Use of an Innovative Simple Method for Anaerobiosis in the Diagnosis and Management of Infections in Two Unusual Cases
- *Corresponding Author:
- Prof. Maiti PK
Department of Microbiology, Institute of Post Graduate Medical Education and Research, 244, AJC Bose Road, Kolkata - 700 020, West Bengal, India.
E-mail: pkmaitiipgmer@gmail.com
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
Citation Haldar J, Pal N, Ray R, Maiti PK. Use of an innovative simple method for anaerobiosis in the diagnosis and management of infections in two unusual cases. Ann Med Health Sci Res 2016;6:56-8.
Abstract
Technical limitations restrict routine anaerobe isolation from clinical materials in resource‑limited laboratories. An innovative two steps combustion candle jar technique may be suitable for such setup. This system was tried with one case of chronic osteomyelitis developed on supracondyler compound fracture. Porphyromonas spp. was isolated and identified. Vancomycin was recommended based on in vitro sensitivity test, but the leg was amputed after receiving a resistant drug gentamycin. While in another child with hydrocephalous, V‑P shunt associated infection by Peptostreptococcus anaerobius was successfully controlled by sensitive drug vancomycin. These two eye‑opener cases insisted us for large scale application of the technique.
Keywords
Anaerobiosis, Candle-jar technique, Osteomyelitis, Porphyromonas spp, V-P shunt infection
Introduction
The available methods for anaerobiosis are costly and cumbersome. That is why empirical treatment of anaerobic infections has become a routine practice[1] in most centers, often resulting drug abuses, drug resistance development, and fatal outcome. A simple, cost-effective method of anaerobiosis may contribute toward the rational treatment of such infections for resource-limited centers. A candle jar system for rapid combustion coupled with acidified steel-wool for purging most part of residual oxygen, has been developed for the purpose.[2] Applying this technique in two different cases of anaerobic infections we timely identified which became our eye-opener cases for effective management.
Case Reports
Case 1
A 62-year-old male patient sustained a fall injury in 2012, resulting in right supracondylar compound fracture of femur and patella. He was treated outside with K-wire fixation. However, it did not heal completely even after 6 months. Patient attended the orthopedic department at our institution. Radiological examination showed features of osteomyelitis of the lower end of the right femur. The patient was otherwise healthy. His hematological parameters and blood sugar, urea, and creatinine level were within normal limits. The HIV status was seronegative. Sample from the wound showed plenty of pus cells and few Gram-negative bacilli, but no acid-fast organism. There was the growth of Pseudomonas aeruginosa in aerobic culture. The patient was treated with debridement and gentamicin bead implantation in accordance with the sensitivity pattern of the organism, which resulted in no improvement. A very offensive smell of the wound also persisted. In the absence of conventional anaerobic culture set-up first time, we tried to perform isolation, identification, and susceptibility test for anaerobes by a user-friendly method recently developed in our laboratory.[2]
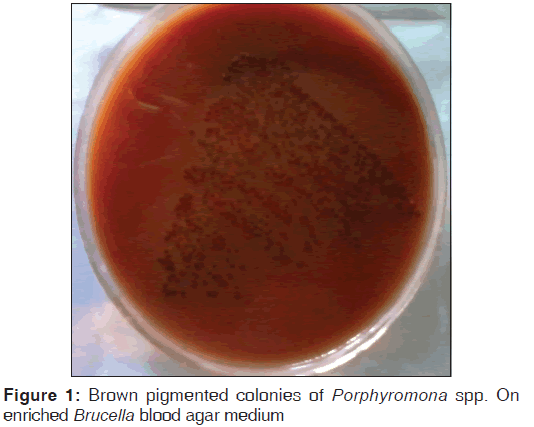
After 72 h of incubation, brown-black pigmented colonies of Gram-negative bacilli were noted in enriched blood-agar media [Figure 1] which failed to grow aerobically. Biochemical tests[3] were done in anaerobic investigation medium, which showed indole test positive, esculin hydrolysis negative, and glucose nonfermenter. Assessment of susceptibility to special potency identification discs was performed by disc diffusion method using the same candle jar technique. The organism was sensitive to vancomycin, whereas it was resistant to kanamycin and colistin. Isolate was provisionally identified as Porphyromonas spp.
By E-strip susceptibility testing [4] when interpreted according to CLSI 2013 standard for Bacteroides spp., this was sensitive to vancomycin, Penicillin G and metronidazole while resistant to kanamycin, gentamicin, cefoxitin, and colistin.
Unfortunately, the limb was amputed without waiting for in vitro susceptibility result, due to uncontrolled infection and intolerable suffering. This could be avoided in the presence of suitable laboratory back-up for routine anaerobic culture.
Case 2
A 4-year-old male child was admitted to our Neurosurgery Department for management of hydrocephalous. His developmental milestones were grossly delayed; body weight was 17 kg, head circumference 102 cm with anterior fontanelle open. There was downgaze palsy without meningeal signs. Fundoscopy examination showed optic disc pallor, but no papilledema. Other systemic examinations were within normal limits. The patient was afebrile on admission. Aqueductal stenosis with gross obstructive hydrocephalus was noted by magnetic resonance imaging. The patient was operated for implantation of V-P shunt which had to remove after 2 days for blockage. Re-implantation of V-P shunt was done, and the cerebrospinal fluid (CSF) sample was sent for microbiological investigation which was sterile at that time. The patient developed a fever from second postoperative days. Gram-stain examination of CSF sample showed Gram-positive cocci in chains with plenty of pus cells but aerobic culture showed no growth after 48 h of incubation in blood agar plate. Another CSF sample was collected through V-P shunt and processed at bedside by our technique for anaerobic culture. Growth after 48 h incubation was examined and found Gram-positive, short chain cocci. The organism failed to grow on aero-tolerance testing. Isolate was indole test negative glucose, sucrose, and maltose fermenter, whereas lactose and cellobiose nonfermenter. It was sensitive to vancomycin (5 μg) while resistant to kanamycin (1000 μg) and colistin (10 μg) by special potency disc test. Also, it showed susceptible to sodium polyanethol sulfonate disc, giving inhibition zone of 13 mm and was provisionally identified as Peptostreptococcus anaerobius. By E-test method this was sensitive to vancomycin, penicillin, cefoxitin and metronidazole and resistant to gentamycin and levofloxacin. Blood culture showed no growth both aerobically and anaerobically. After 2 weeks of parenteral vancomycin therapy, a third CSF sample was collected, which showed no growth either in aerobic or anaerobic culture, and the patient improved clinically.
Discussion
Successful isolation of anaerobes from clinical materials requires quick inoculation in prereduced media, prompt incubation as well as the use of a device ensuring early attainment of critical low oxygen level in incubation system. In modified candle-jar system, first step combustion approximately consumes 90% of atmospheric oxygen instantaneously almost at zero cost, creating vacuum equivalent to about 15% air with the addition of another 4% carbon-dioxide. Residual oxygen is brought down in next step to a critical level by use of acidified copper coated steel-wool. After standardization, the technique was applied for testing of clinical materials. Two anaerobic pathogens, Porphyromonas spp. and P. anaerobius were isolated from a case of orthopedic injury and V-P shunt infections, respectively, which prompted us for wide scale application of the innovative technique.[5]
The spectrum of infections[6] with Porphyromonas spp. includes pulmonary, intracranial, gastrointestinal infections, bacteremia (mostly of oropharyngeal origin), head and neck infections, osteomyelitis, uro-genital, and skin or soft tissue infections. In one case, Porphyromonas gingivalis was isolated from osteomyelitis of the ulna.[7] However in this case, the organism could not be obtained in cultures from intra-operative specimen but was identified by polymerase chain reaction. So far our knowledge goes; the present orthopedic infection caused by Porphyromonas spp. is the first reported one from India.
Most CSF shunt infections are caused by normal flora of the skin. A variety of microorganisms are responsible for these infections, but Staphylococcus epidermidis are the most common infecting agent.[8] However, isolation of anaerobes from clinical specimens is difficult due to technical limitations.[9] In some studies, Clostridium perfringens have been isolated from several infants with ventriculoperitoneal shunts.[10]
Financial support and sponsorship
Department of Science and Technology, Government of West Bengal, India.
Conflicts of interest
There are no conflicts of interest.
References
- Bharadwaj R. Anaerobic microbiology: Time to rejuvenate. Indian J Med Microbiol 2012;30:3-5.
- Maiti PK, Haldar J, Mukherjee P, Dey R. Anaerobic culture on growth efficient bi-layered culture plate in a modified candle jar using a rapid and slow combustion system. Indian J Med Microbiol 2013;31:173-6.
- Sutter VL, Citron DM, Edelstein MA, Finegold SM. Wadsworth Anaerobic Bacteriology Manual. 2nd ed. Belmont, California: Star Publishing Company; 1986.
- Citron DM, Ostovari MI, Karlsson A, Goldstein EJ. Evaluation of the E test for susceptibility testing of anaerobic bacteria. J Clin Microbiol 1991;29:2197-203.
- Haldar J, Mukherjee P, Mukhopadhyay S, Maiti PK. Isolation of bacteria from diabetic foot ulcers with special reference to anaerobe isolation by simple modified candle jar technique. Indian J Med Res 2016;143. [Accepted for publication: 10.02.2015].
- Finegold SM. Anaerobic Bacteria in Disease. Orlando, FL: Academic Press; 1977.
- Welkerling H, Geissdörfer W, Aigner T, Forst R. Osteomyelitis of the ulna caused by Porphyromonas gingivalis. J Clin Microbiol 2006;44:3835-7.
- Shurtleff DB, Foltz EL, Weeks RD, Loeser J. Therapy of Staphylococcus epidermidis: Infections associated with cerebrospinal fluid shunts. Pediatrics 1974;53:55-62.
- Arank A, Syed SA, Kenney EB, Freter R. Isolation of anaerobic bacteria from human gingiva and mouse cecum by means of a simplified glove box procedure. Appl Microbiol 1969;17:568-76.
- Borowsky AD, Stein SM, Tulipan NB, Dermody TS. Meningitis covered by anaerobic species complicating Tethered cord syndrome. Clin Infect Dis 1995;21:706-7.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.