Vitamin D, Immune System and Its Relationship with Diseases
2 Department of Nutrition and Dietetics, Istanbul Medipol University, Istanbul, Turkey
Received: 04-May-2022, Manuscript No. AMHSR-22-62949; Editor assigned: 07-May-2022, Pre QC No. AMHSR-22-62949(PQ); Reviewed: 21-May-2022 QC No. AMHSR-22-62949; Revised: 04-Jul-2022, Manuscript No. AMHSR-22-62949(R); Published: 11-Jul-2022
Citation: Sanliera N, et al. Vitamin D, Immune System and Its Relationship with Diseases. Ann Med Health Sci Res. 2022;12:1-11.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Objective: Vitamin D is classi ied as an immunomodulatory hormone synthesized on skin exposure to sunlight. It is known to come into play during the regulation of hormone secretion, immune functions, cell proliferation, and differentiation. Its de iciency can cause plentiful diseases along with their associated pleiotropic effects. In this direction, the available line of research as to the effects of vitamin D absorption, metabolism, health effects/bene its, effects/health bene its, de iciency of vitamin D, VDR, and 25- hydroxyvitamin D was reviewed.
Methods: The literature review was conducted through selected websites, involving Medline, Embase, Web of Science, Cochrane Central, PubMed, Science Direct, Google Scholar, EFSA, plus the World Health Organization.
Results: The mentioned aspects, vis-àvis its eminent function toward adaptive immune response and innate immune response, vitamin D levels are ascribed to immune tolerance. Vitamin D is found effective fort he regulation of hormone secretion, immune functions, cell proliferation along with differentiation. Its role as an immune modulator is based on the presence of receptors on a good number of immune cells and the synthesis of its active metabolite from these cells. Vitamin D, an immune system modulator, inhibits cell proliferation and stimulates cell differentiation.
Conclusion: There is not enough safety data to be able to give appropriate advice to individuals through a risk bene it analysis regarding the potential advantages of vitamin D against possible risks. To get the most bene its for health from vitamin D, more studies are needed on individual consumption and the relation of dose response.
Keywords
Diseases; Health; Immune system; Vitamin D
Introduction
Vitamin D is the sole vitamin that acts as a hormone and is synthesized in the skin through sunlight exposure. Only 10-20% of the daily requirement of vitamin D can be met by dietary intake. Vitamin D deficiency has pleiotropic effects on the human body and is attributed to related health issues and diseases such as bone and dental health, cardiovascular disease, hypertension, some types of cancer, type 1,2 diabetes, obesity, multiple sclerosis, cognitive decline, dementia, depression, rheumatoid arthritis, allergy, frailty, infectious diseases, autism. The association with these diseases suggests that vitamin D is a fundamental immune modulator. Vitamin D turns out basically effective during the regulation of hormone secretion, immune functions, cell proliferation and differentiation. Its immunomodulatory role depends on the presence of receptors on numerous immune cells and the synthesis of its active metabolite from immune cells. The Vitamin D Receptor (VDR) together with metabolizing together with metabolizing enzymes is expressed by a variety of immune cells, lymphocytes, monocytes, macrophages, and dendritic cells to name a few (DC). It responds to innate and adaptive immunity by providing immunomodulation of monocytes, macrophages, Dendritic Cells (DC) as well as T and B lymphocytes. 1, 25 (OH) 2D acts as a strategic agent in the regulation of immune system homeostasis [1-5].
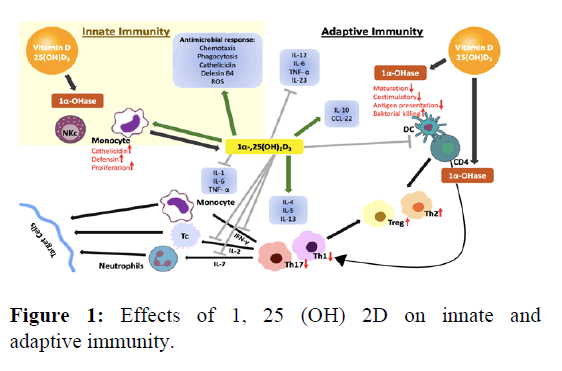
Although vitamin D suppresses the activation of the adaptive immune system, it activates the innate immune system, in particular monocytes and macrophages. Vitamin D, an immune system modulator, inhibits cell proliferation and stimulates cell differentiation. Thusly, whilst providing innate immunity support for antimicrobial functions in the immune system, it reduces inflammatory activity and the capacity to initiate an adaptive immune response. The major impacts belonging to vitamin D regarding the regulation of immune functions are known as the presence of VDR in active inflammatory cells as accompanied by the ability of the active form of vitamin D to inhibit T cell proliferation. Vitamin D boosts anti-inflammatory cytokine levels (IL-4, IL-5, IL-10, TGF-beta) via stimulating T Helper (Th) 2 cells on the inflammatory system and by inhibiting Th1 and Th17 cells, proinflammatory cytokines (IL-2, IL- 3, Inflame, TNFalpha) production. Added, 1, 25 (OH) 2D has an inhibitory effect on the maturation of dendritic cells by preventing the dissimilitude of B cell precursors into plasma cells. CD4 T cells can transform into regulatory (Treg) and suppressor T cells alongside Th1 and Th2 cells. Active vitamin D elevates the conversion of CD4 T cells into treg cells. In cases where there exists no vitamin D, the number and activity of Treg diminish, and the incidence of autoimmune diseases escalates. It is also underpinned to have anti-infective properties against infectious diseases and there is an interaction between the host immune response against pathogens and vitamin D signaling in response to bacterial infection, VDR and 1-alpha hydroxylase activity increase in monocytes and macrophages. Thus, a serious resistance is formed against intracellular microorganisms such as mycobacteria. It has been divulged that through providing immunomodulation of monocytes, macrophages, DCs apace with T and B lymphocytes, it responds to innate and adaptive immunity. A healthy immune system is acknowledged the most important weapon against viral infections. Howbeit, when there is an irregularity in the response of immune system toward viral mechanisms, the rate of the inflammatory process rises, which leads to death. It has been indicated trough the literature that it can generate an irregular immune reaction, specifically on lymphocytes. Vitamin D is believed to raise natural immunity by increasing antimicrobial peptides, namely, cathelicidin as a response to infection [6,7]. Relatively recent research reveals that vitamin D is indeed a prominent element of the immune system since it actively takes part in the blocking of infections of several sorts also regulating the operations within the immune system. Vitamin D is in fact necessary for monocyte function. The very role of vitamin D during immune system regulation has become more established through the following discovery: VDRs are found in virtually all immune system cells, including antigen presenting cells like activated CD4+ and CD8+ T cells, B cells, neutrophils, monocytes, macrophages, and DCs. A large number of immune system cells, such as monocytes, macrophages, DC, B and T lymphocytes, have the capacity to express CYP27B1 and translate 25 (OH) D to 1,25 (OH) 2D. Vitamin D is crucial for the production of AMPs locally through the 1, 25 (OH) 2D/VDR complexes against infection. The linkage of monocytes and vitamin D is realized by CYP27B1 enzyme. Monocytes perform immune reactions through phagocytizing foreign body in conjunction with the use of Toll-Like Receptors (TLRs) coupled with pattern recognition receptors pertaining to some other classes with a view to recognizing foreign presence [8-10]. It has been proved that there is an elevation of CYP27B1 activity when this event occurs. The very situation pinpoints an increase in 1, 25 (OH) 2D, which is locally produced, that binds up to the endogenous VDR and monitors gene expression in monocytes. VDR-driven innate immunity is mediated by CYP27B1 through the stimulation of TLR1/2 Pathogen-Recognizing Receptors (PRR) and the activation of TLR-4 ligand Lipo Poly Saccharides (LPS). Pathogens are identified by Pathogen-Associated Molecular Patterns (PAMPs) that bind to these PRRs. Vitamin D participates in the differentiation of monocytes into macrophages with the addition of natural immunity by increasing the phagocytic capacity of macrophages. It creates a natural immune response by fighting pathogens with TLR2 and TLR4 inhibition. In light of the aforementioned situations, the relevant information italicizes the momentousness of vitamin D while protecting the organism against pathogens. Considering the highly significant function of Vitamin D for both adaptive immune response and innate immune response, vitamin D level is referred to as immune tolerance. These effects on immune cells may explain the beneficial effect of vitamin D observed against several autoimmune diseases. However, although vitamin D supplementation has beneficial effects on curtailing the severity of disease activity, there is no consensus on the dose to be used. More randomized controlled trials are needed so as to evaluate clinical efficacy, duration of treatment, type, and dose of the compound to be administered. In this context, the current review intends to cast light on the possible influence of vitamin D on the immune system and its relationship with the existing literature. Impacts of 1, 25 (OH) 2D on innate in tandem with adaptive immunity is outlined in (Figure 1).
Materials and Methods
Literature review belonging to the period prior to May 2021 was conducted through selected websites, involving the Medline, Embassy, Web of Science, Cochrane Central, www.ClinicalTrials.gov, PubMed, Science Direct, Google Scholar, EFSA, plus the World Health Organization (WHO). Reference articles were searched and reviewed by the following keywords: Vitamin D absorption, metabolism, health effects/benefits, effects/health benefits, vitamin D deficiency, VDR, 25-hydroxyvitamin D; insulin; diabetes mellitus obesity, immunity, infection, immune system, autoimmune diseases, autoimmune thyroid diseases, multiple sclerosis, inflammatory bowel diseases, autoimmune rheumatic diseases, rheumatoid arthritis, psoriasis, infectious diseases, COVID 19 and vitamin D, vitamin D replacement therapy. The same keywords are resorted to by means of combinations, disease names, using gene, polymorphism, and other immune disease names. Sub-references of the articles were selected according to the keywords searched and these articles were also checked. Animal studies and clinical human studies on vitamin D treatments and immune diseases were primarily reviewed in this sense. These research articles, reviews, reviews of systematic nature, and metaanalyses constituted the groundwork of the present study [11-14].
Vitamin d and disease interaction
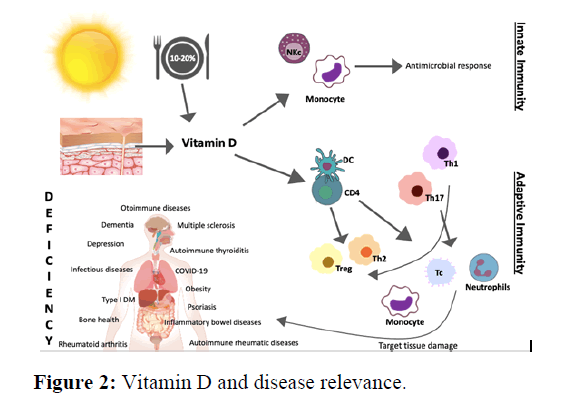
The relation between vitamin D and disease is presented in (Figure 2).
Obesity and vitamin D
Vitamin D functions during extracellular Ca concentration regulation and calcium enters into pancreatic β-cells. With this mechanism, the synthesis of vitamin D receptors besides 1-alpha-hydroxylase enzymes are triggered in β-cells that secrete insulin from the pancreas, and at the same time, insulin production and secretion are facilitated. Apart from this, since the renin-angiotensin system activity increases in cases of incremented adiposity, accordingly low adiponectin levels are detected in the adipose tissue and intensification in this regard can be achieved through vitamin D impact. High circulating levels of leptin and interleukin-6 can inhibit vitamin D synthesis. A bidirectional association is corroborated between vitamin D and obesity. It is considered that adipose tissue is metabolically active tissue and is able to coordinate vitamin D or that vitamin D can execute the aforementioned act for adipose tissue. Verily, vitamin D directly suppresses the expression of peroxisome proliferatoractivating receptor γ2, PPARγ2, which provides lipogenesis and differentiation of pre adipocytes. It has been declared that 1, 25 (OH) 2D suppresses lipogenesis and blocks pre adipocyte differentiation by stimulating insulindependent gene-2 Insight to expression. With these key factors, 1, 25 (OH) 2D can control adipose storage. Vitamin D is believed to affect obesity through cell signaling mechanism. A consistent relationship was found between increasing body mass index and decreasing serum 25 (OH) D concentrations. Vitamin D, in its active form, 1, 25 (OH) 2D, supports the synthesis of catecholamine’s by activating the gene expression of the tyrosine hydroxylase enzyme. It has also been highlighted that it may contribute to cholinergic functions by increasing the choline acetyl transferase enzyme activity, which is the key enzyme in acetylcholine synthesis. It has also been put forth that high adiposity is assigned to low serum vitamin D, and weight loss mitigates peripheral uptake and leads to an increase in vitamin D concentration. Low vitamin D level upsurges the differentiation of pre adipocytes into mature adipocytes. Mature adipocytes cannot express VDR. The vitamin D receptors are expressed in adipocytes, and aside from this, are sensitive towards the fatsoluble 1, 25 (OH) 2D vitamin. In view of the fact that vitamin D metabolites are fat-soluble, they are retained in adipose tissue. Vitamin 25 (OH) D and its conversion to inactive metabolites cause exaggerated hypovitaminosis in obese individuals [15,16].
Results and Discussion
Type 1 diabetes mellitus and vitamin D
Relevant research has hitherto predicated that vitamin D is effective in increasing insulin secretion and insulin sensitivity, and a negative relationship between low serum 25OHD levels and prediabetes, diabetes and metabolic syndrome is evident. As known, vitamin D can affect insulin resistance and β-cell function directly through the VDR and indirectly by affecting calcium homeostasis. It is stated that the presence of vitamin D dependent calcium-binding protein in pancreatic tissue coupled with the relationship between vitamin D innate and adaptive immunity are influential in this mechanism of action. The influence of vitamin D over glucose metabolism includes changes in extracellular and intracellular calcium concentrations in pancreatic β cells. Insulin secretion is considered as a calcium-dependent process that is mediated by 1, 25 (OH) 2D with the addition of Para Thyroid Hormone (PTH). It has also been punctuated that serum 25 (OH) D deficiencies and consequent increase in PTH may cause difficulties in the capacity of β cells to convert pro-insulin to insulin and diabetes may develop with apoptosis of cells. The deficiency of Vitamin D has been imputed to boost insulin resistance, to scale down insulin production and also to metabolic syndrome. It has been reported that calcitriol supplementation decreases antibodies’ serum levels and delays β-cell destruction succession. Therefore, complement through vitamin D or analogues of it become preventive instead of being curative against the disease. A systematic meta-analysis announced improvements in glycemic control indices after vitamin D supplementation in diabetic patients and supported its use as adjunctive therapy against this disease. Amongst the environmental factors considered protective against type 1 DM is the early intake of vitamin D supplementation. One other meta-analysis demonstrated type 1 DM risk was lessened in infants who were given vitamin D supplements, to a considerable extent as opposed to those who were not. The dose-response impact is also verified because those who use relatively bigger amounts of vitamin D have a diminished risk of developing type 1 DM. That being said, studies evaluating the gains out of maternal vitamin D supplementation in preventing type 1 diabetes in children could not show this relationship. In conclusion, intensifying intake of vitamin D intake during early childhood years to assure serum 25 (OH) D stay within the optimal range appears to be a guard against developing type 1 diabetes. Supplementation of vitamin D assists in controlling disease activity, if not acting curatively. With that being said, no affirmation is observed that is pertinent to the longer-term impacts of supplementation of vitamin D over morbidity or mortality speaking of patients with type 1 diabetes. In recent years, genes mixed up in vitamin D metabolism have also attracted attention due to their relationship with vitamin D deficiency. To cite an example, susceptibility to type 2 diabetes was documented to increase with the VDR gene. Other than that, vitamin D deficiency is found to be linked to microvascular complications of diabetes such as neuropathy and retinopathy [17,18].
Sjogren’s syndrome and vitamin D
Sjogren's syndrome is an autoimmune disease that usually impacts the exocrine glands and glandular manifestations in around half of the related patients. Vitamin D levels, in lowered forms, were detected in patients with sjogren's syndrome in contrast to control groups. Even so, another study found no significant difference. Although there is no consensus over the impact of vitamin D supplementation on the patients in question, supplementation is recommended as a prophylactic measure.
Autoimmune thyroiditis and vitamin D
It has been determined that there is interdependence between deficiency of vitamin D and thyroid autoimmunity. The thyroid hormone is effective in sustaining sufficient vitamin D levels, and the immunomodulatory function of vitamin D may affect the development of autoimmune thyroid disease. Therefore, it was associated with a decline in the levels of anti-thyroperoxidase antibodies, which may have a beneficial impact on these diseases, yet no meaningful correspondence was spotted with others viewed in this way. In Hashimoto's thyroiditis, cellular immunity is impaired by virtue of genetic defects in suppressor T cells. As a result of this defect, the appropriate T lymphocytes cannot suppress the other T lymphocytes i.e., the helper. Helper T lymphocytes, when activated, bond together B lymphocytes and secrete many cytokines (INF-γ). Activated B lymphocytes construct antibodies that respond with thyroid antigens. Low serum 25 (OH) D level is spotted in persons with Hashimoto's thyroiditis. Low serum 25 (OH) D level is fastened to high ant thyroid antibody level, thyroid functions with abnormal characteristics, augmented thyroid volume combined with raised TSH level. Nevertheless, a weak-to-non-relationship between low vitamin D levels and thyroid autoimmunity has been marked. In a study, low levels of serum 25 (OH) D levels were correlated negatively with serum anti-thyroid peroxidase antibodies in thyroid patients having Hashimoto's thyroiditis. Following a 4 months supplementation of vitamin D, serum anti-TPO levels in patients with 25 (OH) D levels of 30 ng/mL curtailed in considerable fashion [19].
Multiple sclerosis and vitamin D
Multiple Sclerosis (MS) is categorized as an autoimmune Central Nervous System (CNS) disease in the literature, which is detailed through inflammation, demyelination, with axon damage. MS is a chronic disease. Whereas some patients suffer from attacks, for some the disease is witnessed progressively. Vitamin D in low levels has been allotted to a higher risk of disease development and reoccurrence. Positive effects have been unearthed for vitamin D supplementation as long as doses ranged between 500 and 2000 IU/day, and on top of that, a fall in optic neuritis to go with the rate of recurrence. Nonetheless, higher doses, that is, 5000-10.000 IU/ day, have been denounced for worse outcomes. Supplementation of vitamin D in combination with interferon β has been identified to produce a synergistic beneficial effect. In (2006) work it has been shared that a 41.0% decrement in MS risk for every 20 ng/mL (50 nmol/L) an incensement in serum 25 (OH) D levels above 24 ng/Ml. It has been signaled that females consuming more than 400 IU of vitamin D daily have 41.0% attrition in the risk of MS development. Thereupon, vitamin D deficiency is believed to take over an essential role in developing dysregulated T helper cells, CTL, NK cells, B cells, which cause central nervous system auto inflammation that harms neurons and oligodendrocytes inherent in MS.
Inflammatory bowel disease and vitamin D
The deficiency of vitamin D has been assigned to the start of intestinal diseases portrayed with progressive chronic inflammation of the gastrointestinal tract. In Crohn's disease, vitamin D has been found to inhibit the production and Th17 T helper lymphocyte subpopulations and inflammatory cytokines in the gastrointestinal tract, deplete inflammation, and protect the gut microbiota that is of crucial importance in the functioning of the mucosal immune system. Intestinal homeostasis has been imputed to VDR expression that restricts the production of IL-6 by epithelial cells. In truth, vitamin D supplementation is an influential and safe treatment at doses that are to be evaluated through case-by-case. VDR polymorphisms have been displayed in Inflammatory Bowel Diseases (IBD). The most known variations in IBD are BsmI, FokI, TaqI, ApaI polymorphisms. Nevertheless, polymorphism may differ according to races. It is clear through the bulk of literature that patients with IBD are more inclined toward vitamin D deficiency and that these patients have a greater risk of developing osteomalacia, osteoporosis and fragility fractures. For they are not capable of efficiently forming micelles and chylomicrons that allows for the absorption of vitamin D in their gastrointestinal tract. To this end, patients need to be screened for vitamin D deficiency. It is underlined that they can be treated with greater doses of vitamin D to attain a normal serum 25 (OH) D level of at least 30 ng/mL (75 nmol/ L). This points to that the relationship between vitamin D status and IBD may be of bidirectional character. It has been accentuated that persons residing in lower latitudes have a consistently low risk of developing IBD compared to the ones in high latitudes. The maximum quartile of estimated serum levels of 25 (OH) D has been published to be associated with a 46% decreased risk of crohn's disease and a 35.0% decreased risk of ulcerative colitis. Another meta-analysis study unveiled that vitamin D supplementation in IBD patients was connected to an alleviated relapse rate, backing up the therapeutic role of vitamin D as a collateral therapy for IBD. Patients with IBD are not able to absorb vitamin D effectively and hence their case entails 2-3 times higher doses of vitamin D supplementation to reach normal serum 25 (OH) D levels. Sufficing vitamin D supplementation in IBD is recognized as an adjunctive immunomodulatory agent unfolded that not only limits the risk of osteoporosis, osteomalacia, as well as fragility fractures, but also one that improves disease activity.
Autoimmune rheumatic diseases and vitamin D
Vitamin D deficiency is commonly perceived in patients with autoimmune rheumatic disease comprising more than 100 inflammatory, degenerative, and autoimmune diseases and is condemned for joint damage, severe pain, disability, and death. De la Torre Lossa (2020) promulgated that vitamin D deficiency is more common among individuals with rheumatoid arthritis and can foster its beginning or advancement. Mateen viewed that these patients were with lower calcidiol and higher inflammatory cytokine levels. Calcidiol is discovered to no longer able to perform its immunomodulatory role at depleted concentrations and that the heightening of cytokines results in the augmentation of disease severity. Notwithstanding, in spite of an improvement in disease activity, findings to date have appeared not sufficient to expound the immunomodulatory role of vitamin D to the full extent.
Rheumatoid arthritis and vitamin D
An inverse relationship was found between serum 25 (OH) D and 1, 25 (OH) 2D levels and disease activity, severity, as well as functional disability in patients with Rheumatoid Arthritis (RA). In a prospective cohort study, an inverse correlation was recognized between the risk to develop RA and vitamin D intake. The most common mutations associated with RA are the FokI, BsmI, TaqI and ApaI polymorphisms in the VDR gene and rheumatoid arthritis. It is depicted with the production of several cytokines, including TNFα, IL-6, IL-15, IL-17, and IL-1β. Vitamin D is of utmost importance for the etiopathogenesis of RA; by inhibiting Th1 cells, it can attenuate the production of IL-1, IL-6, IL-17 and TNF-α and the release of IL-2 and IFN-γ from CD4 cells. Inhibiting IFN- γ secretion down regulates NF-κB and ultimately decreases IL-12 synthesis. Th1 cells and Th17 cells are both rudimentary in the pathogenesis of experimental arthritis.
Psoriasis and vitamin D
Psoriasis is classified as a chronic inflammatory autoimmune disease distinguished by hyper proliferation of VDRexpressing keratinocytes. Amon remarked low serum vitamin D levels in patients with psoriasis and caught low vitamin D concentrations in 44.0% of them. Topical treatment using the vitamin D analog calcitriol can adjust the expression of proinflammatory cytokines (eg, TNF-α, IFN-γ, IL-2 and IL-8) as well as psoriasis with coebnericin, proteins which potentiate reactions of inflammatory nature in psoriasis. Conversely, a rise in IL-10, an anti-inflammatory cytokine inhibiting proinflammatory cytokine synthesis by T lymphocytes and macrophages, is produced Similarly, in another study it was ascertained that the calcitriol analog maxacalcitol lowered psoriasiform inflammation of the skin via giving rise to T-regulatory cells and curbing the production of IL-23 and IL-17, cytokines that are foundational for psoriasis, in the company of other diseases. It has been pointed out that serum 25 (OH) D deficiencies is a part of the known independent risk factors for psoriasis. The investigation has confirmed a relatively high prevalence of the deficiency of vitamin D amongst patients with psoriasis in contrast to the general population, regardless of the presence of succeeding adjustment for confounders in a multivariate analysis. Patients with psoriasis emerge as the ones who are less likely to expose themselves to sunlight which is the basic resource for vitamin Don the flip side; treated vitamin D deficiency may be beneficial.
Infectious diseases and vitamin D
Epidemiological data enable to connect vitamin D deficiency to defective functioning of the immune system with proliferated risk of infection as well as susceptibility to autoimmune disease. Relationships have been disclosed between 25 (OH) D deficiency and enlarged risk of infection with mycobacterium tuberculosis in parallel with respiratory tract infections, specifically in the case of infection. Another study validated that vitamin D supplementation was protective against acute respiratory tract infections in a population with 25 (OH) D deficiency, particularly those who received daily or weekly supplementation. The mechanism through which vitamin D forms a shield against respiratory infections is derived from in vitro research showing that 1, 25 (OH) 2D leads to uplifted cathelicidin expression, regulation of cytokine release, and suppression of the adaptive response via inflating innate immunity. Results of the study pointed out that vitamin D, along with antibiotics in children as well as adults had no additional positive impact for the treatment of acute bacterial pneumonia, but children had low baseline 25 (OH) D levels. As vitamin D has a considerable influence over macrophages, great efforts have been made to bind vitamin D to tuberculosis. 25 (OH) D deficiency has been confirmed to lift the risk of active tuberculosis development. Reasons assumed herein are that 1, 25 (OH) 2D induces the activation of macrophages and increased mycobactericidal activity by induction of cAMP and DEFB4. Also, adding vitamin D supplementation to anti-tuberculosis therapy has turned out to have a positive effect. Even in Chronic Obstructive Pulmonary Disease (COPD), patients with COPD have been attested to be more likely to suffer from 25 (OH) D deficiencies than compatible healthy smokers, with degeneration of COPD classification and a further disease related exacerbation rate.
COVID-19 and vitamin D
Severe Acute Respiratory Syndrome Coronavirus-2 (SARSCoV- 2) is an enveloped, single-stranded Ribo Nucleic Acid (RNA) virus. Coronavirus Disease (COVID-19) as an outcome of SARS-CoV-2 was first chronicled in Wuhan, China in late December 2019 and has rapidly been spread across the world then. COVID-19 can yield serious infections, pneumonia, kidney failure and death. Whereas the function of vitamin D has been asserted as paramount with the inspection of the pathophysiology of COVID-19, a lot is yet to be learned concerning its role of it in preventing the infection in conjunction with lethality. Vitamin D supports cellular immunity by reducing the cytokine storm experienced as part of the disease. The emergence of vitamin D in relation to COVID-19 is chiefly related to its effect on Th cell response. In case of viral or bacterial infection, the immune system responds by releasing anti-inflammatory and pro-inflammatory cytokines. The cytokine storm taking place with the excessive secretion of these cytokines is related with the severity of COVID-19 and is illustrated as an exigent cause of COVID-19 mortality. Vitamin D decreases the Th1 response and increases the Th2 and regulatory response. Thence, while the release of proinflammatory cytokines decreases the release of anti-inflammatory cytokines is increased. It has been delineated that vitamin D can prevent cytokine storm and acute respiratory distress syndrome with this regulatory effect on the immune system. As another mechanism, it has been proposed that vitamin D may reduce the severity of COVID-19 via rising up the expression of Angiotensin-Converting Enzyme 2 (ACE-2) while reducing pulmonary vasoconstriction. In a study, a relationship was encountered between vitamin D status and COVID-19 mortality. Although randomized controlled trials and research with large populations evaluating serum vitamin D levels and the severity of COVID-19 still waiting to be completed, there is proof that is worthy of sharing that points to the fact that vitamin D positively affects the very course of the disease. SARS-CoV-2 initially employs immune avoidance mechanisms, which in some patients are associated with elevated pro-inflammatory cytokine release, boosted risk of pneumonia. Sepsis and acute respiratory distress syndrome, often resulting in death. While many factors become functional when determining the outcome of COVID-19 patients, serum vitamin D levels have been determined to be correlated with disease incidence as well as mortality. Conditions associated with vitamin D deficiency, that is to say, diabetes and hypertension may be indirectly associated with the severity of COVID-19 Retrospective, multicenter research put forwards that COVID-19 patients with vitamin D deficiency had poorer outcomes overall, while those with higher vitamin D levels had better outcomes. Rhodes et al. (2021) punctuated that there is strong evidence to relate vitamin D deficiency to the severity of COVID-19 infection. Another study noted that African Americans with vitamin D deficiency and those with more severe COVID-19 disease may benefit from supplementation. In a smaller cohort observational study placed in Singapore, 43 COVID-19 patients receiving combined oral doses of vitamin D, Mg, and vitamin B12 showed critical protective effects against clinical worsening, even after adjustments made as regards age, sex, and comorbidities. Low vitamin D levels have also been measured in patients with pre-existing disease with severe COVID-19. In Belgium, a retrospective observational study with 186 positive cases and 2717 negative controls found significantly lower serum vitamin D levels in COVID-19 patients in comparison to control subjects. That said, there is not sufficient evidence to ensure there exists a link between serum vitamin D levels and COVID-19 severity and mortality, and randomized controlled and large-scale cohort research is required to test the mentioned hypothesis.
Vitamin D and its relationship with other diseases
In addition to activating the antimicrobial defense system in the body, vitamin D has some anti-inflammatory activities. In monocytes and macrophages, IL-1β, IL-6, IL-8 and TNF have a pivotal effect on reducing the production of proinflammatory mediators and regulating the host inflammatory response against pathogens. Vitamin D performs its biological activity through VDR and it has been proclaimed that the induced VDR negatively regulates NF-κB activation and reduces the inflammatory response What is more, it is accepted that the seal I polyformism in vitamin D receptors prevents the formation of anti-inflammatory signals originating from vitamin D and predisposes patients to respiratory syncytial virus-associated bronchiolitis. A similar anti-inflammatory effect has been recorded for lung epithelial cells infected with the influenza virus. Vitamin D at lower levels has also drawn a parallel with lifelong immune system development in humans, respiratory infections, wheezing, transfer of Human Immunodeficiency Virus (HIV) in infants, bacterial vaginosis in pregnancy. More recently data have been published with respect to the potential function that vitamin D holds towards an increased resistance to HIV infection. Exclusively, HIV-exposed seronegative patients produced more cAMP in the oral mucosa and peripheral blood and had higher CYP24A1 mRNA in the vaginal mucosa. CYP24A1 is indicative of high levels of 1, 25 (OH) 2D. Low serum vitamin D has been linked with HIV/AIDS progression and mortality. Vitamin D can modulate the innate immune system, together with improving the phagocytic ability on immune cells, and strengthen the physical barrier role that epithelial cells carry, substantially 1, 25 (OH) 2D, corneal and intestinal epithelial barrier function. Data on nonmycobacterial infections in humans are also correlated with urinary and respiratory infections and sepsis. This hints at the susceptibility to urinary tract infection in children who have low vitamin D levels owing to decreased cAMP production and defense of β2. Likewise, cAMP levels and other antimicrobial peptides in patients with COPD have been connected to a widened risk of acute exacerbations. Commensuration with the abovementioned data, treatment with 1, 25 (OH) 2D is functional in lessening respiratory infections in asthma patients through increased cAMP expression and modulation of inflammatory cytokines. Results reflecting findings on the role of vitamin D status and vitamin D supplementation in sepsis are accessible in pediatric and adult patients: a clear role for 25 (OH) D and cAMP in pediatric patients has not been manifested yet. Whereas, in adults, 25 levels are lower (OH) D sepsis and cAMP increases, circulating vitamin D increases, and higher doses decrease inflammatory cytokines such as IL-6 and IL-1β. Patients with autoimmune hepatitis also have a high frequency of vitamin D deficiency. Serum vitamin D level was found to be low in patients with chronic liver disease and there is a negative correlation between vitamin D level and disease progression. Jointly, it has been identified that vitamin D deficiency contributes to the pathogenesis of the disease in these patients in genetic and non-genomic ways. It has been unearthed that autoimmune liver diseases are associated with BsmI and TaqI polymorphisms. The liver has an important place in the synthesis steps of vitamin D. Having said that, it is not certain whether vitamin D deficiency in chronic liver diseases is a cause or a result of disease-related events. Scholars have been convinced that vitamin D deficiency is associated with disease activity in patients with Systemic Lupus Erythematous (SLE). In these patients, photosensitivity provides less sun exposure, reduces the synthesis of vitamin D from the skin, and chronic use of corticosteroids changes vitamin D metabolism. It accredits that deficiency of vitamin D deficiency can be an impetus to the production of autoantibody raising the risk of developing autoimmune disease in patients with a genetic predisposition. Vitamin D supplementation is recommended for the prevention of SLE-related morbidity. It has been underscored that vitamin D supplementation yields integral output, to illustrate, inhibition of DC activation, suppression of IgG production by B cell and regulation of CD4 helper T cell responses (Table1).
Table 1: Vitamin D and effects on immune-related diseases. |
|||||
|---|---|---|---|---|---|
| Immunologic Diseases | Study type | Study sample | Study method | Effects of vitamin D supplementation | Conclusion |
| Obesity | Randomized placebo-controlled | 54 overweight or obese, vit. D deficient adults | Bolus oral 100,000 IU vit. D followed by 4000 IU vit.D or placebo for 16 wk | Vit. D and placebo groups did not differ in change in insulin sensitivity or first-phase insulin secretion. Results remained nonsignificant after adjustment for age, sex, percentage of body fat, sun exposure, physical activity, and dietary vit. D intake. | Vit. D supplementation would be an effective strategy for reducing diabetes risk even in vit. D deficient populations. |
| Obesity | Double-blind, Randomized placebo-controlled | 66 overweight and obese women | Oral vit. D 50,000 IU per 25 day or placebo for 3 m | The mean difference of anthropometric indices, serum Ca, 25(OH)D and serum PTH between the groups were significant. However, no significant differences in serum phosphorus between the groups were seen. | Vit. D supplementation resulted in a significant reduction in anthropometric indices in obesity and overweight women with normal vit D. serum levels. |
| Obesity | Double-blind, Randomized placebo-controlled | 192 healthy men with serum 25(OH)D levels <75 nmol/L | 20,000 IU of vit. D/wk. or placebo for 12 wk | Significant treatment effect on fasting glucose/fasting insulin ratio was detected, whereas no significant effect on the remaining outcome parameters was observed. In subgroup analyses of men with baseline serum vit. D levels <50 nmol/L, significant effect on waist circumference, waist-to-hip ratio, total body fat, and android fat was found. | In middle-aged healthy men, vit. D treatment had a negative effect on insulin sensitivity. In vit. D deficient men, vit. D has an unfavorable effect on central obesity and body composition. |
| Type I DM (Araujo et al. 2020) | Prospective, Phase II, Open trial, Pilot study | 13 adults with recent onset T1D | ASCs (1 × 106 cells/kg) and vit. D 2000 UI/d for 3 m or standard insulin therapy | After 3 months vit. D group had lower insulin requirement and HbA1c than control group. In vit. D group, 2 patients became insulin free (for 4 and 8 weeks) and all were in honeymoon after 3 months. C-peptide variations did not differ between group. | Allogenic ASCs + vit. D without immunosuppression was associated with stability of CP and unanticipated mild transient adverse events in patients with recent onset T1D. |
| Thyroid | Double-blind, Randomized placebo-controlled | 42 women with HT disease | 50 000 IU vit. D or placebo pearls weekly for 3 m | Significant reduction of anti-Tg Ab and TSH hormone in the vit. D group compared to the start of the study; however, there was no significant reduction of anti-TPO Ab in the vit. D group compared to the placebo group. No significant changes were observed in the serum levels of T3 and T4 hormones. | Vit. D supplementation can be helpful for alleviation of the disease activity in HT patients. |
| MS | Double-blind, Randomized placebo-controlled | 94 RRMS patients | 50,000 IU vit. D or placebo every five days for 3 m. | Vit. D group had a significant difference in mental health composite with placebo group. Change in health was higher in vit. D group. | Mental QOL improved significantly after taking high dose vit. D for 3 months in vit. D group relative to placebo. |
| MS | Randomized placebo-controlled | 68 persons with relapsing remitting MS | 20,000 IU vit. D or placebo weekly | Serum vit. D levels more than doubled in the vit. D group and parathyroid hormone decreased in the vit. D group compared to the placebo group at week 48 and week 96. There was no effect on bone formation as measured by procollagen type I N propertied (PINP), or on bone resorption as measured by C-terminal cross-linking telopeptide of type I collagen (CTX1). Neither PINP nor CTX1 predicted bone loss from baseline to week 96. | Vit. D supplementation does not prevent bone loss in persons with MS who are not vit. D deficient. |
| IBD | Double-blind, Randomized placebo-controlled, Pilot study | 50 mild to moderate active UC patients | 2000 or 1000 IU/d vit. D for 12 w | Serum level of hs-CRP decreased non-significantly in both groups. Serum level of TNF-α in the high dose group was reduced non-significantly. In the low dose group, a significant increase in serum TNF-α concentration was observed. After adjusting for the effect of confounders, the significance effect was disappeared. Activity of NF-κB increased in both groups while this increase was significant in the low dose group compared to the baseline, and to high dose group. After adjustment for confounders, the difference between groups remained significant. | 12 weeks supplementation with 2000 IU/day vit. D prevents from systematic inflammation, while decreasing disease activity in patients with mild to moderate active UC. |
| IBD | Double-blind, Randomized placebo-controlled, Pilot study | 90 mild to moderate UC patients | A single muscular injection of 7.5 mg vit. D or 1 mL normal saline as placebo | There was a significant increase in vit. D only in the vit. D group. Compared to placebo, vit. D had significant decreasing effects on serum TNF-α, IFN-γ, and IL12p70 levels, but it had no significant effect on serum levels of IL4 and IL10. Vit. D seems to inhibit Th1 immune responses and have no effect on Th2 responses. | The findings of this study support several in vitro studies, which suggest a therapeutic immunomodulatory potential of vit. D. |
| IBD | Double-blind, Randomized placebo-controlled, Pilot study | 34 mild to moderate UC patients | High-dose vit D at 10,000 IU or 1000 IU daily for 12 m | High-dose vit. D significantly improved vit. D levels. On an intention-to-treat basis, the rate of relapse was not significantly different between groups. In per-protocol analysis, clinical relapse of CD was less frequently observed in patients receiving a high dose. Improvement in anxiety and depression scores and a good safety profile were observed in both groups. | Oral supplementation with high-dose vit. D at 10,000 IU daily significantly improved serum vit D. levels. Rates of clinical relapse were similar between both groups. |
| IBD | Double-blind, Randomized placebo-controlled | 94 patients with CD in remission | 1200 IU vit. D or placebo daily during 12 m | Oral vit. D treatment increased serum vit. D levels. The relapse rate was lower among patients treated with vit. D. | Vit. D treatment might be effective in CD. |
| RA | Prospective, Double-blind, Randomized placebo-controlled | 59 vit. D deficient non-remission RA patients whose treatment was not expected to be changed over a 3-month period | 100,000IU vit. D ampoules or placebo | At 6 months, HAQ scores tended to be increased in the placebo group, while slightly decreased in the vit. D group. After adjusting for age, gender, season, and initial vit. D status, the between-group difference achieved statistically significance. After adjusting for age, gender, season, and initial vit. D status, there was no significant difference in the secondary criteria between the 2 groups except for ESR and CRP respectively. | Patients with RA and vit. D deficiency, high doses of vit. D resulted in a statistically significant improvement in functional disability at month 6, which, however, was clinically not relevant. |
| RA | Open-labeled, Randomized placebo-controlled | 121 patients with treatment naive early RA | Triple Disease-Modifying Anti-Rheumatic Drug (DMARD) + 500 IU vit. D + Ca or triple DMARD and Ca | Patients on the vit. D group had higher pain relief. There was no significant difference in the time taken for initial pain relief. Occurrence of hypovitaminosis D in RA patients (68.1%) is comparable to published normal Indian prevalence. There was no correlation between serum vit. D levels and disease activity. | Supplementation of 500 IU vit. D daily to previously DMARD-naïve patients with early RA along with triple DMARD therapy results in a significantly higher pain relief at the end of 3 months. |
| Psoriasis | Double-blind, Randomized placebo-controlled | 65 Psoriasis patients aged 50-84 years | 100,000 IU vit. D monthly | There were no significant differences between the groups in all of the psoriasis outcome measures. | Vit. D supplementation (100,000 IU per month) is not recommended as a treatment for mild psoriasis. |
| COVID-19 | Randomized placebo-controlled | 69 hospitalized patients with mild to moderate COVID-19 disease | 5000 IU oral vit. D or 1000 IU oral vit. D once daily for 2 w | Vit. D supplementation for 2w caused a significant increase in serum vit. D levels in the 5000 IU group only. Within-group comparisons showed a significant decrease in BMI and IL-6 levels overtime in both groups but was not clinically significant in between-group comparisons. 5000 IU group had a significantly shorter time to recovery than the 1000 IU group in resolving cough, even after adjusting for age, sex, baseline BMI, and D-dimer, and ageusia. | The use of 5000 IU vit. D as an adjuvant therapy for COVID-19 patients with suboptimal vit. D status, even for a short duration, is recommended. |
| COVID-19 | Multicenter, Double-blind, Randomized placebo-controlled | 237 hospitalized patients with moderately to severely ill COVID-19 disease | A single oral 200 000 IU of vit. D or placebo | Median length of stay was not significantly different between groups. The difference between the groups was not significant for in-hospital mortality, admission to the intensive care unit or need for mechanical ventilation. Mean serum levels of vit. D significantly increased after a single dose of vit. D. | The findings do not support the use of a high dose of vit. D for treatment of moderate to severe COVID-19. |
Conclusion
Current day signposts that deficiency of vitamin D is a compelling issue of public health. Related research advocates vitamin D has an invaluable role as an immunomodulation. While innate immunity enhances the capacity of the immune system to fight against pathogens, its effect on the modulation of the adaptive immune system is still controversial. There indeed are uncertainties about the role of vitamin D in the pathogenesis of autoimmune diseases. It is unclear if vitamin D deficiency is a cause or consequence of autoimmune diseases. At the same time, the increase in vitamin D deficiency at epidemic rates and its coincident with the increasing prevalence of autoimmune diseases support this relationship. More comprehensive studies are being carried out on the pathophysiology of diseases, immune system functioning and the effect of vitamin D.
The salient role of vitamin D in maintaining immune balance should not be overlooked. Low serum vitamin D levels are associated with many immune-related diseases, covering autoimmune disorders and infectious diseases. Studies until now have determined that sustaining a healthy serum vitamin D level is central to regulating the body's immune function. EFSA (European Food Safety Authority) publicized daily Adequate Intake (AI) as 10 mcg in 7-1-month babies and 15 mcg in all age groups 1 year and older. Clinical studies are called for to further explain the effects of vitamin D in human beings and to reveal how many these risks can be prevented by treatment in individuals on vitamin D supplementation or with deficiency of it.
Credit Authorship Contribution Statement
NS: Visualization, Investigation, Supervision, Writing-review and editing.
MGC: Methodology, Investigation, Conceptualization, Writing - original draft.
Data Availability
The following information was supplied regarding data availability: There is no raw data in this review.
Declaration of Competing Interest
The authors declare that they have no conflict of interest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Ethics Statement
The papers cited involving the use of human subjects, have been carried out in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki) for experiments involving humans. The papers cited involving animal research have been carried out in accordance with the U.K. Animals (Scientific Procedures) Act, 1986 and associated guidelines, EU Directive 2010/63/EU for animal experiments, or the National Institutes of Health guide for the care and use of Laboratory animals (NIH Publications No. 8023, revised 1978).
Author Agreement
The authors declare no competing interests.
Conflict of Interest
The authors declare that they have no conflict of interest.
References
- Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357:266‑281.
- Kidder VD. Vitamininin kardiyovasküler ve metabolik etkileri. J Clin Exp Investig. 2013:4:398-404.
- Wei R, Christakos S. Mechanisms underlying the regulation of innate and adaptive immunity by vitamin D. Nutrients. 2015;7:8251-8260.
[Crossref]
- Baeke F, Takiishi T, Korf H. Vitamin D: modulator of the immune system. Curr Opin Pharmacol. 2010;10:482-496 .
- Korf H, Decallonne B, Mathieu C. Vitamin D for infections. Curr Opin Endocrinol Diabetes Obes. 2014;21:431-436.
- Cutolo M, Paolino S, Sulli A. Vitamin D, steroid hormones, and autoimmunity. Ann Ny Acad Sci. 2014;1317:39-46.
- Şahin S, Gurgen SG, Yazar U. Vitamin D protects against hippocampal apoptosis related with seizures induced by kainic acid and pentylenetetrazol in rats. Epilepsy Res. 2019;149:107-116.
- Jayewardene R, Jeyakumar DT, Francis TV. Impact of the vitamin D deficiency on COVID-19 infection and mortality in Asian countries. Diabetes Metab Syndr Clin Res Rev. 2021;15:757-764.
- Qin B, Xu B, Ji N, Yao S, Pawlish K, Llanos AA, et al. Intake of vitamin D and calcium, sun exposure, and risk of breast cancer subtypes among black women. Am J Clin Nutr. 2020;111:396-405.
- Kamen DL, Tangpricha V. Vitamin D and molecular actions on the immune system: modulation of innate and autoimmunity. J Mol Med. 2010;88:441-450.
- Medrano M, Carrillo-Cruz E. MonteroI, Perez-Simon JA. Vitamin D: effect on haematopoiesis and immune system and clinical applications. Int J Mol Sci. 2018;19:2663.
- Prietl B, Treiber G, Pieber T, Amrein K. Vitamin D and immune function. Nutrients. 2013;5:2502–2521.
- Zhang Y, Leung DYM, Richers BN, Liu Y, Remigio LK, Riches DW, et al. Vitamin D inhibits monocyte/macrophage proinflammatory cytokine production by targeting MAPK phosphatase-1. J Immunol. 2012;188:2127–2135.
- Nurminen V, Seuter S, Carlberg C. Primary vitamin D target genes of human monocytes. Front Physiol. Front Physiol. 2019;10:194.
- Adams JS, Ren S, Liu PT, Chun RF, Lagishetty V, Gombart AF, et al. Vitamin D-directed rheostatic regulation of monocyte antibacterial responses. J Immunol. 2009;182:4289-4295.
- Sassi F, Tamone C, Amelio DP. Vitamin D: nutrient, hormone, and immunomodulator. Nutrients 2018; 10:1656.
- Bikle DD. Vitamin D metabolism, mechanism of action, and clinical applications. Chem Biol. 2014;21:319–329.
- Holick MF. Vitamin D: A millenium perspective. J Cell Biochem. 2003;88:296–307.
- Whayne TF. Vitamin D: popular cardiovascular supplement but benefit must be evaluated. Int J Angiol. 2011;20:63–72.




 The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.
The Annals of Medical and Health Sciences Research is a monthly multidisciplinary medical journal.