Unresolving Short Stature in a Possible Case of Mucopolysccharidosis
- *Corresponding Author:
- Dr. Adaeze C Ayuk
Department of Pediatrics, University of Nigeria Teaching Hospital, Enugu, Enugu State, Nigeria.
E-mail: adaraymond@yahoo.com
Citation: Ayuk AC, Obu AO, Ughasoro MD, Ibeziako NS. Unresolving short stature in a possible case of mucopolysccharidosis.Ann Med Health Sci Res 2014;4:38-42.
Abstract
We present a metabolic disorder with main complaints of unresolving short stature following prolonged treatment for rickets. ES is a 4‑year‑old male who first presented to our hospital on self‑referral but had been seen previously at another tertiary health facility. The complaints were a swelling on the back and poor growth since 1 year of age with associated skeletal deformities: Chest wall, wrists, knees and ankle joints, which were progressive. Examination revealed a severely stunted child with a large head and caput quadratum, craniofacial disproportion, coarse facial features, saddle‑shaped nose, thick lips and bilateral corneal clouding/opacities. He had very poor language development for his age. His diagnoses based on clinical and radiological assessment was in keeping with Hurlers type of mucopolysaccharidoses. We highlight this case to emphasize the need for early consideration of other possible rare differential diagnoses in metabolic conditions in children.
Keywords
Child, Metabolic, Mucopolysaccharidoses, Short stature
Introduction
Mucopolysaccharidoses (MPS) are lysosomal storage disorders, each due to an inherited deficiency of a lysosomal enzyme required for glycosaminoglycan (GAG, acid mucopolysaccharides) degradation.[1,2] Distended lysosomes accumulate in the cell, interfere with cell function, and lead to a characteristic pattern of clinical, radiologic, and biochemical abnormalities. MPS occurs in 3.5‑4.5/100,000.[1] Skeletal dysplasia (dysostosis multiplex) leading to short stature is a major clinical presentation in all types of MPS.[1‑3] Spot tests for increased urinary GAG fragments excretion are useful for initial evaluation. Chemical quantification of uronic acid‑containing substances is required to assess the total urinary excretion of GAG. Quantitative analysis of single GAGs by various methods, or of oligosaccharides by tandem mass spectrometry, reveals type‑specific profiles.[1] In resource‑poor countries where these tests may not be available,[4] a high clinical acumen and index of suspicion is required, to be able to differentiate it from other metabolic disorders that may also present with short stature. This case attempts to highlight the important clinical and radiological findings that are useful in making a plausible diagnosis in the absence of definitive advanced tests.
Case Report
The present case is about ES a 4‑year‑old male, an only child of his parents. He first presented to our children’s out‑patient department at the University of Nigeria Teaching Hospital Enugu in January 2008, on self‑referral but had been previously seen at the University of Port Harcourt Teaching Hospital Port Harcourt, Nigeria. The complaints were swelling at the back, first noted at 2 weeks of age as a bony protuberance over the mid‑spine. Other complaints were poor growth, especially of height, that parents had noted when ES was 1 year old. There were associated skeletal deformities: Chest wall, wrists, knees and ankle joints that were progressive over the next 3 years. The back swelling was non‑tender and was not increasing in size; however parents had noted the head increased in size in relation to the rest of the body.
Of note in prenatal and post natal history of ES is that mother’s pregnancy was properly supervised in a tertiary hospital and the index pregnancy was said to have lasted 11 months and culminated in an emergency caesarian section. ES cried at birth and weighed 4.2 kg. He was exclusively breast‑fed for 6 months, after which appropriate and adequate complementary feeding was started. He was also exposed appropriately to outdoor sunshine. Motor mile‑stones were noted to be delayed. The child achieved head control at 6 months and walked at 17 months. Other milestones were within normal range. The child was fully immunized.
For the current illness, medical attention was first sought at a tertiary hospital where he was assessed and the diagnosis of rickets was made. He was started on high dose oral vitamin D and calcium for 1 year with regular follow‑up visits. There was however no obvious improvement and the patient presented to our hospital.
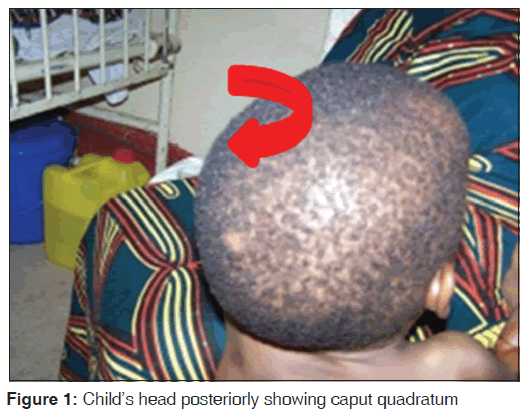
General examination revealed [Figure 1] a child who appeared short for his age with caput quadratum, craniofacial disproportion, coarse facial features, saddle shaped nose and thick lips with bilateral corneal clouding/opacity. ES was fully conscious, well‑oriented but apprehensive with limited language skills. The anterior fontanel was closed. The cranial nerve examination revealed no abnormality and the deep tendon reflexes were not exaggerated. He had a normally erupted set of teeth that were stained dark yellow. His abdomen was protuberant with an enlarged smooth non‑tender liver, 5 cm below the right costal margin but without splenomegaly. Hearing was not impaired. The cardio‑respiratory examination was normal.
The musculoskeletal system revealed a severely stunted child with an apparent large head. The weight was 13.2 kg which was above the 50th centile with a height of 80 cm, which was below 3rd centile. The occipito‑frontal circumference of 56 cm was at the upper limit of normal.
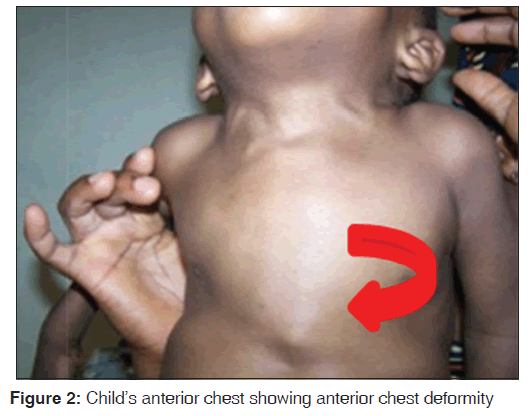
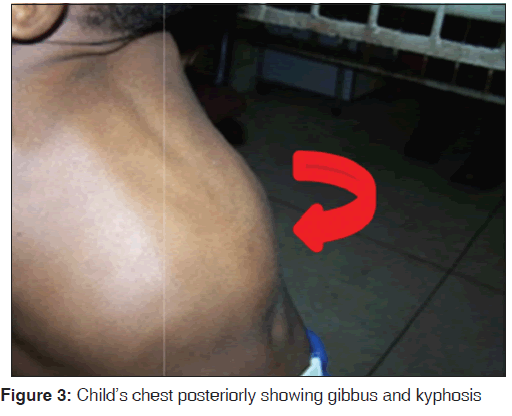
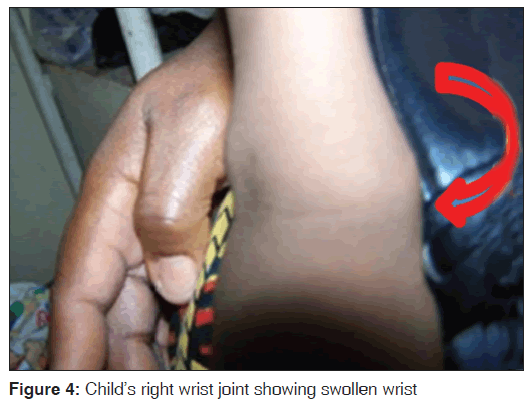
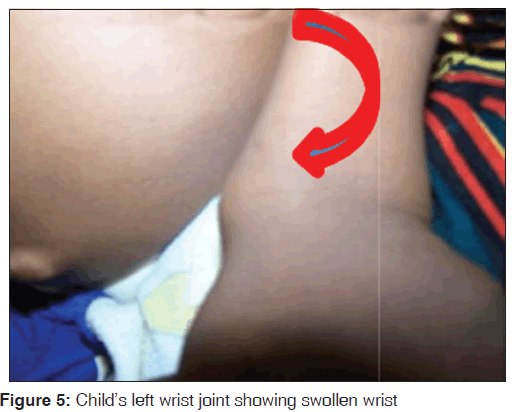
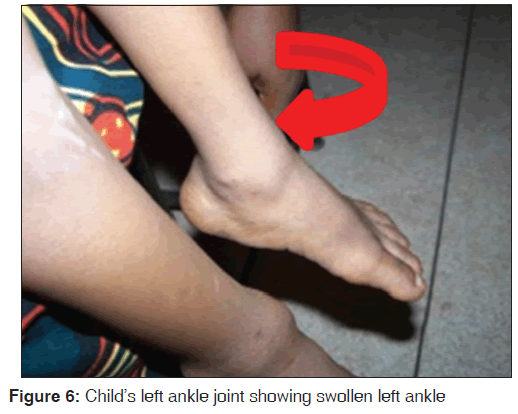
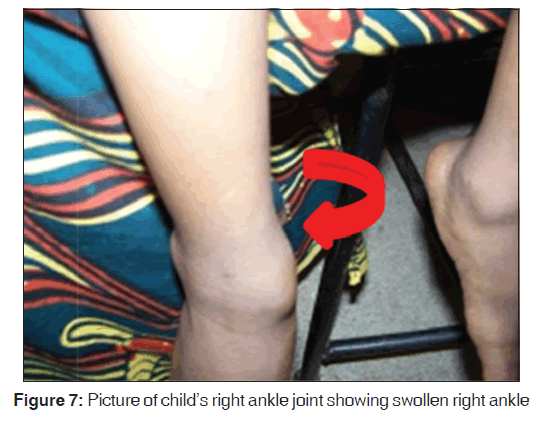
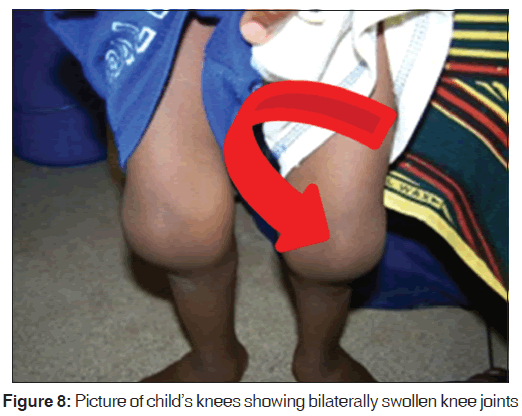
The upper to lower body segment ratio [US/LS] was 1.3. He had pectus‑carinatum [Figure 2] with Harrison sulcus but no costo‑chondral beading. Other deformities noted included bilateral swelling of wrists and ankle joints [Figures 4-7], a thoracolumbar gibbus [Figure 3], bilateral genu valgum [Figure 8]. He walked with a waddling gait.
A diagnosis of short stature of uncertain etiology was made with the following possible differentials: Vitamin Dresistant rickets, achondroplasia and MPS (probably Hurler syndrome due to the corneal clouding). An endocrine abnormality involving the thyroid gland was also considered.
Alkaline phosphatase assay done was at a level of 59 mmol, which was normal. Serial serum electrolytes, calcium and phosphate levels as well as complete blood count were also done and were within normal ranges as well.
The 24‑hr urine protein and uric acid was performed and were normal. The erythrocyte sedimentation rate was 14 mm in 1st hr westergreen; the thyroid function tests revealed a high normal T3, moderately elevated T4 and very low TSH titer. The growth hormone (hGH) was elevated at 2.3 ng (0.02‑1). The mucopolysachariduria analysis and urinary electrophoresis for GAGs were however not done due to lack of facilities.
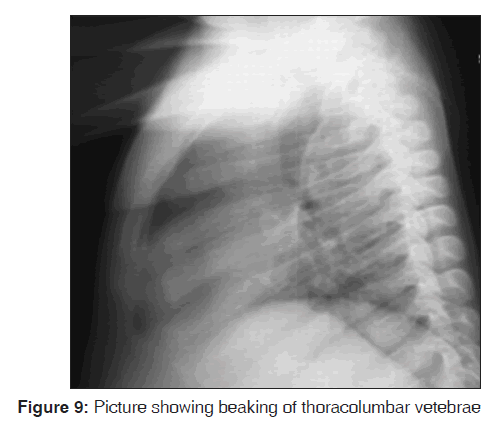
A full skeletal radiologic survey of all affected bones (skull, cervical, thoro‑columbar spines and limbs) was done and X‑ray images sent for expert radiological interpretation. This revealed the following: Osteoporotic spatulate shaped ribs which were pinched and constricted at the vertebral ends and flexed at the sternal ends [Figure 9]. The iliac bones were flared at the upper end and constricted at the acetabular ends. The acetabular angles were widened and the Menano‑Sheriton lines undisturbed. The proximal femoral epiphyses were flattened. Femoral necks were flayed and the shaft angles widened causing significant valgus deformity.
X‑ray of the radius and ulna bones revealed an acute angle, substended by the axis of both bones tilting toward each other. The 2‑5th metacarpal bones were noted to be constricted, pointing proximally with distal phalangeal hypoplasia. The spine showed localized kyphosis, hypoplasia of L1 body and vertebra. These findings were noted to be in keeping with dysostosis multiplex of MPS.
He was referred to the orthopedic surgeons for bone tissue biopsy and assay and discharged to outpatient follow‑up. He was however lost to subsequent follow‑up.
Discussion
This case highlights the importance of “thinking outside the box”, as in this case; a child thought to have rickets and was treated for 1 year. Lack of response to treatment should have prompted earlier review of possible alternative diagnoses and management.
MPS has a very low incidence; thus diagnosis is often missed. In a retrospective study in Australia,[5] between 1980 and 1996, the frequency was 1 in 22,500. The incidence in Nigeria is not known.
The clinical findings in the various types of MPS may include mental deficiency, coarse facial features, corneal clouding, viscera‑megaly, short stature, which our patient had. Joint contractures, dysostosis multiplex, leucocyte inclusions and mucopolysacchariduria are the other known features.[1,6‑10] Colville and Bax [11] found in their study that for 45% of the parents, the first symptoms noted were abnormal appearance of their children and developmental/ mental delay.
Our patient had features suggestive of Hurler disease, a form of MPS I (MPS I‑H), which is a severe, progressive disorder with multiple organ and tissue involvement that results in premature death by 10 years of age. An infant with Hurler syndrome appears normal at birth, but inguinal hernias are often present. Diagnosis is usually made between 6 and 24 months of age with evidence of hepatosplenomegaly, coarse facial features, corneal clouding, large tongue, prominent forehead, joint stiffness, short stature and skeletal dysplasia.[1,6] The patient’s first symptom was however noted by 2nd week of life which is much earlier than expected.
The patient had been managed for as long as 1 year as a case of rickets with no remarkable response to therapy. This was probably due to the skeletal abnormalities with short stature, highlighting the need for a high index of suspicion and extensive laboratory backup.
Following clinical evaluation, diagnosis of MPS requires laboratory confirmation by enzyme assay.[12]
This is not available in our center. The other investigations done including skeletal survey and the clinical findings were highly suggestive of MPS. Thyroid function tests, alkaline phosphatase and hGH assays did not conform to alternative diagnoses.
Bone marrow transplantation results in significant clinical improvement of somatic disease in MPS I, II, and VI. Clinical effects include increased life expectancy, resolution or improvement of growth failure, hepato‑splenomegaly, joint stiffness, facial appearance, pebbly skin changes in MPS II, obstructive sleep apnea, heart disease, communicating hydrocephalus, and hearing loss.[1] Enzyme replacement using recombinant enzymes is approved for patients with MPS I, MPS II, and MPS VI.[1,13] It reduces organo‑megaly, ameliorates low rate of growth and joint mobility and reduces the number of episodes of sleep apnea and urinary GAG excretion. The combination of enzyme replacement therapy and stem cell transplantation may offer the best treatment.[1] The treatment of complications by the orthopedic team presently forms the major care for this patient. Early treatment of short stature in MPS with bone marrow transplantation before the irreversible pathology was shown by Meikle et al.[4] in their study, to have a good outcome.
We could not offer our patient any of these as these are not available in our center and possibilities for referral could not be explored as the patient was loss to follow‑up. Primary prevention through genetic counseling and tertiary prevention to avoid/treat complications remains the mainstay of supportive pediatric care.[1]
Conclusion
In the absence of advanced diagnostic tests, genetic counseling/ prenatal detection that enables early and appropriate interventions, the physician’s clinical acumen becomes an invaluable tool in thinking early of alternative diagnoses, especially for such rare disorders as MPS which may mimic vitamin D deficiency.
Source of Support: Nil.
Conflict of Interest: None declared.
References
- Spranger J. Mucopolysaccharidoses. In: Berhrman RE, Kliegman RM, Jenson HB, editors. Nelson Textbook of Pediatrics. 18th ed. Ch. 88 Philadelphia, PA: Saunders Elsevier, 2007.
- Barr DG. Metabolic diseases. In: Hendrickse GR, Harrison J, editors. Pediatrics in the Tropics. Oxford: Blackwell; 1991.
521-39. - Wit JM, Rekers-Mombarg LT, Cutler GB, Crowe B, Beck TJ, Roberts K, et al. Growth hormone (GH) treatment to final height in children with idiopathic short stature: Evidence for dose effect. J Pediatr 2005;146:45-53.
- Meikle PJ, Hopwood JJ, Clague AE, Carey WF. Prevalence of lysosomal storage disorders. JAMA 1999;281:249-54.
- Kakkis ED, Muenzer J, Tiller GE, Waber L, Belmont J, Passage M, et al. Enzyme-replacement therapy in mucopolysaccharidosisI. N Engl J Med 2001;344:182-8.
- Johnson BK, Oski FA. Lysosomal storage disorder. In: Oski?s Essential Pediatrics. New York: Lippincott Publishers; 1994.
- 516-8. Mutesa L, Muganga N, Lissens W, Boemer F, Schoos R, Pierquin G, et al. Molecular analysis in two siblings African patients with severe form of Hunter Syndrome: Identification of a novel (p.Y54X) nonsense mutation. J Trop Pediatr 2007;53:434-7.
- Sly WS, Quinton BA, McAlister WH, Rimoin DL. Beta glucuronidase deficiency: Report of clinical, radiologic, and biochemical features of a new mucopolysaccharidosis. J Pediatr 1973;82:249-57.
- Muenzer JL. The mucopolysaccharidoses, a heterogenous group of disorders with variable predictive presentation. J Pediatr 2004;144:27-34.
- Nuefeid EF, Muenzer JL. The mucopolysaccharidoses. In: Beaudet AL, Sly WS, Scries CR, editors. The Metabolic and Molecular Bases of Inherited Diseases. New York: McGraw Hill; 2001. p. 3421-52.
- Colville GA, Bax MA. Early presentation in the mucopolysaccharide disorders. Child Care Health Dev 1996;22:31-6.
- Wilcox WR. Lysosomal storage disorders: The need for better pediatric recognition and comprehensive care. J Pediatr 2004;144:S3-14.
- Hein LK, Meikle PJ, Dean CJ, Bockmann MR, Auclair D, Hopwood JJ, et al. Development of an assay for the detection of mucopolysaccharidosis type VI patients using dried blood-spots. Clin Chim Acta 2005;353:67-74.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.