SARS - CoV-2, Pandemic COVID 19: A Brief Review
1Department of Medicine, King Khalid University, College of Medicine, Abha, Saudi Arabia
2Department of Medicine, GMC Srinagar, Jammu and Kashmir, Kashmir
3Department of Medicine, GMC Baramulla, Jammu and Kashmir, Kashmir
- *Corresponding Author:
- Mir Nadeem
Veterinary Research Division
Department of Medicine
King Khalid University
College of Medicine, Abha, Saudi Arabia
Tel: +966599103476
E-mail: mirnadeem44@gmail.com
Citation:Nadeem M, et al. SARS - CoV-2, Pandemic COVID 19: A Brief Review. Ann Med Health Sci Res. 2020; 10: 846- 856.
This is an open access article distributed under the terms of the Creative Commons Attribution‑NonCommercial‑ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non‑commercially, as long as the author is credited and the new creations are licensed under the identical terms.
Abstract
As a novel coronavirus now known as SARS-COV-2 first reported in Wuhan china in late December 2019, the severe corona respiratory syndrome (SARS-CoV) is responsible for an acute human respiratory syndrome and is linked to Wuhan wholesale sea food market. The virus spread rapidly involving who country and with span of three months WHO had to declare it as pandemic. Although its origins are not entirely understood, these genomic analyses suggest that SARS-CoV-2 probably evolved from a strain found in bats. SARS-COV-2 is zoonotic but human-to-human transmission is also possible. Surveillance and phylogenetic researches indicate SARS-CoV-2 to be closely associated with bats’ corona viruses, suggesting bats as reservoirs, although unconfirmed. With no vaccine currently available for SARS-COV-2 nor approved prophylactics, its global spread to over 150 countries with high virulence highlights its role as ongoing public health threat. An articulated action plan ought to be taken, preferably from a One Health perspective, for appropriately advanced counter measures against COVID 19.
Keywords
Attenuation map; Attenuation correction; 3-D MLEM; Reconstruction; SPECT images
Introduction
Viral infections continue to represent one of the most pressing health is-sues worldwide. Reemerging and novel viruses often lead to widespread outbreaks such as the SARS epidemic of 2003 and the MERS epidemic of 2012. Presently, the outbreak of a severe respiratory syndrome attributed to a novel coronavirus SARS-CoV-2 which was first reported in Hubei province of China in December 2019 and the earliest cases were linked to the Huanan wholesale seafood and wet animal market. Within a span of few days the virus spread rapidly involving the whole country and overwhelming the medical system of the world’s 2nd biggest economy. Soon cases were reported in other countries and the sheer rapidity with which this Infection has spread throughout the world lead WHO to declare it as. A pandemic on 12th March 2020, within 3 months of the first case reported in China. As per the data available, around 550,000 confirmed cases have been reported till March 27th 2020, where’s the number of death attributed to this are around 24,906 and around 176 countries worldwide have been affected by the COVID-19 pandemic and the pandemic continues to evolve as one of the biggest challenge to the health services worldwide.
In this article we try to review the current knowledge about this novel virus as to understand what can be done to prevent its further spread.
Virology and Pathogenesis
Coronaviruses (coronam: Latin: crown) are enveloped positivestranded RNA viruses with a nucleo-capsid core and a crownlike appearance under an electron microscope. This is due to the presence of spike glycoproteins on the envelope giving the appearance of crown. [1] The Coronoviridae family (order Nidovirales) with sub family Orthocoronaviridae is further classified into four genera of CoVs: c (alpha-CoV), Betacoronavirus (beta-CoV), Delta-coronavirus (delta-CoV), and Gammacoronavirus (gamma-CoV). Furthermore, the beta-CoV genus is divided into five sub-genera or lineage. [2,3] Extensive and careful genomic analyses has shown that most likely bats and rodents are the gene sources of alpha-CoVs and beta-CoVs while avian species are probably the gene sources of delta CoVs and gamma CoVs. Although these viruses can cause all types of diseases (Enteric, Respiratory, Neurological) in different animals like bats, cattle, camels and cats. [2,3] however, Till now, a total of seven CoVs have been identified that are capable of infecting humans. however, in general, studies have estimated that 2% of the population may be healthy carriers of coronaviruses and these viruses may be responsible for about 5 to 10% of acute respiratory infections of varying severities. [4]
Common human CoVs: HCoV-OC43, and HCoV-HKU1 (beta- CoVs of the A lineage); HCoV-229E, and HCoV-NL63 (alpha- CoVs). [5,6] They usually cause common colds and self-limiting upper respiratory infections in immunocompetent individuals and lower respiratory infections such as pneumonia’s in the elderly and immunodeficient patients. However, there are other human CoVs such as the SARS-CoV, MERS-CoV and more recently the SARS-CoV 2 (beta-CoVs of the B and C lineage,) that have caused epidemics of variable clinical severity with respiratory and extra-respiratory manifestations and with SARS-CoV, MERS-CoV, the mortality rates have been up to 10% and 35%, respectively. [7,8] Although 2019-nCoV is similar to some beta- coronaviruses detected in bats, it is distinct from SARS-CoV and MERS-CoV. The three 2019-nCoV coronaviruses from Wuhan, together with two bat-derived SARS-like strains, ZC45 and ZXC21, form a distinct clade. SARS-CoV strains from humans and genetically similar SARSlike coronaviruses from bats collected from southwestern China formed another clade within the subgenus sarbecovirus. [9] Since the sequence identity in conserved replicase domains (ORF 1ab) is less than 90% between 2019-nCoV and other members of betacoronavirus, the 2019-nCoV — the likely causative agent of the viral pneumonia in Wuhan — is a novel beta-coronavirus belonging to the sarbecovirus sub- genus of Coronaviridae family [9] This novel coronaviruses was named by the international Committee on Taxonomy of viruses as SARSCoV- 2 and 2019-nCoV by the WHO and based on its genetic proximity to 2 bat-derived SARS-like coronaviruses has likely originated in chrysanthemum bats. The virus uses a densely glycosylated spike (S) protein to enter host cells and binds with high affinity to the angiotensin-converting enzyme 2 (ACE2) receptor in humans in a manner similar to SARS-CoV. [10]
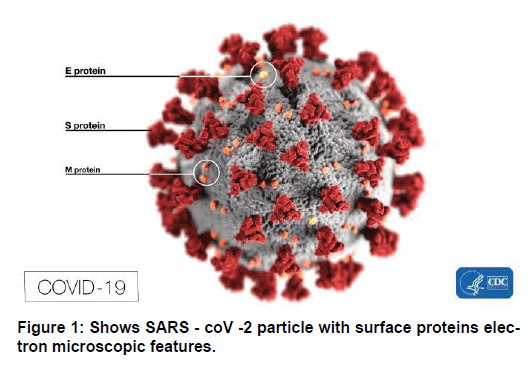
Thus, in order to understand the COVID19, considering its genetic makeup and structural variations is important [Figure 1].
Genetics and Pathophysiology
CoVs are the largest know RNA viruses till now and consist of positive single stranded RNA of approximately 30kb length and with a 5′-cap structure and 3′-poly-A tail. It has round or elliptic and often pleomorphic form, and a diameter of approximately 60–140 nm. [11] Chan et al. have proven that the genome of the new HCoV, isolated from a cluster-patient with atypical pneumonia after visiting Wuhan, had 89% nucleotide identity with bat SARS-like-CoVZXC21 and 82% with that of human SARS-CoV. For this reason, the new virus was called SARS-CoV-2. Its single-stranded RNA genome contains 29891 nucleotides, encoding for 9860 amino acids. Although its origins are not entirely understood, these genomic analyses suggest that SARS-CoV-2 probably evolved from a strain found in bats. [12] Starting from the viral RNA, the synthesis of polyprotein 1a/1ab (pp1a/pp1ab) in the host is released. The transcription works through the replication-transcription complex (RCT) organized in double-membrane vesicles and via the synthesis of subgenomic RNAs (sgRNAs) sequences. However, there are certain transcriptions regulatory sequences located between these so called open reading frames (ORFs) that work as templates for the production of subgenomic mRNAs. In the atypical CoV genome, at least six ORFs can be present. Among these, a frameshift between ORF1a and ORF1b guides the production of both pp1a and pp1ab polypeptides that are processed by virally encoded chymotrypsin-like protease (3CLpro) or main protease (Mpro), as well as one or two papain-like proteases for producing 16 non-structural proteins (nsps). Apart from ORF1a and ORF1b, other ORFs encode for structural proteins, including spike, membrane, envelope, and nucleocapsid proteins. [3] and accessory protein chains. Different CoVs present special structural and accessory proteins translated by dedicated sgRNAs. Among the structural elements of CoVs, there is the spike glycoproteins composed of two subunits (S1 and S2). Homotrimers of S proteins compose the spikes on the viral surface, guiding the link to host receptors. [13]
Thus, for understanding the pathophysiology and virulence of the CoVs in general and SARS-CoV2 in particular, its important to understand the genomics and the functions of the nsps and the structural proteins. For example, research has underlined that nsp is able to block the host innate immune response. [14] Among functions of structural proteins, the envelope has a crucial role in virus pathogenicity as it promotes viral assembly and release. However, many of these features (e.g., those of nsp 2, and 11) have not yet been fully understood and described. Moreover the matter of viral mutations is of utmost importance in assessing disease relapses and virulence. For example, according to recent research, a spike mutation, which probably occurred in late November 2019, triggered jumping to humans. Angeletti et al. compared the Sars-Cov-2 gene sequence with tSars-CoV and compared the trans membrane helical segments in the ORF1ab encoded 2 (nsp2) and nsp3 and found that position 723 presents a serine instead of a glycine residue, while the position 1010 is occupied by proline instead of isoleucine. [15] Thus, further research into the genome and a gene mapping is of fundamental importance to allow the researchers to trace the phylogenetic tree of the virus and, above all, the recognition of strains that differ according to the mutations is of utmost importance.
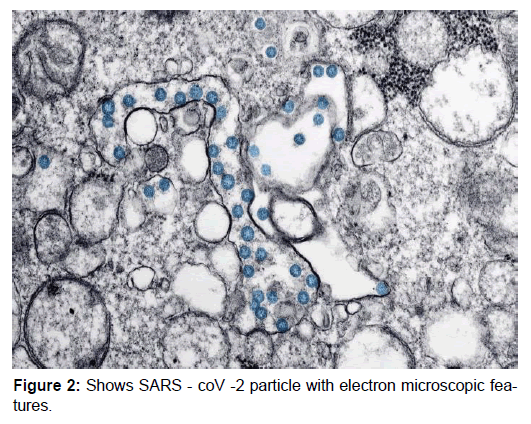
Structural analysis suggests that the virus is able to bind to the angioten-sin-converting enzyme-2 (ACE2) receptor in humans, which suggests that it may have a similar pathogenesis to SARS. [10] The key determinant of SARS coronavirus host specificity is the Spike glycoprotein, an envelope-anchored trimeric protein responsible for binding human Angiotensin Converting Enzyme 2 (ACE2) as the principle receptor for virus docking and entry. SARS-CoV S glycoprotein also binds C-type lectins like DCand/ or L-Sign as a co-receptor, an interaction which is blocked by mannose binding lectin. [16,17] However, a unique structural feature of the spike glycoprotein receptor binding domain of SARS-CoV-2 (which is responsible for the entry of the virus into host cells) confers potentially higher binding affinity for ACE2 on host cells compared to SARS-CoV. [18] A furin- like cleavage site has been identified in the spike protein of the virus; this does not exist in other SARS-like coronaviruses. [19] The viral load starts to increase in the patient as soon as the infection begins and is similar to that of influenza. Even asymptomatic patients have been seen with similar viral loads as symptomatic patients. [20] The Pathophysiological implications of these findings still need to be elucidated in bigger clinical studies and whether patients on ACE inhibitors are at a higher risk is still a question that needs to be answered [Figure 2].
Epidemiology
Viral infections have time and again presented a challenge to the global healthcare systems, more specifically, the novel strains that tend to cause outbreaks of varying intensities. Mostly, these present as the common cold or flu like symptoms, however, in the last twenty years, several viral epidemics have been recorded such as the severe acute respiratory syndrome coronavirus (SARS-CoV) in 2002 to 2003, and H1N1 influenza in 2009, the Middle East respiratory syndrome coronavirus (MERS-CoV) in Saudi Arabia in 2012. Andmost recently, the severe acute respiratory syndrome coronavirus -2 (CoV-2) which was first reported from the Hubei province of China.
Tracing the timeline of the current pandemic, the first cases were reported in the Wuhan town of. Hubei province of China, on December 31 2019 and were linked to the Huanong wholesale seafood market which is a “wet” wholesale seafood market that is where live animals are sold including bats, rats, snakes etc. [21,22] During the initial outbreak, no known causal organism could be elucidated; hence, The Chinese Centre for Disease Control and Prevention (CDC) and local CDCs then organized an intensive outbreak investigation program. As the causal organism wasn’t identified during the initial outbreak, these first cases were labeled as “pneumonia’s of unknown ethology”. The aetiology of this illness has now been attributed to a novel virus belonging to the coronavirus (CoV) family, COVID-19.
On February 11, 2020, the WHO Director-General, Dr. Tedros Adhanom Ghebreyesus, announced that the disease caused by this new CoV was a “COVID-19,” which is the acronym of “coronavirus disease 2019”. Subsequently, the task of experts of the International Committee on Taxonomy of Viruses (ICTV) termed it the SARS-CoV-2 virus as it is very similar to the one that caused the SARS outbreak (SARS-CoVs).
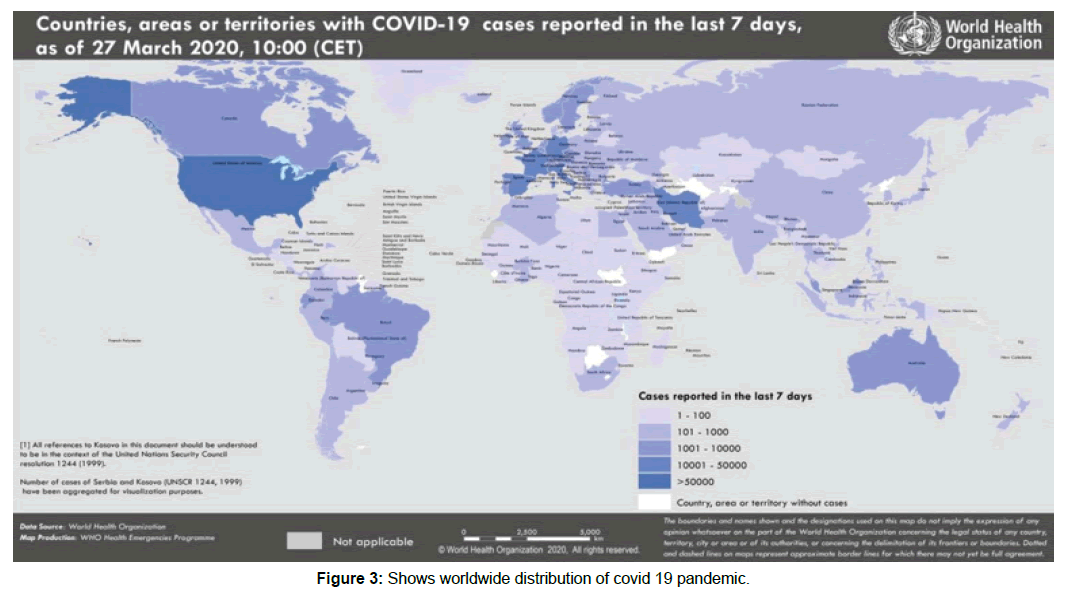
In a meeting on January 30, 2020, per the International Health Regulations (IHR, 2005), the outbreak was declared by the WHO a Public Health Emergency of International Concern (PHEIC) as it had spread to 18 countries with four countries reporting human-to-human transmission. An additional landmark occurred on February 26, 2020, as the first case of the disease, not imported from China, was recorded in the United States. Keeping in view the rapidity with with the infection and spread and suspecting that more countries would be involved, WHO declared the COVID-19 as a pandemic on 12 March 2020 [Figure 3].
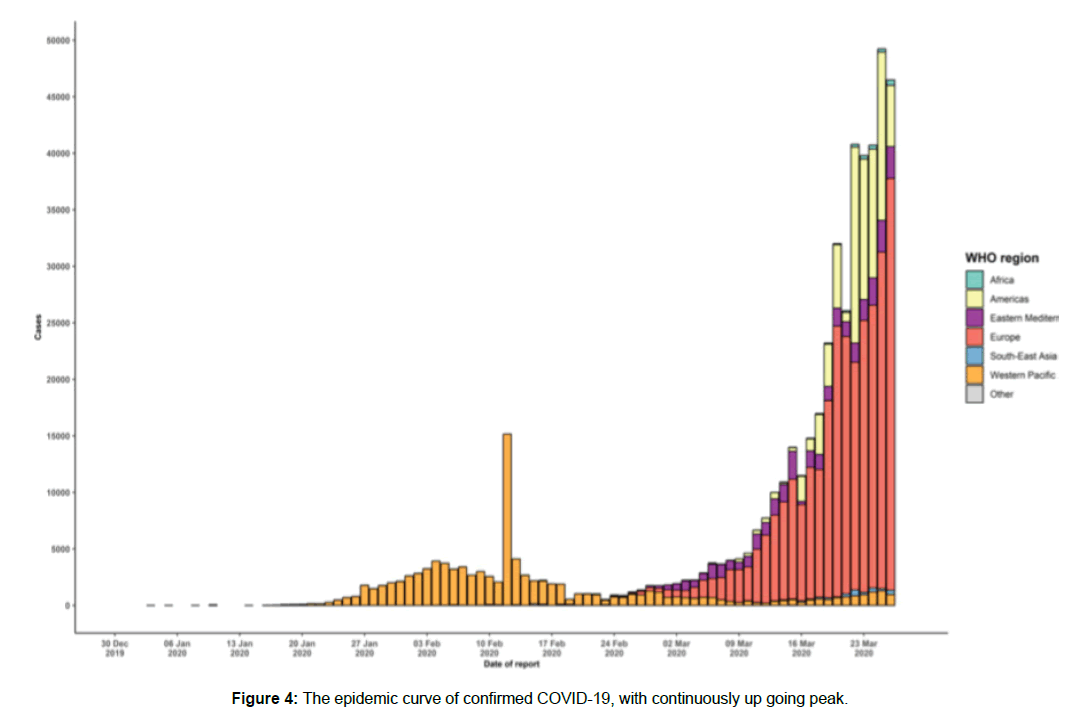
Data provided by the WHO Health Emergency Dashboard (March 20, 8: 36 pm) report confirmed 550,000 cases worldwide since the beginning of the pandemic. Of these, 24,908 (4.58%) have been fatal. in the initial stages of the pandemic almost all confirmed cases (About 92%) were recorded in China, where almost all the deaths were also recorded (96.5%). However, with the rapid spread through rest of the world these dynamics have rapidly changed with an increasing number being reported from Italy, Spain, USA and Iran. As of now, USA has soared past China in the number of confirmed cases worldwide with 85,906 confirmed cases. As of now, 80,589 confirmed cases have been reported in Italy and 64,059 in Spain. Of note, these countries have also reported a number of casualties ie 8215 in Italy and 1307 deaths in USA. Currently Italy stands at the highest mortality rate. According to the World Health Organization and the US Centres for Disease Control and Prevention, the incubation period is estimated to range from 1 to 14 days while the median incubation period has been estimated to be 5 days. However, the transmission may be possible even during the incubation period. Preliminary reports suggest that the reproductive number (R0), the number of people who acquire the infection from an infected person, is approximately 2.2. [23] However, as the situation is still evolving, the R0 may actually be higher or lower. Like other CoVs, it is sensitive to ultraviolet rays and heat. Furthermore, these viruses can be effectively inactivated by lipid solvents including ether (75%), ethanol, chlorine-containing disinfectant, peroxyacetic acid and chloroform except for chlorhexidine [Figure 4].
Source of Infection and Transmission
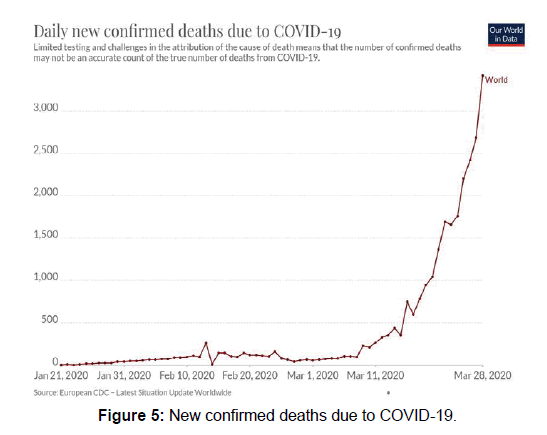
COVID-19 is a viral infection that started possibly as a zoonotic transmission from the Huanan market in China. However, since then the infection has spread from human to human contact although the complete details are still under consideration. Extensive research into the genetic and phylogenetic tree of the virus points towards bats as a reservoir of the infection. However, further research to determine if an intermediate host exists is still required [Figure 5].
COVID-19 is transmitted via dropletsand fomites duringclose unprotected contact between humans. Airborne spread has not been reported for COVID-19 and it is not believed to be a major driver of transmission based on available evidence; however, it can be envisaged if certain aerosol-generating procedures are conducted in health care facilities. Fecal shedding has been demonstrated from some patients, and viable virus has been identified in a limited number of case reports. However, the fecal-oral route does not appear to be a driver of COVID-19 transmission; its role and significance for COVID-19 remains to be determined. [24]
China
The Chinese Centre for Disease Control and Prevention has recently published the largest case series to date of coronavirus disease 2019 (COVID-19) in mainland China (72314 cases, updated through February 11, 2020) [25]. Among these, 62% were confirmed cases on positive nuclei acid test result, in 15% diagnosis was made on clinical history and exposure, 22% were suspected cases in whom testing could not be done and 1% ie 889 cases were asymptomatic. Most case patients were 30 to 79 years of age (87%), 1% were aged 9 years or younger, 1% were aged 10 to 19 years, and 3% were age 80 years or older. Most cases were diagnosed in Hubei Province (75%) and most reported Wuhan-related exposures (86%; ie, Wuhan resident or visitor or close contact with Wuhan resident or visitor). [25] Most cases were classified as mild (81%; ie, non-pneumonia and mild pneumonia). However, 14% were severe 5% were critical (ie, respiratory failure, septic shock, and/or multiple organ dysfunction or failure) the overall case-fatality rate (CFR) was 2.3% (1023 deaths among 44 672 confirmed cases). No deaths occurred in the group aged 9 years and younger, but cases in those aged 70 to 79 years had an 8.0% CFR and cases in those aged 80 years and older had a 14.8% CFR. No deaths were reported among mild and severe cases. The CFR was 49.0% among critical cases. CFR was elevated among those with preexisting comorbid conditions-10.5% for cardiovascular disease, 7.3% for diabetes, 6.3% for chronic respiratory disease, 6.0% for hypertension, and 5.6% for cancer. Among the 44 672 cases, a total of 1716 were health workers (3.8%), 1080 of whom were in Wuhan (63%). Overall, 14.8% of confirmed cases among health workers were classified as severe or critical and 5 deaths were observed. [25]
Thus COVID-19 in China rapidly spread from a single city to the entire country within just a month. The sheer pace of both the abrupt in-crease in number of cases and the vast geographic spread across the country has surprised and quickly overwhelmed health and public health services in China, particularly in Wuhan City and Hubei Province. Epidemic curves reflect what may be a mixed outbreak pattern, with early cases suggestive of a continuous common source, potentially zoonotic spillover at Huanan Seafood Wholesale Market, and later cases suggestive of a propagated source as the virus began to be transmitted from person to person.
Italy
As the pandemic is evolving,, Italy has become one of the worst hit country after China. Since the identification of COVID-19 travel-related cases in northern Italy, the number of people confirmed to be infected in Italy Has increased rapidly. As of 16th march 2020, Italy has had 27,980 cases of SARS-CoV-2 infections, with most cases in Northern Italy in the Lombardia and Veneto regions. [26] Travel-related cases were the main source of COVID-19 cases during the early stages of the current epidemic in Italy. The month of February, however, has been dominated by two large clusters of outbreaks in Northern Italy, south of Milan, with mainly local transmission the source of infections. The outbreak illustrates how rapidly and easily a new virus can spread through local transmission in a naive population. The region of Lombardy has the highest number of cases of coronavirus in Italy and appears to be the epicentre of the Italian outbreak. It has been suggested that a 38-year-old man from the city of Codogno, who presented at the hospital on 20 February 2020 with a serious respiratory infection, could be the first local human-to-human transmission case in the region, as he had dined with a friend who had been in China. [27]
Latest public made official data from the IstitutoSuperiore di Sanita, 4th leading scientific technical body of the Italian National Health Service, show that the clinical status of 2539 cases is known, of which 25% are labelled critical or severe, 30% have mild symptoms, and 10% are asymptomatic (the rest are either pauci- symptomatic or the severity level is not specified). Currently, 21% of cases have been admitted to hospital, and 1545 patients are in intensive care. The median age of those in intensive care is 69 (age 51-70: 46%; age >70: 44%), with no cases under the age of 18. However, a significant percentage of patients are under 30, which confirms how crucial this age group is in transmitting the virus. [28]
The rapidity of spread of the epidemic and a sudden increase in the number of patients has overwhelmed the medical and preventive policies of the country and the country has witnessed an increased fatality rate with a case fatality rate of 10.1% (8215/ 80589) which is much higher than that for China. [28,29] On 21st March, Italy recorded the highest number of deaths ie 793 due to coronavirus in a single day.
While it is being debated upon that why Italy has fallen short in combating the pandemic, a number of issues have come up. For example, Italy is experiencing a chronic shortage of healthcare workers. Doctors and healthcare workers have been working multiple shifts and it has put a toll on the already deficient health care system. The long recovery time from pneumonia caused by the coronavirus is also contributing to overcrowding in already deficient intensive care units. Pneumonia is 5-10% lethal among admitted patients, rising to 30% in those admitted to intensive care. Intubation may be required in a patient admitted for more than two weeks, thus occupying a bed that cannot be used by others. [28] Another reason perhaps is the demographic distribution of the country. The Italian population has around 23% people in the elderly group aged 65 years or older. Moreover, it has been seen that the COVID-19 has an increased mortality in this age group. This may partly explain the increased fatality of the disease in the country. [30]
The most important lacuna perhaps, has been a delayed response to the outbreak, missing the crucial first weeks in curtailing the spread of the infection. All this has perhaps led to more than twice case fatality rate in Italy as compared to China. However, as the disease continues to unfold newer trends may become apparent.
USA
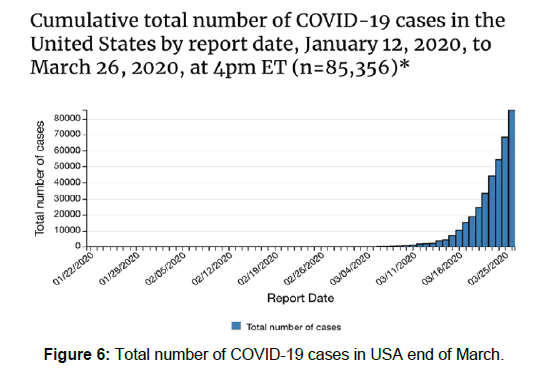
CDC and WHO reports suggest Unites states of America have already crossed the 100,000 confirmed cases, surpassing the case burden in China. A total of 1704 deaths have also been reported. As more data becomes available, these numbers will continue to change [Figure 6].
Clinical Features
The clinical spectrum of SARS-CoV2 is varied and ranges from asymptomatic to a mild disease to severe conditions characterized by respiratory failure that necessitates mechanical ventilation and ICU care and multi organ dysfunction and this diversity in presentation has in-fact caused complications in diagnosis and recognition of the disease at the earliest. Approximately 80% of patients present with mild illness, 14% present with severe illness, and 5% present with critical illness. Early reports suggest that illness severity is associated with older age and the presence of underlying health conditions. [31,32] Some patients may be minimally symptomatic or asymptomatic.
An analysis of the recent case series reveals that the most common symptoms are fever, cough, dyspnea, myalgia, fatigue while some less commonly encountered symptoms were chest pain, rhinorrhea, diarrhea, hemoptysis, headache, confusion, dizziness, anorexia, sputum production, abdominal pain. [22,23,33]
Approximately 90% of patients present with more than one symptom, and 15% of patients present with fever, cough, and dyspnoea. [23] It appears that fewer patients have prominent upper respiratory tract or gastrointestinal symptoms compared with SARS, MERS, or influenza. [22,23] Patients may present with nausea or diarrhea 1 to 2 days prior to onset of fever and breathing difficulties. Most children present with mild symptoms, without fever or pneumonia. However, they may have signs of pneumonia on chest imaging despite having minimal or no symptoms. [34-36]
Accumulating evidence suggests that a subgroup of patients with severe COVID-19 might have a cytokine storm syndrome, identification and treatment of hyperinflammation using existing, approved therapies with proven safety profiles to address the immediate need to reduce the rising mortality. A cytokine profile resembling Secondary haemophagocyticlymphohistiocytosis (sHLH) is associated with COVID-19 disease severity, characterized by increased interleukin (IL)-2, IL-7, granulocyte- colony stimulating factor, interferon-γ inducible protein 10, monocyte chemo- attractant protein 1, macrophage inflammatory protein 1-α, and tumour necrosis factor-α [32]. Predictors of fatality from a recent retrospective, multi-center study of 150 confirmed COVID-19 cases in Wuhan, China, included elevated ferritin (mean 1297•6 ng/ml in non- survivors vs. 614•0 ng/ml in survivors; p<0•001) and IL-6 (p<0•0001). [35] Suggesting that mortality might be due to virally driven hyper inflammation.
Retrospective reviews of pregnant women with COVID-19 found that the clinical characteristics in pregnant women were similar to those reported for non-pregnant adults. [37,38] A retrospective case series of 62 patients in Zhejiang province found that the clinical features were less severe than those of the primary infected patients from Wuhan City, indicating that second-generation infection may result in milder infection. Similar findings have been reported with MERS.
Approximately 2% of the patients had a history of direct contact with wildlife, whereas more than three quarters were either residents of Wuhan, had visited the city, or had contact with city residents.
Conventional routes of transmission of SARS- CoV, MERSCoV, and highly pathogenic influenza consist of respiratory droplets and direct contact, mechanisms that probably occur with SARS-CoV-2 as well. Because SARS-CoV-2 can be detected in the gastrointestinal tract, saliva, and urine, these routes of potential transmission need to be further studied.
The Chinese CDC reports have divided the clinical manifestations of the disease by their severity: Mild disease: non-pneumonia and mild pneumonia (81% of cases). Severe disease: dyspnea, respiratory frequency ≥ 30/min, blood oxygen saturation (SpO2) ≤ 93%, PaO2/FiO2 ratio [the ratio between the blood pressure of the oxygen (partial pressure of oxygen, PaO2) and the percentage of oxygen supplied (fraction of inspired oxygen, FiO2)] < 300, and/or lung infiltrates > 50% within 24 to 48 hours; ( 14% of cases) Critical disease: respiratory failure, septic shock, and/or multiple organ dysfunction (MOD) or failure (MOF); (5% of cases).
However, these are the data from the earlier reported cases and the presentations and complications may change as further countries are affected.
Evaluation
The diagnosis of a COVID 19 presents a major challenge because of the varied clinical presentations. And most countries have now come up with some type of epidemiological or clinical survey to determine whom to test. According to the U.S. CDC, most patients with confirmed COVID-19 have developed fever and/or symptoms of acute respiratory illness (e.g., cough, difficulty breathing). If a person is a PUO, it is recommended that practitioners immediately put in place infection control and prevention measures. Initially, they recommend testing for all other sources of respiratory infection. Additionally, they recommend using epidemiological factors to assist in decision making.
There are epidemiological factors that assist in the decision on who test. This includes anyone who has had close contact with a patient with laboratory confirmed COVID-19 within 14 days of symptom onset or a history of travel from affected geographic areas (presently China, Italy, Iran, Japan, and South Korea) within 14 days of symptom onset.
The WHO recommends collecting specimens from both the upper respiratory tract (naso and oropharyngeal samples) and lower respiratory tract such as expectorated sputum, endotracheal aspirate, or bronchoalveolar lavage. The collection of BAL samples should only be performed in mechanically ventilated patients as lower respiratory tract samples seem to remain positive for a more extended period. The samples require storage at four degrees Celsius. In the laboratory, amplification of the genetic material extracted from the saliva or mucus sample is through a reverse polymerase chain reaction (RT-PCR). The probes used are based on the initial gene sequence released by the Shanghai Public Health Clinical Centre & School of Public Health, Fudan University, Shanghai, China on Virological. org, and subsequent confirmatory evaluation by additional labs. If the test result is positive, it is recommended that the test is repeated for verification. In patients with confirmed COVID-19 diagnosis, the laboratory evaluation should be repeated to evaluate for viral clearance prior to being released from observation.
Apart from this, routine clinical investigations such as CBC, KFT, LFT, ESR, CRP and chest radiographs must be ordered in all suspected patients. In the early stages of the disease, a normal or decreased total white blood cell count and a decreased lymphocyte count has been demonstrated. Lymphopenia has been mentioned as a negative prognostic factor. Increased values of liver enzymes, LDH, muscle enzymes, and C-reactive protein can be found, however, patients mostly have a normal procalcitonin value. In critical patients, multi-organ dysfunction has been seen with a raised d-dimer.
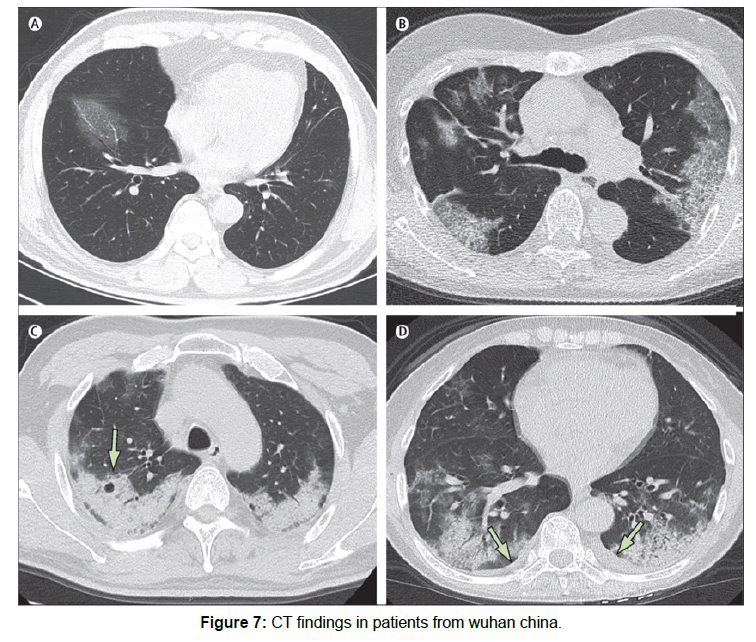
Ct findings: Wu J al studied Ct images total of 130 patients diagnosed with COVID-19 from seven hospitals in China. They reported unilateral lesions in 10.7% and bilateral in 89.3% of cases. Most commonly, the lesions were distributed sub pleurally (78.4%), centrilobular lesions in 76.1% and diffuse in 6.1% of cases. Ground glass opacity with or without consolidation was the most common finding in the scans. Moreover, they reported some accompanying signs, most importantly, the “pleural parallel sign” in 75.3%. Other signs included the vascular thickening, reversed halo sign, halo sign, pneumatocoeles. [39] Thus is of utmost importance that a high index of suspicion is held while diagnosing a patient with SARS-CoV2. All clinical, radiological and laboratory parameters have to be kept in consideration while diagnosing and triage of these patients so that supportive and preventive measures can be initiated at the earliest.
Figure 7 CT images of 56-year-old man, day 3 after symptom onset: focal ground-glass opacity associated with smooth interlobular and interlobular septal thickening in the right lower lobes. (B) 74-year-old woman, day 10 after symptom onset: bilateral, peripheral ground-glass opacity associated with smooth interlobular and intralobularseptal thickening (crazypaving pattern). (C) 61-year-old woman, day 20 after symptom onset: bilateral and peripheral predominant consolidation pattern with a round cystic change internally (arrow). (D) 63-year-old woman, day 17 after symptom onset: bilateral, peripheral mixed pattern associated with air bronchograms in both lower and upper lobes, with a small amount of pleural effusion (arrows).
Screening and Testing
Both the WHO and CDC recommend screening based on travel to China (especially Hubei Province) or close contact with a person diagnosed or under investigation for SARS-CoV-2 in the past 14 days with fever and/or respiratory symptoms. However, given the increasing number of cases in other countries such as South Korea, Italy, and Japan, and the recent report of a case in California with no history of travel or known contact with someone who traveled, has changed the scenario.
People who may have been exposed to individuals with suspected COVID-19 (including healthcare workers) should be advised to monitor their health for 14 days from the last day of possible contact, and seek immediate medical attention if they develop any symptoms, particularly fever, respiratory symptoms such as coughing or shortness of breath, or diarrhea. [40] Some people may be put into voluntary or compulsory quarantine depending on the guidance from local health authorities.
All patients with severe COVID-19 should be screened for hyper-inflammation using laboratory trends (eg, increasing ferritin, decreasing platelet counts, or erythrocyte sedimentation rate) and the HScore, [41] To identify 5 the subgroup of patients for whom immunosuppression could improve
mortality. Therapeutic options include steroids, intravenous immunoglobulin, selective cytokine blockade (eg, anakinra or tocilizumab) and JAK inhibition.
Preventive Measures
At times like this, preventive measure are the basic block to prevent the spread of the disease into the community and thus to decrease the morbidity and mortality associated with it. The only way to prevent infection is to avoid exposure to the virus [42,43] and people should be advised to:
• Frequent hand wash with soap and water or use of an alcohol based sanitize
• Avoid touching the eyes, nose, and mouth with unwashed hands.
• Maintain a distance of at least 1 meterbetween each other, social distancing is one of the most important aspects of preventive measure to stop spread in community
• Respiratory hygiene is also an important part, always cover your mouth and nose while sneezing and coughing.
• Seek medical care early if they have a fever, cough, and difficulty breathing, and share their previous travel and contact history with their healthcare provider.
• Avoid the consumption of raw or undercooked animal products, and handle raw meat, milk, or animal organs with care as per usual good food safety practices.
• The World Health Organization (WHO) does not recommend that people wear a medical mask in community settings if they do not have respiratory symptoms as there is no evidence available on its usefulness to protect people who are not ill. Individuals with fever and/or respiratory symptoms are advised to wear a mask, particularly in endemic areas. [44]
• Enforced quarantine has been used in some countries to isolate easily identifiable cohorts of people at potential risk of recent exposure (e.g., groups evacuated by aero plane from affected areas, or groups on cruise ships with infected people on board). The psychosocial effects of enforced quarantine may have long-lasting repercussions. [45,46]
Treatment and Management
At present, there is no confirmed definitive treatment of the SARS-CoV-2. So the mainstay of treatment is proper triage of patients and proper supportive care focusing on the systemic complications. Patients should be managed in a hospital setting where possible; however, home care may be suitable for selected patients with mild illness. Immediate isolation of all patients suspected to have COVID 19 is a must to prevent further spread.
Prompt admission of patients with pneumonia or respiratory distress to an appropriate healthcare facility and proper supportive care depending on the clinical presentation is important in management. The median time from onset of symptoms to hospital admission is reported to be approximately 7 days. [22,32] Patients with impending or established respiratory failure should be admitted to an intensive care unit. Between 23% to 32% of hospitalized patients require intensive care for respiratory support. [22,23,32] However, with each day these figures continue to change and as more cases continue to emerge, these figures will probably change.
Supportive therapies
Supportive measures are the most important treatment options as long as definitive measures to combat the disease emerge. Some of these are:
• Oxygen: In patients with hypoxaemia, respiratory distress and shock, supplemental oxygen at a rate of 5L/ minute or higher to reach a target SpO2 > 90% [47]
• Fluids: Judicious use of iv fluid in absence of shock is recommended as overloading with it fluid may lead to worsening of hypoxaemia. [47]
• Antipyretic for symptomatic relief of fever. However, use of Ibuprofen has been associated with worse outcomes and is not recommended. [47]
• Broad spectrum anti microbials depending on the local epidemiological data should be started within the first hour in patients suspected to have secondary sepsis. [47]
• Neuraminidase inhibitors should be given until influenza is ruled out [47]
However, these are all supportive measures and patients need careful monitoring to predict and manage systemic complications as they arise. [47]
Mechanical ventilation
Intubation and mechanical ventilation may be required in patients who cannot maintain oxygenation (SpO2>90%) despite supplemental oxygen and in patients with impending respiratory arrest. However, precautions must be taken while intubation and experienced health workers should undertake it under complete precautions. [47]
Emerging treatments
Antivirals: Various antivirals have been tried in patients on trial basis including oseltamivir, lopinavir/ritonavir, ganciclovir, favipiravir, baloxavirmarboxil, umifenovir, interferon alfa); however, at present there are no data to support their use. [48-51]
Remdesivir has shown in vitro activity against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and has been used to treat patients in China, as well as the first patient in the US. [52,53] Clinical trials with remdesivir have started in the US and in China.
Intravenous immunoglobulin is also being tried in some patients with COVID-19; however, there are no data to support this.
Chloroquine and hydroxychloroquine are being trialled in some patients with COVID-19; however, there are no data to support this. Chloroquine shows in vitro activity against SARS-CoV-2. [54] Chloroquine is likely to be added to the updated version of the Chinese management guidelines.
Kevzara (Sarilumab) is a fully human monoclonal antibody, which inhibits the interleukin-6 (IL-6) pathway and is being tried on trial basis to treat severe COVID-19 patients in the New York, US. IL 6 is associated with the overactive inflammatory response in the lungs of severely or critically ill COVID-19 patients. However, at present no clear guidelines are present to guide treatment.
As during previous pandemics (severe acute respiratory syndrome and Middle East respiratory syndrome), corticosteroids are not routinely recommended and might exacerbate COVID- 19-associated lung injury [54], however, in hyper inflammation, immunosuppression is likely to be beneficial. A multi-center, randomized controlled trial of tocilizumab (IL-6 receptor blockade, licensed for cytokine release syndrome), has been approved in patients with COVID-19 pneumonia and elevated IL-6 [37] in China (ChiCTR2000029765). [55,56] Janus kinase (JAK) inhibition could affect both inflammation and cellular viral entry in COVID-19. [41]
Stem cell therapy
Stem cell therapy is being investigated to treat patients with COVID-19 in clinical trials. It is thought that mesenchymal stem cells can reduce the pathological changes that occur in the lungs, and inhibit the cell- mediated immune inflammatory response. [57]
Angiotensin-II receptor antagonists
Angiotensin-II receptor antagonists such as losartan are being investigated as a potential treatment because it is thought that the angiotensin-converting enzyme-2 (ACE2) receptor is the main binding site for the virus. [58]
Convalescent plasma
Convalescent plasma from patients who have recovered from viral infections has been used as a treatment in previous virus outbreaks including SARS, avian influenza, and Ebola virus infection. [59] A clinical trial to determine the safety and efficacy of convalescent plasma in patients with COVID-19 has started in China; however, there is no data on its use as yet. [60]
Secondary prevention
Early recognition of new cases is the cornerstone of prevention of trans-mission. Immediately isolate all suspected and confirmed cases and implement recommended infection prevention and control procedures ac-cording to local protocols, including standard precautions at all times, and contact, droplet, and airborne precautions while the patient is symptomatic. [61] COVID-19 is a notifiable disease; report all suspected and confirmed cases to your local health authorities. [62]
Both primary and secondary prevention is an important aspect of combating this pandemic. Immediate recognition of cases and their contacts seems to be the only way to halt the pandemic from claiming more lives.
Discussion
Emerging novel viral infections have time and again proved to be a challenge to the health care systems worldwide. While earlier viruses have caused epidemics of varying proportions, SARS-CoV-2 has transformed into a pandemic affecting more than a 2lakh patients globally. Why this virus reached such huge proportion is a question that needs to be both asked and answered.
As the disease continues to unfold, perhaps, a comprehensive interdisciplinary effort is needed to combat and control the infection. Understanding of human, environmental and animal contributions to this pandemic need to be understood. Multidisciplinary teams with collaboration be-tween public health officers and veterinarians ought to be primarily organized. This must also include researchers, geneticists and phylogeneticists so that a better understanding of the genome of the virus is made and possibly research into targeted drug therapies and the development of a vaccine on large scale. Secondly, and importantly, proper laboratory equipment and diagnostic assays should be made available at the onset of the outbreak. Guidelines for testing and screening of cases and their contacts must be tailored specific to each country as one size cannot fit all. Every region has its own demographic differences and these must be taken into consideration while forming policies. Also, the role of bats as reservoirs and sources of infection needs to be further elaborated. Third, and perhaps, the most important point in a disease with such international implications is the isolation and quarantine of the travelers. Every country must have a check on the inflow of patients from affected regions and facilities for their proper quarantine and testing. Finally, it is a global concern and hence, international cooperation is a must if we have to combat the virus.
Conclusion
This article has discussed the various aspects of SARS-CoV-2 and its impact in the global scenario. We also stress the need of the hour is a comprehensive and quick first response to the pandemic to check its growth and to prevent spread into community. Education about preventive measures and isolation and self-quarantine will also help debunk the load over the medical system. Moreover, strengthening of the public health system both medical and paramedical is the need of the hour.
Competing Interests
The authors declare that they have no competing interests.
References
- Li F. Structure, function, and evolution of coronavirus spike proteins. Annu Rev Virol. 2016; 3: 237-261.
- Enjuanes L, Almazan F, Sola I, Zuniga S. Biochemical aspects of coronavirus replication and virus-host interaction. Annu Rev Microbi-ol. 2006; 60: 211-230.
- Perlman S, Netland J. Coronaviruses post-SARS: update on replication and pathogenesis. Nat Rev Microbiol. 2009; 7: 439-450
- Chen Y, Liu Q, Guo D. Emerging coronaviruses: Genome structure, replication, and pathogenesis. J. Med. Virol. 2020; 92: 418-423.
- Adams MJ, Carstens EB. Ratification vote on taxonomic pro-posals to the International Committee on Taxonomy of Viruses (2012). Arch Virol 2012; 157: 1411-1422.
- De Groot RJ, Baker SC, Baric RS, Brown CS, Drosten C, Enjuanes L, et al. Middle East Respiratory Syndrome Coronavirus (MERS-CoV): Announcement of the Coronavirus Study Group. J Virol 2013; 87: 7790-7792.
- WHO. Summary of probable SARS cases with onset of illness from 1 November 2002 to 31 July 2003. Dec 31, 2003.
- WHO. Middle East respiratory syndrome coronavirus (MERS-CoV). November, 2019.
- Lu R, Zhao X, Li J, Niu P, Yang B, Wu H, et al. Genomic characterization and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. The Lancet. 2020; 395: 565-574.
- Li W, Moore MJ, Vasilieva N, Sui J, Wong SK, Berne MA, et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003; 426: 450-454.
- Su S, Wong G, Shi W, Liu J, Lai AC, Zhou J, et al. Epidemiology, genetic recombination, and pathogenesis of coronaviruses. Trends in microbiology. 2016; 24: 490-502.
- Chan JF, Kok KH, Zhu Z, Chu H, To KK, Yuan S, et al. Genomic characterization of the 2019 novel human-pathogenic coronavirus isolated from a patient with atypical pneumonia after visiting Wuhan. Emerg Microbes Infect. 2020; 9: 221-236.
- Song W, Gui M, Wang X, Xiang Y. Cryo-EM structure of the SARS coronavirus spike glycoprotein in complex with its host cell receptor ACE2. PLoSPathog. 2018; 14: e1007236.
- Lei J, Kusov Y, Hilgenfeld R. Nsp3 of coronaviruses: Structures and functions of a large multi-domain protein. Antiviral Res. 2018; 149: 58-74.
- Angeletti S, Benvenuto D, Bianchi M, Giovanetti M, Pascarella S, Ciccozzi M. COVID-2019: The role of the nsp2 and nsp3 in its pathogenesis. J. Med. Virol. 2020.
- Jeffers SA, Tusell SM, Gillim-Ross L, Hemmila EM, Achenbach JE, Babcock GJ, et al. CD209L (L-SIGN) is a receptor for severe acute respiratory syndrome coronavirus. ProcNatlAcadSci USA. 2004; 101: 15748-15753.
- Zhou Y, Lu K, Pfefferle S, Bertram S, Glowacka I, Drosten C, et al. A single asparagine-linked glycosylation site of the severe acute respiratory syndrome coronavirus spike glycoprotein facilitates inhibition by mannose-binding lectin through multiple mecha-nisms. Journal of Virology. 2010; 84: 8753-8764
- Chen Y, Guo Y, Pan Y, Zhao ZJ. Structure analysis of the receptor binding of 2019-nCoV. Biochemical and biophysical research communications. 2020.
- Coutard B, Valle C, De Lamballerie X, Canard B, Seidah NG, Decroly E. The spike glycoprotein of the new coronavirus 2019-nCoV contains a furin-like cleavage site absent in CoV of the same clade. Antiviral research. 2020; 176: 104742.
- Zou L, Ruan F, Huang M, Liang L, Huang H, Hong Z, et al. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. New England Journal of Medicine. 2020; 382: 1177-1179.
- Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The Lancet. 2020; 395: 497-506.
- Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. The Lancet. 2020; 395: 507-513.
- Li Q, Guan X, Wu P, Wang X, Zhou L, Tong Y, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus–infected pneumonia. New England Journal of Medicine. 2020.
- Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19).
- Wu Z, Mc Googan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. Jama. 2020.
- Italian Ministry of Health. COVID-19 – Current situation in Italy. 2020.
- En24. Coronavirus, first infection in Italy: 38-year-old hospi-talised in Codogno, is serious. Quarantine for 70. 2020.
- COVID-19: on the front lines of coronavirus: the Italian response to COVID-19; B Marta paterlini, MJ 2020.
- Italy’s virus fatality rate higher than China’s. NHK World 2020 Mar 15.
- Case-Fatality Rate and Characteristics of Patients Dying in Relation to COVID-19 in Italy Graziano Onder, Giovanni Rezza.
- Novel Coronavirus Pneumonia Emergency Response Epidemiology Team. The epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (COVID-19) in China. Zhonghua Liu Xing Bing Xue ZaZhi. 2020; 41 (2): 145-15.
- Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. Jama. 2020; 323: 1061-1069.
- Chen ZM, Fu JF, Shu Q, Chen YH, Hua CZ, Li FB, et al. Diagnosis and treatment recommendations for pediatric respiratory infection caused by the 2019 novel coronavirus. World Journal of Pediatrics. 2020; 5: 1-7.
- Shen KL, Yang YH. Diagnosis and treatment of 2019 novel corona-virus infection in children: a pressing issue. World J Pediatr. 2020.
- Wang XF, Yuan J, Zheng YJ, Chen J, Bao YM, Wang YR, et al. Retracted: Clinical and epidemiological characteristics of 34 children with 2019 novel coronavirus infection in Shenzhen. Zhonghuaerkezazhi= Chinese Journal of Pediatrics. 2020; 58: E008.
- Ruan Q, Yang K, Wang W, Jiang L, Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med 2020.
- Chen H, Guo J, Wang C, Luo F, Yu X, Zhang W, et al. Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: A retrospective review of medical records. The Lancet. 2020; 395: 809-815.
- Zhu H, Wang L, Fang C, Peng S, Zhang L, Chang G, et al. Clinical analysis of 10 neonates born to mothers with 2019-nCoV pneumonia. Translational Pediatrics. 2020; 9: 51.
- Novel coronavirus pneumonia (COVID-19) CT distribution and sign features.
- Wu J, Feng CL, Xian XY, Qiang J, Zhang J, Mao QX, et al. World Health Organisation. Home care for patients with suspected novel coronavirus (COVID-19) infection presenting with mild symptoms, and management of their contacts. February 2020.
- Fardet L, Galicier L, Lambotte O, Marzac C, Aumont C, Chahwan D, et al. Development and validation of the HScore, a score for the diagnosis of reactive hemophagocytic syndrome. Arthritis & Rheumatology. 2014; 66: 2613-2620.
- World Health Organization. Corona virus disease (COVID-19) Advice for the public. 2020.
- Centers for Disease Control and Prevention. Corona virus Disease 2019 (COVID-19): prevention and treatment. February 2020.
- World Health Organization. Advice on the use of masks in the com-munity, during home care and in health care settings in the context of the novel coronavirus (2019-nCoV) outbreak. January 2020.
- Mahase E. China corona virus: what do we know so far? BMJ. 2020; 368: m308.
- Brooks SK, Webster RK, Smith LE, Woodland L, Wessely S, Greenberg N, et al. The psychological impact of quarantine and how to reduce it: rapid review of the evidence. The Lancet. 2020.
- World Health Organization. Clinical management of severe acute respiratory infection when novel coronavirus (nCoV) infection is suspected. 2020.
- Chinese Clinical Trial Registry. A randomized, open-label, blank-controlled trial forth efficacy and safety of lopinavir-ritonavir and inter-feron-alpha 2b in hospitalization patients with 2019-nCoV pneumonia (novel coronavirus pneumonia, NCP). 2020.
- Chinese Clinical Trial Registry. A randomised, open-label, multi-centre clinical trial evaluating and comparing the safety and efficiency of ASC09/ritonavir and lopinavir/ritonavir for confirmed cases of novel coronavirus pneumonia (COVID-19). 2020.
- Chinese Clinical Trial Registry. Clinical study for safety and efficacy of favipiravir in the treatment of novel coronavirus pneumonia (COVID-19). 2020.
- Chinese Clinical Trial Registry. Clinical study of arbidolhydrochlo-ride tablets in the treatment of novel coronavirus pneumonia (COVID-19). 2020.
- Clinical Trials.gov. Mild/ moderate 2019-nCoV remdesivir RCT. 2020.
- Clinical Trials.gov. Severe 2019 -n CoVremdesivir RCT. 2020.
- Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell research. 2020; 30: 269-271.
- Russell CD, Millar JE, Baillie JK. Clinical evidence does not support corticosteroid treatment for 2019-nCoV lung injury. Lancet 2020; 395: 473-475.
- Chinese Clinical Trial Registry. A multicenter, randomized controlled trial for the efficacy and safety of tocilizumab in the treatment of new coronavirus pneumonia (COVID-19). 2020.
- Clinical.trials.gov. Mesenchymal stem cell treatment for pneumonia patients infected with 2019 novel coronavirus. 2020.
- Gurwitz D. Angiotensin receptor blockers as tentative SARS?CoV?2 therapeutics. Drug Development Research. 2020.
- Chen L, Xiong J, Bao L, Shi Y. Convalescent plasma as a potential therapy for COVID-19. The Lancet Infectious Diseases. 2020; 20: 398-400.
- Clinical.trials.gov. Anti-SARS- CoV-2 inactivated convalescent plasma in the treatment of COVID-19. 2020.
- World Health Organization. Infection prevention and control during health care when novel coronavirus (nCoV) infection is suspected. 2020.
- Shi H, Han X, Jiang N, Cao Y, Alwalid O, Gu J, et al. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. The Lancet Infectious Diseases. 2020.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.