The Effect of FASD (Blood-letting) in the Treatment of Sciatica: A Randomized Clinical Trial Study
2 Department of Orthopedics, Shahed University, Tehran, Iran
3 Department of Biostatistics, Faculty of Medical Science, Tarbiat Modares University, Tehran, Iran
Citation: Amini MH, et al. The Effect of FASD (Bloodletting) in the Treatment of Sciatica: A Randomized Clinical Trial Study. Ann Med Health Sci Res. 2018;8:391-396
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Background: Sciatica is a common complication which is associated with disabilities, miss work, high therapeutic costs, and side effects of medications. In this study, we recommend an alternative treatment based on Persian medicine in the treatment of sciatica. We aim to consider the effect of FASD (Blood-letting), which has been suggested as an effective therapeutic method for articular and sciatica pains in Persian medicine, in the treatment of sciatica. Material and Methods: In this randomized clinical trial study, the therapeutic effect of FASD was considered on 50 patients with sciatica in Tehran within the year 2017. Patients were randomly divided into two groups: diclofenac (50- 100 mg based on their body weight, n=25) and diclofenac (50-100 mg based on their body weight + FASD, n=25). Results: In this survey, 76% of patients were male and 24% were female. There was no significant difference in mean of basic demographic data between two groups. Patients who received combined therapy with diclofenac + FASD showed better scores of pain (45.43 vs. 3.6; p<0.001), mental health (17.4 vs. 2.04; p<0.001), quality of life (32.79 vs. 4.29; p<0.001) and Roland Morris score (13.92 vs. 2.28, p<0.001) compared to the diclofenac group. Conclusion: According to the traditional medicine and findings of the current study, FASD can be considered as an effective method for the treatment of articular pains and sciatica.
Keywords
Arthralgia; Blood-letting; Low back pain; Sciatica; Traditional medicine
Introduction
Low back pains are one of the most common causes of disabilities in people under 45 years old which can be correlated to different factors such as lifestyle, nutritional type, inappropriate social behaviors, and occupational habits. [1] The most common reason people go to the doctor is a musculoskeletal disease (MSD), in which back pains and sciatica are most popular. [2] Direct costs of musculoskeletal disabilities treatment and miss work in the USA are estimated billions of dollars each year, in which $100 billion is related to back pain. Statistical analyzes revealed that back pains and sciatica were the most frequent reasons (approximately 62%) that prohibited people go to their workplace in 2006. [3] Therapeutic methods for back pains and sciatica in modern medicine include physiotherapy, exercise training programs, sedative drugs such as steroidal and nonsteroidal anti-inflammatory drugs (NSAIDs), as well as muscle relaxants, anti-seizure drugs, and anti-depression drugs. Eventually, surgery is recommended in case there is no response after drug treatments; however, it reduces pain only in a small number of patients. Previous studies reported that more than 50% of patients who treated with surgery may suffer from sensual and physical disorders. [4] Furthermore, drug use can be associated with different side effects such as gastrointestinal bleeding, heart and brain attacks, hepatic and renal poisoning, as well as dermal, pulmonary, and bloody complications. [5-10] Due to the high prevalence of sciatica, high therapeutic costs and side effects of current medications, other therapeutic methods with lower side effects and costs should be provided. Therefore, we aim to consider the therapeutic effect of FASD, which has been suggested as an effective method for articular pain in Persian medicine, in the treatment of sciatica.
Materials and Methods
In this randomized clinical trial study, the therapeutic effect of FASD (Blood-letting) was considered on 50 patients with sciatica in Tehran- Iran within the year 2017. Participants were patients who referred to traditional medicine healthcare at Shahed University and orthopedic clinic at Shahid Mostafa Khomeini in Tehran. Sciatica was diagnosed according to SLR test. [11]
Inclusion criteria
• Willingness to participate in study,
• Ages 20-60 years old, (Body Mass Index) BMI 18-35,
• A constant weight (a change lower than 6 Kg within the last 6 months),
• Patients who need non-surgical therapeutic methods for sciatica,
• No history of spinal discs surgery and severe sensory and physical symptoms,
• No history of fracture or surgery within the last 3 months,
• No history of inflammation in joints such as rheumatoid arthritis, rheumatic fever, and ankylosing spondylitis,
• No history of back pains caused by infection, rheumatism, glandular, metabolic, malignancies, blood diseases, coagulation diseases, and pelvic deviation
• No history of other spinal diseases such as scoliosis, kyphotic, and spondylolisthesis,
• No history of severe anemia,
• No pregnancy and breastfeeding at the time of study,
• No history of neurology and psychiatry problems,
• No medical problem or allergy to diclofenac, and
• No treatment with anticoagulants at the time of study.
Exclusion criteria
• An unwillingness to participate in the study,
• Being pregnant at the time of the study,
• Having psychological problems,
• Increasing the severity of pain and
• Patient’s disabilities.
In this survey, patients who referred to each of clinical center in Tehran, from June to November 2017 with the complication of back pains or sciatica for at least 4 weeks, were selected for further considerations. After preliminary considerations and inclusion criteria, 50 patients were selected for this survey. Prior to sample collection, all patients signed an informed consent form and filled the questionnaire about demographic characteristics, drug registration form, Roland Morris questionnaire, as well as visual analog scale (VAS) and SF-36 quality of life questionnaires.
Patients were then randomly divided into two groups including treatment with diclofenac (Alborz Darou Pharmaceutical Co) (n=25) and treatment with diclofenac + FASD (n=25). The first group had received diclofenac (50-100 mg/2 times/day based on their body weight) for a period of 7-10 days. Patients in diclofenac + FASD group not only received diclofenac (50-100 mg/2 times/day based on their body weight) for a period of 7-10 days but also they have undergone a Basilic vein FASD by an expert person. The FASD of the dominant side was performed in cases with bilateral sciatica, but a right Basilic vein FASD was done when the pain was the same on both sides.
Incisions (1-2 mm) were made with a bistoury-blade (no. 11) and 100- 150 cc blood was extracted from each patient. All subjects were asked to record their daily diclofenac using in a drug registration form and then deliver it to our researcher. Pain scores were recorded using the VAS questionnaire before the intervention, one day, three days, 10 days, and 4 weeks after the intervention. The amount of diclofenac intake was recorded 30 days after the intervention. Disability score was calculated using Ronald-Morris questionnaire before and 30 days after the intervention. The quality of life score (SF36) was considered before and 30 days after the intervention. The Ethics Review Board of the Shahed University (IR Shahed REC) Tehran, Iran approved the study. This study was also registered and approved in the Iranian Registry of Clinical Trials (IRCT20180131038575N1).
In this survey, pain score was measured using the VAS questionnaire which includes a horizontal straight line with 100 mm in length anchored by 2 verbal descriptors, “no pain” and “marked pain”. Patients are asked to place a line perpendicular to the VAS line at the point that represents their pain intensity. Using a ruler, the score is determined by measuring the distance (mm) on the 100 mm line between the “no pain” anchor and the patient’s mark, providing a range of scores from 0–100. [12,13] A higher score indicates greater pain intensity.
Roland-Morris Disability Questionnaire (RDQ) was used to measure disability score. It consists of 24 statements relating to the person’s perceptions of their back pain and associated disability. RDQ score can range from 0 (no disability) to 24 (maximal disability). Suitable validity and reliability of this questionnaire has been confirmed in the both internal [14] and external [15] studies.
Quality of life was assessed using the quality of life SF-36 questionnaire. This includes a self-report tool with 36 and 8 subscales [physical function (PF), role impairment due to physical health (RP), role impairment due to emotional health (RE), energy/ fatigue (E/F), emotional well-being (EW), social function (SF), and general health (GH). In this questionnaire, a lower score indicates a lower quality of life. This questionnaire has been confirmed in the both internal [16] and external [17] studies.
Statistical analysis
In this study, descriptive analysis (means ± SD, correlation, frequency and ratio) and inferential analysis (chi-square test, independent t-test, Mann-Whitney test, depended t-test, correlation test, frequent schedule test, McNemar’s test, and Friedman test) and Kolmogorov–Smirnov test (for normality and non-normality distribution test) were used. Data were analyzed using SPSS, version 21. A probability of less than 0.05 was considered significant. A total of 21 samples were calculated for each group using the formula, in which α=0.05, β=0.1 and d=1. Since some subjects who enroll in the study may drop out, we enrolled 25% more subjects (6 samples for each group) that the sample size calculation called for. Eventually, 27 samples were entered into each group.
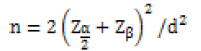
Results
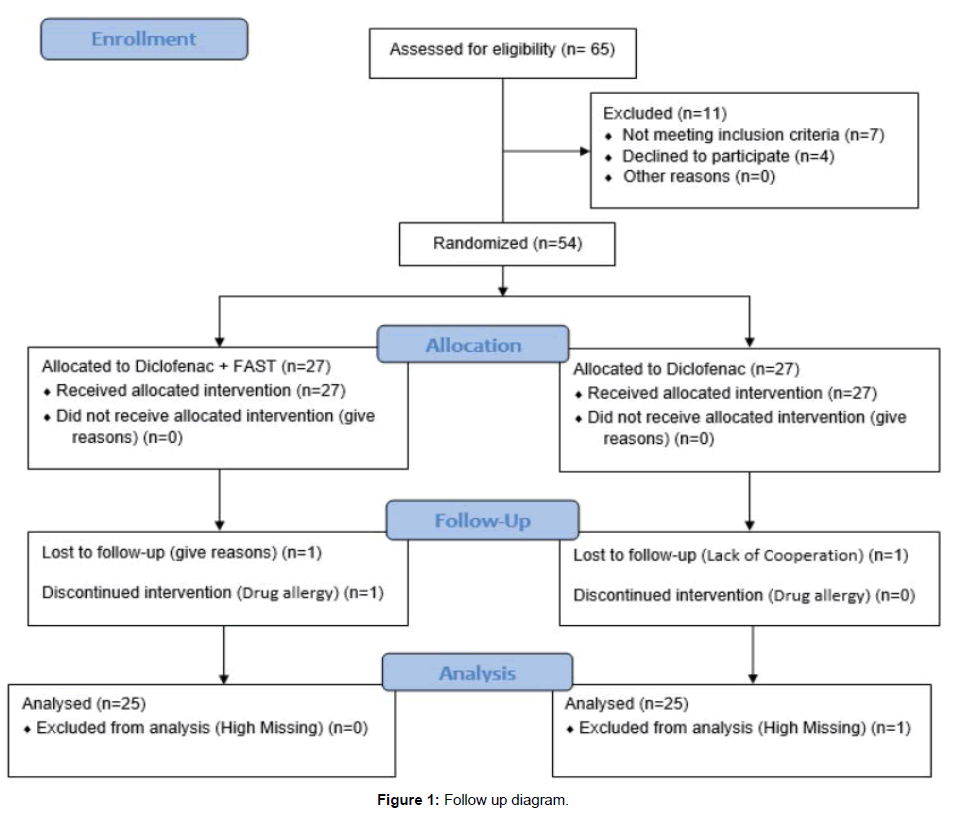
A total of 54 patients were entered into the survey in which 4 subjects were unable to continue the study (Figure 1). The mean ± SD of demographic data for each group can be seen in Table 1. There was no significant difference in mean of age, sex frequency, educational background, and weight between two groups. The mean ± SD of pain score, disabilities, amount of diclofenac consumed by patients in each group, quality of life score and its sub-scales before intervention are shown in Table 2. There was no significant difference in mean values of these baseline parameters between two groups. Table 3 shows that FASD has a significant effect on these parameters by increasing the quality of life and decreasing the disabilities and contents of consumed diclofenac, while diclofenac consumption alone had no significant effect on these parameters. Table 4 compares the mean of pain scores between two groups. The mean of pain scores in FASD group were significantly decreased compared to the diclofenac group.
| Categorical variable | Group | p-value | ||||
|---|---|---|---|---|---|---|
| Diclofenac + FASD | Diclofenac | |||||
| N | % | N | % | |||
| Gender | Male | 19 | 76.0% | 19 | 76.0% | 0.629 |
| Female | 6 | 24.0% | 6 | 24.0% | ||
| Education | Under Diploma | 9 | 37.5% | 8 | 34.8% | 0.743 |
| Diploma | 8 | 33.3% | 10 | 43.5% | ||
| Upper Diploma | 7 | 29.2% | 5 | 21.7% | ||
| Continues variable | Mean | SD | Mean | SD | ||
| Age | Year | 37.82 | 10.69 | 42.88 | 11.11 | 0.114 |
| BMI | kg/m2 | 25.98 | 3.76 | 26.18 | 3.05 | 0.420 |
The P-value base on Chi-Square test, T-test or Mann-Whitney U test; SD: Standard Deviation
Table 1: The frequency and percent of gender, education and mean and SD of age and BMI according to the two the groups.
| Group | p-value | ||||
|---|---|---|---|---|---|
| Diclofenac + FASD | Diclofenac | ||||
| Mean | SD | Mean | SD | ||
| Physical functioning | 32.15 | 33.69 | 32.00 | 26.46 | 0.717 |
| Role limitations due to physical health | 18.75 | 29.72 | 13.00 | 24.07 | 0.570 |
| Role limitations due to emotional problems | 40.78 | 43.73 | 45.33 | 42.90 | 0.811 |
| Energy/ fatigue | 51.93 | 20.62 | 51.00 | 17.26 | 0.953 |
| Emotional well being | 62.76 | 17.18 | 61.28 | 16.15 | 0.815 |
| Social functioning | 39.74 | 26.24 | 49.50 | 27.36 | 0.195 |
| Pain | 24.90 | 20.85 | 29.40 | 26.33 | 0.595 |
| General health | 52.73 | 25.08 | 47.87 | 14.60 | 0.284 |
| TOTAL Quality of Life | 40.45 | 20.50 | 40.40 | 15.79 | 0.915 |
| VAS (Pain) | 8.76 | 1.45 | 9.16 | .90 | 0.509 |
| Roland Morris (Disability) | 16.44 | 7.32 | 18.40 | 6.11 | 0.249 |
SD: Standard Deviation; p-value base on T-test or Mann-Whitney U test
Table 2: The base line variable of study variables according to the two groups.
| Pre | Post | Diff | p-value 1 | p-value 2 | ||||
|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | |||||
| Physical functioning | Diclofenac + FASD | 32.15 | 33.69 | 81.33 | 23.33 | 49.18 | <0.001 | <0.001 |
| Diclofenac | 32.00 | 26.46 | 37.60 | 28.21 | 5.6 | 0.460 | ||
| Role limitations due to physical health | Diclofenac + FASD | 18.75 | 29.72 | 63.67 | 40.16 | 44.92 | <0.001 | <0.001 |
| Diclofenac | 13.00 | 24.07 | 20.00 | 26.02 | 7 | 0.314 | ||
| Role limitations due to emotional problems | Diclofenac + FASD | 40.78 | 43.73 | 75.76 | 40.08 | 34.98 | 0.004 | 0.048 |
| Diclofenac | 45.33 | 42.90 | 54.67 | 40.69 | 9.34 | 0.421 | ||
| Energy/ Fatigue | Diclofenac + FASD | 51.93 | 20.62 | 68.62 | 16.96 | 16.69 | 0.002 | 0.001 |
| Diclofenac | 51.00 | 17.26 | 52.80 | 17.45 | 1.8 | 0.708 | ||
| Emotional well being | Diclofenac + FASD | 62.76 | 17.18 | 75.52 | 10.36 | 12.76 | 0.001 | 0.001 |
| Diclofenac | 61.28 | 16.15 | 62.88 | 16.91 | 1.6 | 0.721 | ||
| Social functioning | Diclofenac + FASD | 39.74 | 26.24 | 64.39 | 20.76 | 24.65 | <0.001 | <0.001 |
| Diclofenac | 49.50 | 27.36 | 52.50 | 27.24 | 3 | 0.719 | ||
| Pain Quality of life | Diclofenac + FASD | 24.90 | 20.85 | 70.33 | 24.87 | 45.43 | 0.006 | <0.001 |
| Diclofenac | 29.40 | 26.33 | 33.00 | 25.87 | 3.6 | 0.609 | ||
| General Health | Diclofenac + FASD | 52.73 | 25.08 | 70.13 | 20.34 | 17.4 | <0.001 | <0.001 |
| Diclofenac | 47.87 | 14.60 | 49.91 | 14.32 | 2.04 | 0.356 | ||
| TOTAL Quality of Life | Diclofenac + FASD | 40.45 | 20.50 | 73.24 | 18.41 | 32.79 | <0.001 | <0.001 |
| Diclofenac | 40.40 | 15.79 | 44.69 | 17.75 | 4.29 | 0.356 | ||
| Roland Morris | Diclofenac + FASD | 16.44 | 7.32 | 2.52 | 3.99 | -13.92 | <0.001 | <0.001 |
| Diclofenac | 18.40 | 6.11 | 16.12 | 5.99 | -2.28 | 0.174 | ||
| Number of used Diclofenac pill | Diclofenac + FASD | ------- | ------- | 6.46 | 7.66 | ------- | ------- | <0.001 |
| Diclofenac | ------- | -------- | 52.80 | 13.77 | ------- | -------- | ||
SD: Standard Deviation; p-value 1: base on Paired Sample t-test or Wilcoxon Signed Ranks Test. p-value 2: base on T-test or Mann-Whitney U test
Table 3: The mean and SD of variable before and after of study
| Group | p-value1 | ||||
|---|---|---|---|---|---|
| Diclofenac + FASD | Diclofenac | ||||
| Mean | SD | Mean | SD | ||
| Pain Before study | 8.76 | 1.45 | 9.16 | .90 | 0.509 |
| Pain after one day after treatment | 4.40 | 2.22 | 8.84 | 1.14 | <0.001 |
| Pain after three day after treatment | 4.12 | 2.65 | 8.00 | 1.38 | <0.001 |
| Pain after ten day after treatment | 3.65 | 2.87 | 8.20 | 1.41 | <0.001 |
| Pain after 4 weeks after treatment | 2.04 | 2.64 | 8.16 | 1.77 | <0.001 |
| P-value | <0.001 | <0.001 | |||
SD: Standard Deviation; p-value 1: base on T-test or Mann-Whitney U test; P-value2: base on Friedman Test or repeated measurement ANOVA.
Table 4: The pain during time according to the two groups.
Discussion
The definition of joints pain in the viewpoint of Iranian medicine is the presence of pain in the body corpses, including a kyphosis and scoliosis backache, pelvic pain, arthralgia, gout, hip pain, sciatic pain, varices, elephantiasis, and pain in the heel plantar pain. The important factors that cause articular disorders include effective, impressive and underlie factors. [18-20] Articulate pains treatment in viewpoint of traditional medicine, includes the elimination of diseasecausing factor, excretion of domination humorous, temperament correction and strengthen the body organ. FASD is considered the only or partial therapeutic method for treatment of these abnormalities. [8-14] For this reason, the present study considered the effect of FASD, which has been suggested as an effective therapeutic method for articular pain and sciatica in Persian medicine, in the treatment of sciatica. Our data have revealed that FASD improved the quality of life, pain scores, disabilities and the amounts of diclofenac consumption in patients who treated with this method. Our findings are in agreement with the results obtained from previous studies that confirmed the positive effect of FASD on musculoskeletal disease. [21-25] Previous studies also showed the positive effect of wet-cupping and cupping therapy on back pains, headache, muscular pains, and carpal tunnel syndrome. [26-28] In another research, drug consumption caused bone marrow suppression and reduced number of WBC in patients with rheumatoid arthritis, while combined therapy with drugs and wet-cupping improved WBC number and interleukin receptors. [29] The positive effect of FASD and wet-cupping was also reported in the treatment of musculoskeletal disease, sciatica, and non-specific pains of back, neck and arms. [30-35]
In the viewpoint of Persian medicine, articular diseases and sciatica pain are caused by several factors including:
• Effective factors such as simple miss-temperaments (e.g. warm, cold and dry miss-temperaments) and physical miss-temperaments (e.g. four fundamental bodily humors: blood, yellow bile, black bile, and phlegm, and also infection, steam and wind produced of them) and weather conditions.
• Impressive factors such as an intrinsic and active characteristic of the body member.
• Underlie factors such as frequent colds, sinusitis or high accumulation of sputum back of the throat, in-appropriate nutritional style, use of foods that increase abnormal humor production, especially in patients have gastrointestinal and digest problems.
Additionally, bathing, exercise training and sexual activity after eating are the other significant factors. Moreover, drinking during starvation, in the bathroom and just after bathing, during or after exercise, during or after sleep, and after sexual intercourse are important factors. Break the habit of FASD, wet-cupping, sticking, diarrhea, menstrual bleeding, in which individuals excrete poisoning metabolites from the body can be considered as the other factors. Sports leave or over-exercises are also important factors. High sexual activity, especially after eating, sleepless or sleep for long times are the other significant factors. Some sever or chronic diseases, aging, spiritual and mental problems and genetic backgrounds are also significant factors. [18-20]
Since the above-mentioned factors cause accumulation of secondary metabolites in body organs, FASD can excrete these metabolites from the body and improve articular pains and sciatica.
Conclusion
The results of this study showed, FASD can be considered as an effective method for the treatment of articular pains and sciatica.
Conflict of Interest
The authors disclose that they have no conflicts of interest.
REFERENCES
- Stafford M, Peng P, Hill D. Sciatica: A review of history, epidemiology, pathogenesis, and the role of epidural steroid injection in management. British Journal of Anaesthesia. 2007;99:461-473.
- HeliÖvaara M, MÄkelÄ M, Knekt P, Impivaara O, Aromaa A. Determinants of sciatica and low-back pain. Spine. 1991;16:608-614.
- Cannale S. CAMPBLLs operative orthopedics 11th edn 2008:869-914.
- Deyo RA, Mirza SK, Turner JA, Martin BI. Overtreating chronic back pain: time to back off? The Journal of the American Board of Family Medicine. 2009;22:62-68.
- Bjarnason I, Hayllar J. Side effects of nonsteroidal anti-inflammatory drugs on the small and large intestine in humans. Gastroenterology. 1993;104:1832-1847.
- Shim YK, Kim N. Nonsteroidal anti-inflammatory drug and aspirin-induced peptic ulcer disease. The Korean Journal of Gastroenterology. 2016;67:300-312.
- Hoppmann RA, Peden JG, Ober SK. Central nervous system side effects of non-steroidal anti-inflammatory drugs. Aseptic meningitis, psychosis, and cognitive dysfunction. Archives of Internal Medicine. 1991;151:1309-1313.
- Small SP. Preventing sciatic nerve injury from intramuscular injections: literature review. Journal of Advanced Nursing. 2004;47:287-296.
- Singh G. Recent considerations in nonsteroidal anti-inflammatory drug gastropathy. The American Journal of Medicine. 1998;105:31s-38s.
- Whelton A. Renal and related cardiovascular effects of conventional and COX-2-specific NSAIDs and non-NSAID analgesics. American Journal of Therapeutics. 2000;7:63-74.
- Cattley P, Winyard J, Trevaskis J, Eaton S. Validity and reliability of clinical tests for the sacroiliac joint: A review of literature. Australasian Chiropractic & Osteopathy. 2002;10:73.
- Price DD, McGrath PA, Rafii A, Buckingham B. The validation of visual analogue scales as ratio scale measures for chronic and experimental pain. Pain. 1983;17:45-56.
- Ogon M, Krismer M, Söllner W, Kantner-Rumplmair W, Lampe A. Chronic low back pain measurement with visual analogue scales in different settings. Pain. 1996;64:425-428.
- Grotle M, Brox JI, Vøllestad NK. Functional status and disability questionnaires: what do they assess?: a systematic review of back-specific outcome questionnaires. Spine. 2005;30:130-140.
- Asghari A, Golak N. The roles of pain coping strategies in adjustment to chronic pain. 2005.
- Ware JE. SF-36 health survey: manual and interpretation guide. Health Institute. 1993.
- Montazeri A, Goshtasebi A, Vahdaninia M, Gandek B. The Short Form Health Survey (SF-36): translation and validation study of the Iranian version. Qual Life Res. 2005;14:875-882.
- Avicenna. Qanun fi al Tib [Canon of Medicine]. Beiruot: Ehyaol Toras al-Arabi Press; 2010.
- Arzani M. Teb-e-Akbari [Akbari's Medicine] Tehran: Jalaleddin Press; 2008.
- Azam Khan M. Exir Azam Tehran: Institute of Medical History, Islamic Medicine and Complementary Medicine; 2008.
- Lauche R, Cramer H, Hohmann C, Choi K-E, Rampp T, Saha FJ, et al. The effect of traditional cupping on pain and mechanical thresholds in patients with chronic nonspecific neck pain: a randomised controlled pilot study. Evidence-based Complementary and Alternative Medicine. 2012;2012.
- Chang X, Feng Y, Yi S, Zeng X, Lin Y. Effects of pricking bleeding at "Weizhong" (BL40) on sciatic nerve conduction velocity and interleukin-1alpha content in the lumbar vertebral nucleus pulposus in experimental lumbar intervertebral disc protrusion rabbits. Zhen ci yan jiu= Acupuncture research/ [Zhongguo yi xue ke xue yuan Yi xue qing bao yan jiu suo bian ji]. 2007;32:319-322.
- Jiang G, Jia C, Lin M. Effect of bloodletting therapy at local myofascial trigger points and acupuncture at Jiaji (EX-B 2) points on upper back myofascial pain syndrome: a randomized controlled trial. J Tradit Chin Med. 2016;36:26-31.
- Sun H, Wan H, Zhang L, He X. Clinical observation of blood-letting to reduce pressure plus electroacupuncture for acute scapulohumeral periarthritis. Zhongguo Zhen Jiu Chinese acupuncture & moxibustion. 2016;36:933-937.
- Harty LC, Lai D, Connor S, Dunne A, Ali M, Ryan J, et al. Prevalence and progress of joint symptoms in hereditary hemochromatosis and symptomatic response to venesection. JCR: Journal of Clinical Rheumatology. 2011;17:220-222.
- Michalsen A, Bock S, Lüdtke R, Rampp T, Baecker M, Bachmann J, et al. Effects of traditional cupping therapy in patients with carpal tunnel syndrome: A randomized controlled trial. The Journal of Pain. 2009;10:601-608.
- Dal Kwon Y, Cho HJ. Systematic review of cupping including bloodletting therapy for musculoskeletal diseases in Korea. Korean J Oriental Physiology & Pathology 2l. 2007;3:I789-1793.
- Laine C, Horton R, DeAngelis CD, Drazen JM, Frizelle FA, Godlee F, et al. Clinical trial registration—looking back and moving ahead. Mass Medical Soc; 2007.
- Ahmed SM, Madbouly NH, Maklad SS, Abu-Shady EA. Immunomodulatory effects of blood letting cupping therapy in patients with rheumatoid arthritis. The Egyptian Journal of Immunology. 2004;12:39-51.
- Al-Bedah AM, Khalil MK, Posadzki P, Sohaibani I, Aboushanab TS, Al-Qaed M, et al. Evaluation of wet cupping therapy: systematic review of randomized clinical trials. The Journal of Alternative and Complementary Medicine. 2016;22:768-777.
- Zhu Y, Yu H, Pan Y, Yang J, Wu B, Hu X, et al. Clinical observation of pricking blood combined with moxibustion for acute gouty arthritis. Zhongguo Zhen Jiu Chinese Acupuncture & Moxibustion. 2015;35:885-888.
- Zhang SJ, Liu JP, He KQ. Treatment of acute gouty arthritis by blood-letting cupping plus herbal medicine. Journal of Traditional Chinese Medicine. 2010;30:18-20.
- Tian Y, Wang L, Wang X, Sun W. Clinical observation on ankylosing spondylitis treated with acupoint catgut embedding combined vessel pricking therapy. Zhongguo Zhen Jiu Chinese Acupuncture & Moxibustion. 2011;31:601-604.
- Arslan M, Gökgöz N, Dane S. The effect of traditional wet cupping on shoulder pain and neck pain: A pilot study. Complementary Therapies in Clinical Practice. 2016;23:30-33.
- Zeng H, Nie B, Shi L. Observation on therapeutic effect of blood-letting puncture combined with red-hot needle therapy on knee osteoarthritis. Zhongguo Zhen Jiu Chinese Acupuncture & Moxibustion. 2008;28:493-495.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.