The Low-birth weight Infants: Pattern of Morbidity and Mortality in a Tertiary Healthcare Facility in the South Eastern Nigeria
2 Department of Paediatrics, Enugu State University of Science and Technology, Enugu State, Nigeria
3 Griffiths University Medical School, Gold Coast, Australia
Citation: Chidiebere ODI, et al. The Low-birth weight Infants: Pattern of Morbidity and Mortality in a Tertiary Healthcare Facility in the South Eastern Nigeria. Ann Med Health Sci Res. 2018;8:4-10
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Background: Low birth-weight in infants is further categorized as Extreme low-birth weight (ELBW), Very low-birth weight (VLBW) and Low-birth weight (LBW) when weight at birth is <1 kg, ≥ 1 kg to <1.5 kg and ≥ 1.5 kg to <2.5 kg respectively. It is not clear how these various categories predict co-morbidities, duration of hospital stay and survival in affected newborns. Identifying such relationships will help prioritization of care, and this study is designed to explore these. Methods: This is a prospective study conducted over a 45 months period at the neonatal intensive care unit (NICU) of the Enugu State University Teaching Hospital (ESUTH), Enugu, Nigeria. Results: A total of 167 LBW newborns were enrolled, with about 90% born preterm. ELBWs constituted 20 (12.0%), while 47 (28.1%) and 100 (59.9%) were VLBW and LBW respectively. About 50 (32%) of the babies did not survive. Of these number, 16/20 (80%) were ELBW babies, 19/47 (41%) were VLBW while 15/91 (17%) were LBW babies, (P=0.001). Survival analysis showed that ELBW HR 20.27; P=0.001] and VLBW [HR 3.57; P=0.002] babies were respectively about 20 and 4 times more likely to die relative to the LBW group. Recurrent apnoea and the need for respiratory support were significantly more common among ELBWs and VLBWs relative to LBWs (P=0.001), while the LBW babies developed less hypothermia but more hypoglycemia (P=0.001). No significant differences were observed in the incidences of other co-morbidities. Conclusion: We conclude that low mean weights in infants are associated with higher fatal outcomes and prolonged hospital stays as well as a number of co-morbidities.
Keywords
Newborns; Low birth weight; Morbidity; Mortality; Enugu
Introduction
Low birth weight (LBW) has been defined by the World Health Organization (WHO) as weight at birth of less than 2.5 kg (5.5 pounds). In 2002, the World Health Organization adopted ‘A World Fit for Children’ declaration which aimed at reducing the global incidence of low-birth weight by one-third. [1] This declaration was essential because it is estimated that 30 million babies are born annually with low-birth-weight with close to 70 percent of these births occurring in developing countries. Lowbirth weight is a major determinant of mortality, morbidity and disability in infancy and childhood with long-term impact on health outcomes in adult life. [2] Low birth weight usually results when delivery occurs before 37 completed weeks of gestation or due to poor fetal growth in-utero or combination of both. The risk factors that are related to these causes include maternal factors such as poor nutrition, young age, disease conditions etc.; fetal factors such as multiple gestation, congenital malformation, fetal diseases etc., and environmental factors such as smoking, substance abuse, and other pollutants. [2] Due to sub-optimal development, newborns with LBW develop co-morbidities such as hypothermia, electrolyte imbalances, respiratory distress, increased susceptibility to infection resulting to longer hospital stay and increased mortality. Based on epidemiological data, newborns with low-birth weight were 20 times more likely to die than those with birth weight 2.5 kg or more. [3] The consequences of poor health status and inadequate nutritional intake for women during pregnancy not only directly affects women’s health status, but may also have a negative impact on birth weight and early development. [3] The result is a huge economic cost to the health sector that imposes a significant burden on society as a whole. Although the global prevalence of low-birth weight is on the decline, it is still high in many developing countries. In Nigeria for instance, 5-6 million LBW infants are born annually with approximately 100, 000 ending in fatality. [4] Several studies have assessed the socioeconomic factors associated with low birth weight in Nigeria. [5,6] However, none to our knowledge have specifically looked into the various categories of low-birth weight babies further classified as extreme low birth weight (ELBW<1 kg), very low birth-weight (VLBW ≥ 1 kg to <1.5 kg) and low birth-weight (LBW ≥1.5 kg to<2.5 kg). This study investigated how these different categories of low birth-weight predict co-morbidities, duration of hospital stay and survival in LBW babies. This, it is believed, would help in anticipation of co-morbidities and improved management of these vulnerable newborns.
Methodology
Study area and site
This was a prospective study carried out at the Neonatal Intensive Care Unit (NICU) of Enugu State University Teaching Hospital (ESUTH), Parklane. The site is located within Enugu, the capital city of Enugu State, and South-East Nigeria. ESUTH is a tertiary health institution that offers specialized medical services and serves as a referral center to Private, General, Mission hospital and other delivery homes within Enugu and neighboring State. The NICU offers 24 hour services to sick babies born within and outside the hospital and is located in close proximity to the labor ward with an average delivery rate of 144 per month. The NICU is equipped with 4 functional incubators, an open incubator (with radiant warmer) and facilities for Kangaroo Mother Care (KMC). The unit also has a newborn infant ventilator (SC-Y 200 infant ventilator), 6 improvised bubble CPAP devices, a unit of multi-parameter monitor, a unit of hand held pulse oximeter. Other medications and equipment’s available in the unit include caffeine citrate injections, surfactant and oxygen delivery units, several units of Bag-mask-device, manual/electrical suction machines etc. The NICU is manned on rotational basis by 2 consultant neonatologists, 5 resident doctors undergoing rotation in the SCBU, 19 nurses three of which are specialized pediatric nurses.
Newborn participation and enrolment
The study was carried out over a period of 3years and 9 months (January 2013-October 2016). Term and preterm newborn babies of consenting mothers delivered in ESUTH (In-Born) or referred to ESUTH (Out-Born) that had weights less than 2.5 kg were consecutively enrolled and followed up with daily reviews till discharge or death. Newborn babies of mothers with severe medical conditions such as HIV, sickle cell disease and cancer and those with multiple gestations were excluded from the study.
The diagnosis of low-birth weight was based on two measurements using two different weighing scales by two different observers. The weighing scales were re-calibrated before each measurement to ensure accuracy. The concordance rate between the two readings was over 95% in all measurements. For the few cases where there was discrepancy of ± 0.1 kg grams or more between measurements, a third reading was done and where the differences persists, an average between all readings were taken.
Diagnoses of co-morbidities were done based on clinical history, physical examination and laboratory findings where necessary. For this study, these diagnoses were made by a neonatologist not involved in this study. A second neonatologist unaware of the initial diagnosis was also brought in to confirm or refute the diagnosis in all cases. In rare cases, a third specialist is contacted to resolve disparity in diagnosis. For the sake of this study the common co-morbidities encountered in LBW infants were defined as follows;
• Apnea defined as cessation of breathing lasting for 20 seconds or shorter if associated with bradycardia or cyanosis;
• Asphyxia defined as APGAR score at 5th minute less than 7;
• Malaria infection defined as presence of malaria parasite in the blood film report;
• Anemia defined as Hemoglobin level <10 g/dl;
• Hypoglycemia defined as Random blood glucose of <40 mg/dl;
• Hypothermia defined as rectal temperature of <35°C;
• Neonatal sepsis defined as clinical signs and symptoms suggestive of probable sepsis with positive culture results.
• Diagnosis of some co-morbidities such as neonatal jaundice (yellowish discoloration of the sclera and or skin), congenital structural anomalies and Necrotizing Enterocolitis were done clinically.
Measures
Weighing of newborns was done in the labor ward and/or on admission to the NICU for those referred within 24 hours of birth. After skin to skin contact has been established with the mother and drying done, the scale is calibrated to zero. The babies were gently placed on the scales and read off to the nearest 0.01 kg. The weighing of the newborn babies was done by a trained nurses or residents doctors. An electronic infant weighing scale (Kin-lee electronic infant weighing scale, model= EBSL-20 with maximum capacity= 20 kg and minimum of 100 g) in the labor ward was initially used with all readings to the nearest 0.01 kg. The batteries of the scales were checked on alternate days and replaced when necessary to ensure accurate measurement. For the purpose of this study, a second weight measurement was done with a manual weighing scale in NICU (Manual pediatric scale, model= ATZ-10 with maximum capacity= 10 kg minimum capacity= 500 g) and read off by a different reader.
For babies delivered outside ESUTH, the birth weight reported on the referral letter was used with a second done in the SCBU. For those without referrals, permission was requested from consenting mothers to call the hospital where delivery was done. In cases especially from traditional birth homes where no weight was taken, weight was done in the hospital for newborn babies less than 72 hours.
Data entry and analysis
The above measures were documented at presentation in the relevant sections of the questionnaire and subsequently transferred into a Microsoft Excel Sheet. Distribution of the measures of outcome and predictor variables were analyzed and recorded in percentages. Grossly missing data were excluded from the analysis. The Chi-square and logistic regression statistical tools were used to assess variables significantly associated with morbidity and mortality in newborn with lowbirth weight. Data was analyzed using IBM® SPSS version 18.0 (SPSS Inc, Chicago, IL). Statistical significance was set at p<0.05.
Ethical considerations
Ethical clearance was obtained from the Enugu State University Teaching Hospital Ethics Committee. Prior to recruitment of each subject, informed consent was obtained from every mother and newborn pair in their own right. Participation in the study was entirely voluntary and no financial inducement whatsoever was involved. Participants were informed that voluntary withdrawal at any stage of interaction was guaranteed without any adverse effect to themselves or their babies. All information was handled with strict confidentiality.
Results
Description of low-birth weight newborns enrolled in the study
This study enrolled one hundred and sixty-seven (167) low birth weight newborns. Approximately 9-in-10 of the newborns was preterm i.e., delivered before 37 completed weeks while the remaining 10% were term deliveries. There were roughly equal proportion of male and female newborns enrolled with three out of five (≈ 60%) of these babies delivered via spontaneous vaginal delivery. One hundred and forty one mothers (88%) of the newborns attended antenatal care during the pregnancy of the index child and 57% were delivered within Enugu State University Teaching Hospital (ESUTH). About a third of the newborns, 51 (34%) had weights appropriate for gestational age while the remaining, two-thirds were small for gestational age [Table 1].
| Variables | Frequency | Weight categories | P | Variables | Frequency | Weight categories | P | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| ELBW | VLBW | LBW | ELBW | VLBW | LBW | ||||||
| Outcome | N =157 | Neonatal Sepsis | N =146 | ||||||||
| Survived | 107 (68) | 4 (20) | 27 (59) | 76 (83) | 0.001 | Yes | 67 (46) | 7 (44) | 23 (54) | 37 (43) | 0.490 |
| Died | 50 (32) | 16 (80) | 19 (41) | 15 (17) | No | 79 (54) | 9 (56) | 20 (46) | 50 (57) | ||
| Gender | N =166 | Congenital Abnormalities | N =128 | ||||||||
| Male | 86 (52) | 13 (65) | 27 (59) | 46 (46) | 0.164 | Yes | 5 (4) | 1 (7) | 3 (59) | 1 (46) | 0.101 |
| Female | 80 (48) | 7 (35) | 19 (41) | 54 (54) | No | 123 (96) | 13 (93) | 29 (41) | 82 (54) | ||
| Gestational Age | N =164 | Respiratory Support Required | N =163 | ||||||||
| = 32 weeks | 80 (49) | 19 (100) | 35 (75) | 26 (27) | 0.000 | Yes | 103 (63) | 16 (80) | 39 (85) | 48 (49) | 0.000 |
| 33-36 weeks | 68 (42) | 0 (0) | 9 (19) | 59 (60) | No | 60 (37) | 4 (20) | 7 (15) | 49 (51) | ||
| = 37 weeks | 16 (9) | 0 (0) | 3 (6) | 13 (13) | |||||||
| Recurrent Hypothermia | N =149 | ||||||||||
| Antenatal Clinic | N =161 | Yes | 79 (53) | 11 (58) | 30 (71) | 38 (43) | 0.009 | ||||
| Yes | 141 (88) | 15 (75) | 35 (78) | 91 (95) | 0.007 | No | 70 (47) | 8 (42) | 12 (29) | 50 (57) | |
| No | 20 (12) | 5 (25) | 10 (22) | 5 (5) | |||||||
| Recurrent Hypoglycaemia | N =150 | ||||||||||
| Mode of Delivery | N =167 | Yes | 30 (20) | 1 (5) | 16 (39) | 13 (50) | 0.001 | ||||
| Vaginal | 101 (60) | 18 (90) | 33(70) | 50 (50) | 0.001 | No | 120 (80) | 18 (95) | 25 (61) | 77 (86) | |
| Operative | 66 (40) | 2 (10) | 14 (30) | 50 (50) | |||||||
| NNJ ± EBT†2 or Phototherapy | N =123 | ||||||||||
| Place of Birth | N =166 | Yes | 24 (19) | 1 (8) | 9 (26) | 14 (18) | 0.352 | ||||
| In born | 94 (57) | 10 (50) | 26 (57) | 58 (58) | 0.805 | No | 99 (81) | 11 (92) | 25 (74) | 63 (82) | |
| Out born | 72 (43) | 10 (50) | 20 (43) | 42 (42) | |||||||
| Necrotizing Enterocolitis | N =136 | ||||||||||
| Weight for Gestational age | N =152 | Yes | 18 (13) | 2 (15) | 6 (18) | 10 (11) | 0.626 | ||||
| Appropriate for Age (AGA) | 51 (34) | 8 (40) | 14 (33) | 29 (33) | 0.807 | No | 118 (87) | 11 (85) | 28 (87) | 79 (89) | |
| Small for Age (SGA) | 101 (66) | 12 (60) | 29 (67) | 60 (67) | |||||||
| Neonatal Malaria | N =138 | ||||||||||
| Birth Asphyxia | N =156 | Yes | 38 (27) | 1 (6) | 8 (24) | 29 (33) | 0.074 | ||||
| Yes | 33 (21) | 7 (41) | 6 (14) | 20 (21) | 0.071 | No | 100 (73) | 15 (94) | 26 (76) | 59 (67) | |
| No | 123 (79) | 10 (59) | 36 (86) | 77 (79) | |||||||
| Anaemia ± Blood Transfusion | N =150 | ||||||||||
| Recurrent Apnoea | N =158 | Yes | 32 (21) | 4 (22) | 12 (32) | 16 (17) | 0.145 | ||||
| Yes | 50 (32) | 16 (84) | 20 (47) | 14 (15) | 0.001 | No | 118 (79) | 14 (78) | 25 (68) | 79 (83) | |
| No | 108 (68) | 3 (16) | 23 (53) | 82 (85) | |||||||
†1Bold values of P are statistically significant;
†2Neonatal Jaundice with or without Exchange Blood Transfusion
Table 1: Description of enrolled Low-birth weight new-borns and associated co-morbidities diagnosed.
Of the 167 low birth newborns enrolled, 20 (12.0%) had weights <1 kg (ELBW), 47 (28.1%) had weights ≥ 1 kg but less than 1.5 kg (VLBW) while 100 (59.9%) had weights between ≥ 1.5- 2.5 kg (LBW). All the ELBW babies were born at ≤ 32 weeks gestational age compared to 75% of VLBW and 27% of LBW babies delivered at ≤ 32 weeks (P=0.001). Only three quarters of mothers of ELBW babies attended antenatal care in contrast to 78% for VLBW babies and 90% for LBW babies (P=0.007). Caesarean section was the mode of delivery for 10% of ELBW, 30% of VLBW and 50% of the LBW babies (P=0.001). There was no significant differences in the proportion of ELBW babies (40% vs. 60%), VLBW (33% vs. 67%) and LBW newborns (33% vs. 67%) that were appropriate (AGA) or small for gestational age (SGA), P=0.806. Finally, the proportion of ELBWs, VLBWs and LBWs newborns delivered within and outside the hospital was not significantly different (P=0.805).
Co-morbidities and mortality seen among enrolled newborns
Eleven co-morbidities were secondarily diagnosed in the low birth weight babies enrolled in this study. These included birth asphyxia 33/156 (21%), recurrent apnoea 50/158 (32%), neonatal sepsis 67/146 (46%), and neonatal jaundice 24/123 (19%). Others included neonatal malaria 38/138 (27%), anaemia 32/150 (21%), congenital malformations 5/128 (4%), recurrent hypoglycaemia 30/150 (20%), recurrent hypothermia 79/149 (53%) and meconium aspiration syndrome 1/130 (0.8%).
Recurrent apnoeic attacks occurred significantly more in ELBW (84%) and VLBW (47%) babies compared to the LBW babies (15%), P=0.001. Similarly, more ELBW (80%) and VLBW (85%) newborns needed respiratory support compared to the LBW newborns (49%; P=0.001). It was also noted that hypothermia occurred less in LBW (43%) compared to 71% in VLBW and 58% in ELBW newborns while hypoglycaemia was more in LBW compared to VLBW and ELBW (50% vs. 39% vs. 5%, P=0.001). The incidence of other co-morbidities such as birth asphyxia (P=0.071), neonatal sepsis (P=0.490), congenital malformations (P=0.101), neonatal jaundice (P=0.352), necrotizing enterocolitis, NEC (P=0.626), neonatal malaria (P=0.074) and anaemia (P=0.145) were not significantly different
Using a 2-level logistic regression model, it was noted that only gestational age, recurrent apnoea and hypoglycaemia retained significant association with extent of low birth weight [Table 2]. Infants delivered before 37 completed weeks were 14 times more likely to be ELBW compared to those delivered after 37 completed weeks [OR 14.56, CI 1.04-203; P=0.046]. Additionally, VLBW and LBW infants, had less recurrent episodes of apnoeas [OR 0.26, CI 0.07-0.91; P=0.036] compared to ELBW infants. Furthermore, it was noted that episodes of recurrent hypoglycaemia was less common in babies with weight ELBW compared to those with weight of ≥ 1.5 kg [OR 0.26, CI 0.07- 0.94; P=0.046].
| Variables * | Categories | Crude Odd (95% CI) |
P | Adjusted OR¶ (95% CI) |
P |
| Attended Antenatal Care | Yes vs. No† | 0.26 (0.05-1.29) | 0.099 | 0.29 (0.05-1.51) | 0.140 |
| Gestational age at delivery | < 37wks† vs. = 37 wks | 13.85 (1.12-171) | 0.041 | 14.56 (1.04-203) | 0.046 |
| Mode of delivery | Vaginal† vs. Operative | 1.90 (0.71-5.07) | 0.201 | 2.08 (0.73-5.91) | 0.171 |
| Recurrent Apnoea | Yes vs. No† | 0.37 (0.11-1.20) | 0.096 | 0.26 (0.07-0.91) | 0.036 |
| Respiratory support need | Yes vs. No† | 3.29 (1.10-9.80) | 0.032 | 2.58 (0.82-8.16) | 0.107 |
| Recurrent hypothermia | Yes† vs. No | 2.83 (1.09-7.38) | 0.033 | 1.96 (0.72-5.33) | 0.187 |
| Recurrent hypoglycaemia | Yes vs. No† | 0.424 (0.14-1.33) | 0.141 | 0.26 (0.07- 0.94) | 0.040 |
| Outcome | Died vs. Survived† | 0.457 (0.14-1.45) | 0.184 | 0.50 (0.15-1.70) | 0.268 |
*Significantly associated variables on initial analysis
† Reference category
¶Adjusted for sex, place of birth and adequacy of weight for gestational age
OR- Odds ratio and CI- Confidence Interval
Bold values of P statistically significant
Table 2: : Regression analysis of factors associated with morbidity and mortality in LBW.
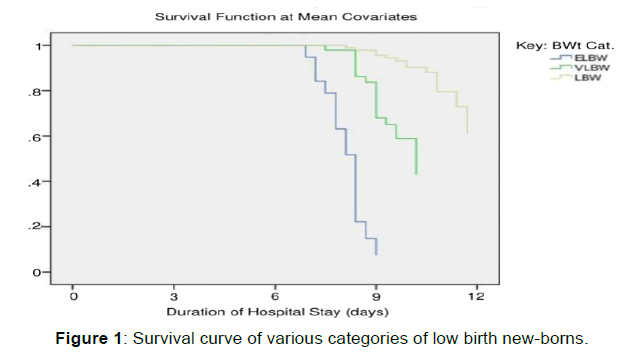
Lastly, 50/151 (32%) of the low birth weight babies admitted to the NICU during the study period did not survive. Of these number, 16/20 (80%) were ELBW babies, 19/46 (41%) were VLBW while 15/91 (17%) were LBW babies, (P=0.001). The survival graph of the babies stratified into weight categories is shown in Figure 1. ELBW and VLBW infants had 20.27 and 3.57 times more likelihood of dying compared to LBW infants, [HR 20.27, CI 5.94-69.17; P=0.001] and [HR 3.57, CI 1.59- 7.99; P=0.002]. Put differently, the chance of survival was 0.05 and 0.28 less likely in EBLW and VLBW compared to LBW infants [HR 0.05, CI 0.014-0.168; P=0.001] and [HR 0.28, CI 0.125-0.629; P=0.002] respectively.
Weight of newborns, duration of hospital stay and clinical-demographic characteristics
Table 3 evaluates the mean weight and duration of hospital stay with some clinical and demographic characteristics of the low birth weight newborns. It was noted that the mean weight of the newborns that survived (1.89 ± 0.32kg) was significantly higher than that of those that died during hospital admission (1.36 ± 0.43kg, P=0.001) Expectedly, low-birth babies that survived also stayed longer in the hospital compared to those that did not (19.33 days vs. 5.88 days, P=0.001).
| Variables | Birth weight Mean± SD/kg |
Min, Max (kg) |
T-value P†1 |
Duration of Hospital Stay Mean ± SD/days |
Min, Max (days) |
T-value P†1 |
|---|---|---|---|---|---|---|
| Outcome | ||||||
| Survived | 1.89 ±0.32 | 1.0, 2.0 | 5.82 | 19.33 ±18.75 | 1, 91 | 4.36 |
| Died | 1.36 ±0.43 | 0.5, 1.9 | 0.001 | 5.88 ±8.67 | 1, 48 | 0.001 |
| Sex | ||||||
| Male | 1.62 ±0.48 | 1.0, 2.0 | 0.44 | 11.86 ±14.32 | 1, 56 | 0.03 |
| Female | 1.67 ±0.45 | 0.6, 2.0 | 0.661 | 11.80 ± 14.61 | 1, 56 | 0.974 |
| Type of LBW | ||||||
| ≤ 1.0 kg (ELBW) | 0.85 ±0.15 | 0.5, 0.9 | 206.29†2 | 2.0 ± 2.45 | 1, 8 | 2.52†2 |
| 1.0-1.5 kg (VLBW) | 1.34 ±0.13 | 1.0, 2.0 | 0.000 | 16.12 ±24.6 | 1, 91 | 0.088 |
| 1.5-2.5 kg (LBW) | 1.92 ±0.27 | 2.0, 2.5 | 11.95 ±10.17 | 1, 39 | ||
| Antenatal Care Use | ||||||
| Yes | 1.72 ±0.41 | 1.0, 2.2 | 2.91 | 14.52 ±16.67 | 1, 91 | 0.13 |
| No | 1.32 ±0.54 | 0.5, 2.0 | 0.005 | 13.94 ±20.38 | 1, 84 | 0.899 |
| Mode of Delivery | ||||||
| Vaginal | 1.53 ±0.45 | 0.5, 2.0 | 2.85 | 12.55 ±16.98 | 1, 39 | 0.53 |
| Operative | 1.84 ±0.40 | 1.0, 2.0 | 0.006 | 10.56 ±10.92 | 2, 67 | 0.600 |
| Recurrent Apnoea | ||||||
| Yes | 1.37 ±0.45 | 0.5, 2.0 | 5.12 | 12.55 ±16.98 | 1, 91 | 0.16 |
| No | 1.86 ±0.34 | 1.0, 2,0 | 0.001 | 11.58 ±10.47 | 1, 39 | 0.875 |
| Birth Asphyxia | ||||||
| Yes | 1.73 ±0.51 | 1.2, 2.4 | 0.69 | 12.33 ±16.05 | 2, 67 | 0.61 |
| No | 1.63 ±0.46 | 0.7, 1.8 | 0.496 | 9.42 ±8.63 | 1, 39 | 0.546 |
| Respiratory Support | ||||||
| Needed | 1.53 ±0.46 | 0.6, 1.9 | 2.75 | 12.65 ±17.40 | 1, 73 | 0.58 |
| Not needed | 1.83 ±0.41 | 1.0, 2.0 | 0.008 | 10.46 ±10.07 | 1, 32 | 0.562 |
| Hypothermia | ||||||
| Recurrent | 1.58 ±0.44 | 0.8, 1.5 | 1.39 | 12.31 ±18.55 | 3, 91 | 0.28 |
| 1-2 episodes | 1.72 ±0.47 | 0.5, 2.0 | 0.202 | 11.30 ±10.10 | 2, 39 | 0.782 |
| Hypoglycaemia | ||||||
| Recurrent | 1.51 ±0.33 | 0.5, 2.0 | 0.86 | 27.33 ±30.23 | 1, 91 | 3.59 |
| 1-2 episodes | 1.657 ±0.47 | 1.0, 2.0 | 0.393 | 9.50 ±9.67 | 1, 69 | 0.006 |
| NNJ ± EBT/Photo†3 | ||||||
| Yes | 1.79 ±0.42 | 1.0, 2.0 | 1.36 | 14.95 ±8.94 | 1, 39 | 0.57 |
| No | 1.61 ±0.46 | 0.6, 2,0 | 0.180 | 12.70 ±17.33 | 2, 67 | 0.569 |
| NEC†4 | ||||||
| Yes | 1.23 ±0.56 | 0.9, 2.0 | 2.08 | 28.83 ±31.78 | 4, 91 | 3.08 |
| No | 1.66 ±0.48 | 0.5, 2.0 | 0.041 | 10.21 ±11.62 | 1, 56 | 0.003 |
| Neonatal Malaria | ||||||
| Yes | 1.79 ±0.34 | 1.2, 2.2 | 1.39 | 26.38 ±22.35 | 3, 89 | 5.21 |
| No | 1.61 ±0.48 | 1.0, 2.0 | 0.174 | 7.43 ±8.11 | 1, 30 | 0.001 |
| Anaemia ± BT†5 | ||||||
| Yes | 1.66 ±0.33 | 1.2, 2.1 | 0.07 | 26.6 ±22.68 | 1, 39 | 5.02 |
| No | 1.65 ±0.49 | 1.0, 2.0 | 0.941 | 7.72 ±8.62 | 2, 67 | 0.001 |
| Neonatal Sepsis | ||||||
| Yes | 1.62 ±0.47 | 0.6, 1.9 | 0.40 | 19.35 ±19.70 | 6, 91 | 3.49 |
| No | 1.65 ±0.43 | 1.0, 2,4 | 0.689 | 7.28 ±8.80 | 1, 33 | 0.008 |
| Place of Birth | ||||||
| In born | 1.65 ±0.45 | 1.0, 2.0 | 0.39 | 8.13 ±10.33 | 1, 56 | 2.42 |
| Out born | 1.62 ±0.45 | 0.6, 1.9 | 0.692 | 16.35 ± 18.45 | 3, 72 | 0.018 |
†1Bold values of P are statistically significant
†2Analysis of Variance (ANOVA)
†3Neonatal Jaundice with or without Exchange Blood Transfusion or Phototherapy
†4Necrotizing Enterocolitis
†5Blood Transfusion
Table 3: The mean weight and duration of hospital stay among LBW neonates.
It was further noted that low-birth weight babies born outside the hospital (16.35 days vs. 8.13 days, P=0.018), and those that had co-morbidities such as one or more episodes of hypoglycaemia (27.33 days vs. 9.50 days, P=0.001), NEC (28.83 days vs. 10.21 days, P=0.003) malaria (26.38 days vs. 7.43 days, P=0.001), anaemia with or without blood transfusion (26.62 days vs. 7.72 days, P=0.001) and neonatal sepsis (19.35 days vs. 7.28 days, P=0.008) had a significantly longer stay in the hospital compared to those without these morbidities.
The mean weight of the ELBW, VLBW and the LBW babies were 0.85 ± 0.15, 1.34 ± 0.13 and 1.92 ± 0.27 kg respectively (P=0.001). Newborn babies whose mother attended antenatal care (1.72 ± 0.41) and those that were delivered through caesarean section (1.84 ± 0.40) had a significantly higher mean birth weight compared low birth weight newborns whose mothers did not attend antenatal care (1.32 ± 0.54; P=0.005) and those delivered via spontaneous vaginal deliveries (1.53 ± 0.45; P=0.006).
Finally, it was observed that only recurrent apnoea and NEC was significantly associated with birth weight of newborns. Infants with recurrent episodes of apnoea (1.37±0.45 vs. 1.86±0.34; P=0.001) and those with NEC (1.23 ± 0.56 vs. 1.66 ± 0.48; P=0.041) had significantly lower birth weights compared to newborns without these co-morbidities.
Discussion
Eleven co-morbidities were identified in the low-birth weight babies enrolled in this study. Recurrent hypothermia and neonatal sepsis occurred most frequently. This is not surprising because these LBW newborns are particularly susceptible to heat loss immediately after birth as a result of a high body surface area–to–body weight ratio, decreased brown fat stores, nonkeratinized skin, and decreased glycogen store. [7] Hypothermia may also lead to hypoglycemia and apnea which were also identified as co-morbidities in our study. De et al. [8] in a study of blood glucose levels in normal and low birth weight newborns reported an overall incidence of hypoglycemia of 32% and noted that hypoglycemia was significantly greater in SGA and preterm compared to AGA and term newborns respectively. In our study, the fact that the lower birth weight babies (VLBW and ELBW) were more likely to receive prolonged glucose containing intravenous fluid could explain the lower incidence of hypoglycemia in the former categories of newborns.
Apnoea of prematurity which is defined as cessation of respiratory activity of more than 20 seconds, with or without bradycardia or cyanosis was understandably commoner in infants with extremely low birth weight. [7] Janvier et al. in their study found that recurrent apnoea during hospitalization in LBW infants was associated with a worse outcome. This finding they attributed to the multiple ischemic brain injuries caused by recurrent hypoxic and bradycardic spells. [9]
A third of the low birth weight babies surveyed did not survive and significant majorities were ELBW babies. Several studies have reported that the low-birth-weight infant is at much higher risk of mortality than the infant with normal weight at birth. [3,10] A meta-analysis by Laswell et al. indicated that VLBW infants and very preterm infants have increased odds of death when not born in level III hospitals. [11] This is in keeping with experience from both developed and developing countries. It has clearly been shown that appropriate care of LBW infants, including their feeding, temperature maintenance, hygienic cord and skin care, and early detection and treatment of infections and complications including respiratory distress syndrome can substantially reduce mortality in these infants. [12]
The finding that babies that survived stayed longer than those that died was not unexpected. The major co-morbidities identified in this study ordinarily are capable of causing death within a short period of time. However, when they survive they are usually admitted for periods ranging days to week to properly treat such co-morbidities except in cases where parents request for discharge against medical advice. The authors have in a previous study reported that more than fifty percent of all newborn death in ESUTH occurred with 24 hours of admission reinforcing the present finding that babies who died spent less time in the hospital than those that survived.[13]
It was also seen in our study that babies delivered through caesarean section and those whose mother attended ANC had significantly higher birth weights compared to babies delivered vaginally and whose mother did not attend ANC. This is also not surprising because it is a common practice during antenatal visits to educate pregnant mothers on healthy behaviours in pregnancy to enhance their general wellbeing as well as the growth of their unborn babies. Such behaviours include but not limited to adequate diet and medications in pregnancy that often times help in adequate fetal weight gain.
Finally, it was shown in this study that recurrent apnoea and NEC occurred more in babies with lower birth weights. Immaturity of the respiratory center and the gut which is a common feature of this group of infants have long been identified as a common causal factor of apnoea and NEC in preterm babies. [14,15] Additionally, a systematic review of NEC in very low birth weight by Bhoomika et al. stated that the risk of NEC is inversely proportional to gestational age and the weight at birth. [16]
Limitations
Firstly, gestational age in some respondents that could not recall their last menstrual period and without any objective proof was estimated using the New Ballard Score. This scoring system is prone to observational errors with reduced accuracy after 48 hours of life. Secondly, estimation of APGAR score for the diagnosis of asphyxia was not feasible for some babies especially those delivered outside the study center by traditional birth attendants. In such cases we resorted to history at birth which is subject to recall bias. Furthermore, even though we tried to minimize measurement error by taking two readings of birth weights, observer’s error could still have resulted in some error leading to inaccuracies in classification. Finally, for weights measurement 1-2 days after birth, weight gain or loss during these window periods (though physiologically minimal) could also have led to classification error.
Conclusion
In summary, the paper concludes that the overall case fatality of low-birth weight infants in our center is high (32%) with fatality rate as high as 80% in infants ELBW, 41% in VLBW infants and 17% in those with LBW. The ELBW and VLBW infants were about 20 and four times respectively more likely to have a fatal outcome compared to LBW infants. We also conclude that the incidences of recurrent apnoea and hypoglycaemia were significantly determined by the extent of low weight in infants and infants that developed necrotizing enterocolitis (NEC) had lower mean weights. Finally, of the co-morbidities seen in this study only recurrent hypoglycaemia, necrotizing enterocolitis, malaria, anaemia and neonatal sepsis caused significant lengthy stay in the hospital. We recommend further studies that would look into factors that would enhance survival in the different sub-categories of low birth weight newborns.
Conflict of Interest
All authors disclose that there was no conflict of interest.
REFERENCES
- World Health Organization, International statistical classification of diseases and related health problems, tenth revision, World Health Organization, Geneva, 1992.
- World Health Organization 2006. Feto-maternal nutrition and low birth weight. Kramer MS. Determinants of Low Birth Weight: Methodological assessment and meta-analysis, Bulletin of the World Health Organization, 1987; 65: 663-737.
- Olu DS. The birth weight of Nigerian babies. J Trop Pediatr 2005; 41: 140-141
- Ugboma H, Onyearugha CN. Low birthweight delivery: Prevalence and associated factors as seen at a tertiary health facility. Niger J Clin Pract 2013; 16: 184-7.
- Onyiriuka AN. Trends in incidence of delivery of low birth weight infants in Benin City, southern Nigeria. Niger Postgrad Med J. 2006; 13: 189-194.
- Subramanian KNS, Barton AM, Montazami S, Seo SC. Extremely Low Birth Weight Infant. Pediatrics: Cardiac Disease and Critical Care Medicine. A publication of Medscape.
- De AK, Biswas R, Samanta M, Kundu CK. Study of blood glucose level in normal and low birth weight newborns and impact of early breast feeding in a Tertiary Care Centre. Ann Nigerian Med 2011; 5: 53-58.
- Janvier A, Khairy M, Kokkotis A, Cormier C, Messmer D, Barrington KJ. Apnea is associated with neuro-developmental impairment in very low birth weight infants. J Perinatol. 2004; 24: 763-768.
- McCormick MC. The contribution of low birth weight to infant mortality and childhood morbidity. N Engl J Med. 1985; 312: 82-90.
- Lasswell SM, Barfield WD, Rochat RW, Blackmon L. Perinatal regionalization for very low-birth-weight and very preterm infants: a meta-analysis. 2010; 304: 992-1000
- WHO. Care of the preterm and/or low-birth-weight newborn.
- Ekwochi U, Ndu IK, Nwokoye IC, Ezenwosu OU, Amadi OF, Osuorah D. pattern of morbidity and mortality of newborns admitted into the sick and special care baby unit of Enugu State University Teaching Hospital, Enugu State. Niger J Clin. Pract.2014; 17: 346-351
- Darnall RA, Ariagno RL, Kinney HC. The late preterm infant and the control of breathing, sleep, and brainstem development: a review. Clin Perinatol. 2006; 33: 883-914.
- Nanthakumar NN, Fusunyan RD, Sanderson I, Walker WA. Inflammation in the developing human intestine: a possible pathophysiologic contribution to necrotizing enterocolities,†proceedings of National Academy of Sciences of United States of America. 2000; 97: 6043-6048.
- Patel BK, Shah JS. Necrotising enterocolitis in very low birth weight infants: A systematic review. Gastroenterol. 2012.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.