. Haematological profile in typhoid fever. Indian Journal of Pathology. 2017.
- Kapoor OP. The Importance of Routine CBC and ESR in Common Fevers. Bombay Hospital Journal. 2008.
- Dhillon SP, Lata N, Singh S, Gotwal V, Joshi T, Singh N. To study haematological profile of Enteric fever patients. Int J Curr Res Med Sci. 2017;3:24-29.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.